Abstract
Background and Objective
Immuno-oncology therapies represent a new treatment opportunity for patients affected by metastatic melanoma. The purpose of this study was to estimate the costs of immune-related adverse events (irAEs) associated with the new anti-PD1 immuno-oncology therapies, with the anti-CTLA-4 immuno-oncology therapy and with the combined therapy (CTLA4 + anti-PD1) in patients affected by metastatic melanoma.
Materials and Methods
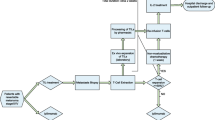
A probabilistic cost-of-illness (COI) model was developed to estimate the management costs of grade ≥ 3 adverse events associated with the new anti-PD1 therapies (pembrolizumab and nivolumab), the anti-CTLA-4 therapy (ipilimumab) and the combined therapy CTLA4 + anti-PD1 (nivolumab + ipilimumab) for the treatment of patients with metastatic melanoma from the National Health Service (NHS) perspective in Italy. Identification of the epidemiological and cost parameters was carried out through a systematic literature review (SLR). Univariate and probabilistic sensitivity analyses were performed to account for uncertainty and variation in the model results.
Results
The model estimated a cost associated with the management of grade ≥ 3 immune-related adverse events in patients with metastatic melanoma equal to €176.2 (95% CI 63.5–335.0) for anti-CTLA-4 therapy, €48.6 (95% CI 40.1–58.5) for the new anti-PDI therapies and €276.8 (95% CI 240.4–316.2) for the combined therapy. Among the innovative therapies for the considered metastatic melanoma, the combined therapy was the most expensive innovative treatment in terms of event management of immune-related grade ≥ 3 adverse events.
Conclusion
This study may represent a useful tool to understand the economic burden associated with the management of irAEs associated with patients affected by metastatic melanoma.




Similar content being viewed by others
References
Italian Oncology Association (AIOM). Immunooncology. Information for the patients. 2016. http://www.aiom.it/pazienti/informazioni-per-pazienti/1,397,1. Accessed 22 Aug 2018.
Spain L, Diem S, Larkin J. Management of toxicities of immune checkpoint inhibitors. Cancer Treat Rev. 2016;44:51–60.
Hodi FS, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363(8):711–23.
Robert C, et al. Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N Engl J Med. 2011;364(26):2517–26.
Robert C, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372(4):320–30.
Weber JS, et al. Nivolumab versus chemotherapy in patients with advanced melanoma who progressed after anti-CTLA-4 treatment (CheckMate 037): a randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2015;16(4):375–84.
Robert C, et al. Pembrolizumab versus ipilimumab in advanced melanoma. N Engl J Med. 2015;372(26):2521–32.
Ribas A, et al. Pembrolizumab versus investigator-choice chemotherapy for ipilimumab-refractory melanoma (KEYNOTE-002): a randomised, controlled, phase 2 trial. Lancet Oncol. 2015;16(8):908–18.
Larkin J, et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Engl J Med. 2015;373(1):23–34.
Zugazagoitia J, et al. Current challenges in cancer treatment. Clin Ther. 2016;38(7):1551–66.
Karlitepe A, Ozalp O, Avci CB. New approaches for cancer immunotherapy. Tumour Biol. 2015;36(6):4075–8.
Kwon ED, et al. Ipilimumab versus placebo after radiotherapy in patients with metastatic castration-resistant prostate cancer that had progressed after docetaxel chemotherapy (CA184-043): a multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol. 2014;15(7):700–12.
Eggermont AM, et al. Adjuvant ipilimumab versus placebo after complete resection of high-risk stage III melanoma (EORTC 18071): a randomised, double-blind, phase 3 trial. Lancet Oncol. 2015;16(5):522–30.
Wiater K, et al. Efficacy and safety of ipilimumab therapy in patients with metastatic melanoma: a retrospective multicenter analysis. Contemp Oncol (Pozn). 2013;17(3):257–62.
Ahmad SS, et al. Ipilimumab in the real world: the UK expanded access programme experience in previously treated advanced melanoma patients. Melanoma Res. 2015;25(5):432–42.
Ascierto PA, et al. Clinical experience with ipilimumab 3 mg/kg: real-world efficacy and safety data from an expanded access programme cohort. J Transl Med. 2014;12:116.
Jung M, et al. Ipilimumab real-world efficacy and safety in Korean melanoma patients from the Korean named-patient program cohort. Cancer Res Treat. 2017;49(1):44–53.
Del Vecchio M, et al. Efficacy and safety of ipilimumab 3 mg/kg in patients with pretreated, metastatic, mucosal melanoma. Eur J Cancer. 2014;50(1):121–7.
Chiarion Sileni V, et al. Efficacy and safety of ipilimumab in elderly patients with pretreated advanced melanoma treated at Italian centres through the expanded access programme. J Exp Clin Cancer Res. 2014;33:30.
Daly LE, et al. The impact of body composition parameters on ipilimumab toxicity and survival in patients with metastatic melanoma. Br J Cancer. 2017;116(3):310–7.
Margolin KA, et al. Effectiveness and safety of ipilimumab therapy in advanced melanoma: evidence from clinical practice sites in the US. J Community Support Oncol. 2015;13(4):131–8.
Eigentler TK, et al. Diagnosis, monitoring and management of immune-related adverse drug reactions of anti-PD-1 antibody therapy. Cancer Treat Rev. 2016;45:7–18.
Ribas A, et al. Updated clinical efficacy of the anti-PD-1 monoclonal antibody pembrolizumab in 411 patients with melanoma. Pigment Cell Melanoma Res. 2014;27(6):1222–3 (abstract).
Robert C, et al. Anti-programmed-death-receptor-1 treatment with pembrolizumab in ipilimumab-refractory advanced melanoma: a randomised dose-comparison cohort of a phase 1 trial. Lancet. 2014;384(9948):1109–17.
Ribas A, Puzanov I, Drummer REA. A randomized controlled comparison of pembrolizumab and chemotherapy in patients with ipilimumab-refractory melanoma. In: SFM research, editor. International Congress, Zurich, Switzerland. 2014.
Robert C, et al. Pembrolizumab (pembro; MK-3475) for advanced melanoma (MEL): randomized comparison of two dosing schedules. In: Annals of oncology, vol 25, no 4. 2014. https://doi.org/10.1093/annonc/mdu438.42.
Vouk K, et al. Cost and economic burden of adverse events associated with metastatic melanoma treatments in five countries. J Med Econ. 2016;19(9):900–12.
Wehler E, et al. Economic burden of toxicities associated with treating metastatic melanoma in eight countries. Eur J Health Econ. 2017;18(1):49–58.
Health Ministry Decree dated 18 October 2012, Tariffs of hospital services for acute patients, by kind of hospitalization/hospital admissions. http://www.trovanorme.salute.gov.it/norme/renderPdf.spring?seriegu=SG&datagu=28/01/2013&redaz=13A00528&artp=1&art=1&subart=1&subart1=10&vers=1&prog=001. Accessed 21 Aug 2018.
Health Ministry Decree dated 18 October 2012, Health care range of fees of specialist out-patient care. http://www.trovanorme.salute.gov.it/norme/renderPdf.spring?seriegu=SG&datagu=28/01/2013&redaz=13A00528&artp=3&art=1&subart=1&subart1=10&vers=1&prog=001. Accessed 21 Aug 2018.
Briggs A, Sculpher M, Claxton K. Decision modelling for health economic evaluation. Oxford: Oxford University Press; 2007.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Funding
This work was supported by an unrestricted grant from MSD Italia.
Rights and permissions
About this article
Cite this article
Mennini, F.S., Bini, C., Marcellusi, A. et al. Cost Estimate of Immune-Related Adverse Reactions Associated with Innovative Treatments of Metastatic Melanoma. Clin Drug Investig 38, 967–976 (2018). https://doi.org/10.1007/s40261-018-0690-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-018-0690-9