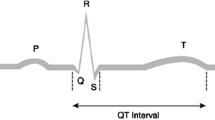
Abstract
The care and treatment of cancer patients has significantly changed in the last decade with a remarkable shift towards novel targeted therapies. These promising new drugs may represent effective and potentially life-saving therapeutic options in cancer patients, but are also emerging in the cardiotoxicity scenario for their arrhythmogenic potential due to their QT-prolonging activity. In this article we review the mechanisms underlying drug-induced QT interval prolongation and the classes of anticancer-targeted therapies most frequently responsible for this adverse event, with a particular focus on tyrosine kinase-targeting molecules. Since up to 49 % of serious adverse drug reactions (ADRs) and 58 % of potentially fatal ADRs may not appear on initial drug safety labels, we also review and discuss data from the post-marketing VigiBase® safety reporting system, the World Health Organization’s global database of ADRs. Finally, we discuss arrhythmic risk stratification and prevention strategies in the complex multiple-risk setting of cancer patients, paying particular attention to drug–drug interactions with common antimicrobial, psychotropic and antiemetic supportive care, and we also provide an electrocardiographic QT monitoring algorithm for patients who are candidates for targeted cancer therapies.


Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2014;136(5):E359–86. doi:10.1002/ijc.29210.
Suter TM, Ewer MS. Cancer drugs and the heart: importance and management. Eur Heart J. 2013;34(15):1102–11. doi:10.1093/eurheartj/ehs181.
Curigliano G, Cardinale D, Suter T, Plataniotis G, de Azambuja E, Sandri MT, ESMO Guidelines Working Group, et al. Cardiovascular toxicity induced by chemotherapy, targeted agents and radiotherapy: ESMO Clinical Practice Guidelines. Ann Oncol. 2012;23(Suppl 7):vii155–66. doi:10.1093/annonc/mds293.
Raschi E, de Ponti F. Cardiovascular toxicity of anticancer-targeted therapy: emerging issues in the era of cardio-oncology. Intern Emerg Med. 2012;7(2):113–31. doi:10.1007/s11739-011-0744-y.
Raschi E, Vasina V, Ursino MG, Boriani G, Martoni A, de Ponti F. Anticancer drugs and cardiotoxicity: insights and perspectives in the era of targeted therapy. Pharmacol Ther. 2010;125(2):196–218. doi:10.1016/j.pharmthera.2009.10.002.
Force T, Kerkelä R. Cardiotoxicity of the new cancer therapeutics–mechanisms of, and approaches to, the problem. Drug Discov Today. 2008;13(17–18):778–84. doi:10.1016/j.drudis.2008.05.011.
Albini A, Pennesi G, Donatelli F, Cammarota R, De Flora S, Noonan DM. Cardiotoxicity of anticancer drugs: the need for cardio-oncology and cardio-oncological prevention. J Natl Cancer Inst. 2010;102(1):14–25. doi:10.1093/jnci/djp440.
Eschenhagen T, Force T, Ewer MS, de Keulenaer GW, Suter TM, Anker SD, et al. Cardiovascular side effects of cancer therapies: a position statement from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2011;13(1):1–10. doi:10.1093/eurjhf/hfq213.
Lal H, Kolaja KL, Force T. Cancer genetics and the cardiotoxicity of the therapeutics. J Am Coll Cardiol. 2013;61(3):267–74. doi:10.1016/j.jacc.2012.05.066.
Hahn VS, Lenihan DJ, Ky B. Cancer therapy-induced cardiotoxicity: basic mechanisms and potential cardioprotective therapies. J Am Heart Assoc. 2014;3(2):e000665. doi:10.1161/JAHA.113.000665.
Ewer MS, Von Hoff DD, Benjamin RS. A historical perspective of anthracycline cardiotoxicity. Heart Fail Clin. 2011;7(3):363–72. doi:10.1016/j.hfc.2011.03.001.
Schwartz PJ, Crotti L, Insolia R. Long-QT syndrome: from genetics to management. Circ Arrhythm Electrophysiol. 2012;5(4):868–77. doi:10.1161/CIRCEP.111.962019.
Kapplinger JD, Tester DJ, Salisbury BA, et al. Spectrum and prevalence of mutations from the first 2,500 consecutive unrelated patients referred for the FAMILION long QT syndrome genetic test. Heart Rhythm. 2009;6(9):1297–303. doi:10.1016/j.hrthm.2009.05.021.
Roden DM, Viswanathan PC. Genetics of acquired long QT syndrome. J Clin Invest. 2005;115(8):2025–32. doi:10.1172/JCI25539.
Kannankeril P, Roden DM, Darbar D. Drug-induced long QT syndrome. Pharmacol Rev. 2010;62(4):760–81. doi:10.1124/pr.110.003723.
Mitcheson JS, Chen J, Lin M, Culberson C, Sanguinetti MC. A structural basis for drug-induced long QT syndrome. Proc Natl Acad Sci USA. 2000;97(22):12329–33. doi:10.1073/pnas.210244497.
Fernandez D, Ghanta A, Kauffman GW, Sanguinetti MC. Physicochemical features of the HERG channel drug binding site. J Biol Chem. 2004;279(11):10120–7. doi:10.1074/jbc.M310683200.
Yeung K-S, Meanwell NA. Inhibition of hERG channel trafficking: an under-explored mechanism for drug-induced QT prolongation. ChemMedChem. 2008;3(10):1501–2. doi:10.1002/cmdc.200800170.
Kolecki PF, Curry SC. Poisoning by sodium channel blocking agents. Crit Care Clin. 1997;13(4):829–48.
Niemeijer MN, Van Den Berg ME, Eijgelsheim M. Pharmacogenetics of drug-induced QT interval prolongation: an update. Drug Saf. 2015;38(10):855–67. doi:10.1007/s40264-015-0316-6.
Antzelevitch C. Cellular basis for the repolarization waves of the ECG. Ann N Y Acad Sci. 2006;1080:268–81. doi:10.1196/annals.1380.021.
Antzelevitch C. Role of spatial dispersion of repolarization in inherited and acquired sudden cardiac death syndromes. Am J Physiol Heart Circ Physiol. 2007;293(4):H2024–38. doi:10.1152/ajpheart.00355.2007.
Frommeyer G, Eckardt L. Drug-induced proarrhythmia: risk factors and electrophysiological mechanisms. Nat Rev Cardiol. 2016;13(1):36–47. doi:10.1038/nrcardio.2015.110.
de Bruyne MC, Hoes AW, Kors JA, Hofman A, van Bemmel JH, Grobbee DE. QTc dispersion predicts cardiac mortality in the elderly: the Rotterdam Study. Circulation. 1998;97(5):467–72.
Molokhia M, Pathak A, Lapeyre-Mestre M, Caturla L, Montastruc JL, McKeigue P. Case ascertainment and estimated incidence of drug-induced long-QT syndrome: study in Southwest France. Br J Clin Pharmacol. 2008;66(3):386–95. doi:10.1111/j.1365-2125.2008.03229.x.
Sarganas G, Garbe E, Klimpel A, Hering RC, Bronder E, Haverkamp W. Epidemiology of symptomatic drug-induced long QT syndrome and Torsade de Pointes in Germany. Europace. 2014;16(1):101–8. doi:10.1093/europace/eut214.
Brell JM. Prolonged QTc interval in cancer therapeutic drug development: defining arrhythmic risk in malignancy. Prog Cardiovasc Dis. 2010;53(2):164–72. doi:10.1016/j.pcad.2010.05.005.
E14 Implementation Working Group. ICH E14 Guideline: The clinical evaluation of QT/QTc interval prolongation and proarrhythmic potential for non-antiarrhythmic drugs. Questions & Answers (R2). Geneva: International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use; 2014.
Drew BJ, Ackerman MJ, Funk M, Gibler WB, Kligfield P, Menon V, American Heart Association Acute Cardiac Care Committee of the Council on Clinical Cardiology, the Council on Cardiovascular Nursing, and the American College of Cardiology Foundation, et al. Prevention of torsade de pointes in hospital settings: a scientific statement from the American Heart Association and the American College of Cardiology Foundation. Circulation. 2010;121(8):1047–60. doi:10.1161/CIRCULATIONAHA.109.192704.
4.0 C version. Common Terminology Criteria for adverse events (CTCAE). http://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-06-14_QuickReference_5x7.pdf. Accessed 22 May 2016.
Naing A, Veasey-Rodrigues H, Hong DS, Fu S, Falchook GS, Wheler JJ, et al. Electrocardiograms (ECGs) in phase I anticancer drug development: the MD Anderson Cancer Center experience with 8518 ECGs. Ann Oncol. 2012;23(11):2960–3. doi:10.1093/annonc/mds130.
Varterasian M, Meyer M, Fingert H, Radlowski D, Asbury P, Zhou X, et al. Baseline heart rate-corrected QT and eligibility for clinical trials in oncology. J Clin Oncol. 2003;21(17):3378–9. doi:10.1200/JCO.2003.99.104.
Varterasian M, Fingert H, Agin M, Meyer M. Consideration of QT/QTc interval data in a phase I study in patients with advanced cancer [letter]. Clin Cancer Res. 2004;10(17):5967–5968; author reply 5968–5969. doi:10.1158/1078-0432.CCR-04-0534.
Zeltser D, Justo D, Halkin A, Prokhorov V, Heller K, Viskin S. Torsade de pointes due to noncardiac drugs: most patients have easily identifiable risk factors. Medicine (Baltimore). 2003;82(4):282–90. doi:10.1097/01.md.0000085057.63483.9b.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–74. doi:10.1016/j.cell.2011.02.013.
Locatelli M, Criscitiello C, Esposito A, Minchella I, Goldhirsch A, Cipolla C, et al. QTc prolongation induced by targeted biotherapies used in clinical practice and under investigation: a comprehensive review. Target Oncol. 2015;10(1):27–43. doi:10.1007/s11523-014-0325-x.
Manning G, Whyte DB, Martinez R, Hunter T, Sudarsanam S. The protein kinase complement of the human genome. Science. 2002;298(5600):1912–34. doi:10.1126/science.1075762.
Druker BJ, Talpaz M, Resta DJ, Peng B, Buchdunger E, Ford JM, et al. Efficacy and safety of a specific inhibitor of the BCR-ABL tyrosine kinase in chronic myeloid leukemia. N Engl J Med. 2001;344(14):1031–7. doi:10.1056/NEJM200104053441401.
Bosutinib, BOSULIF [prescribing information]. http://www.accessdata.fda.gov/drugsatfda_docs/label/2012/203341lbl.pdf. Accessed 29 May 2016.
Ceritinib, ZYCADIA. Supplemental approval. http://www.accessdata.fda.gov/drugsatfda_docs/appletter/2015/205755Orig1s003,s004ltr.pdf. Accessed 29 May 2016.
Crizotinib, XALKORI [prescribing information]. http://www.accessdata.fda.gov/drugsatfda_docs/label/2012/202570s002lbl.pdf. Accessed 29 May 2016.
Dasatinib, SRPYCEL [prescribing information]. http://www.accessdata.fda.gov/drugsatfda_docs/label/2010/021986s7s8lbl.pdf. Accessed 29 May 2016.
Lenvatanib, LENVIMA [prescribing information]. http://www.accessdata.fda.gov/drugsatfda_docs/label/2015/206947s000lbl.pdf. Accessed 29 May 2016.
Nilotinib, TASIGNA [prescribing information]. http://www.accessdata.fda.gov/drugsatfda_docs/label/2007/022068lbl.pdf. Accessed 29 May 2016.
Osimertinib, TAGRISSO [prescribing information]. http://www.accessdata.fda.gov/drugsatfda_docs/label/2015/208065s000lbl.pdf. Accessed 29 May 2016.
Pazopanib, VOTRIENT [prescribing information]. https://www.accessdata.fda.gov/drugsatfda_docs/label/2009/022465lbl.pdf. Accessed 29 May 2016.
Ponatinib, ICLUSIG [prescribing information]. http://www.accessdata.fda.gov/drugsatfda_docs/label/2012/203469lbl.pdf. Accessed 29 May 2016.
Sorafenib, NEXAVAR [prescribing information]. http://www.accessdata.fda.gov/drugsatfda_docs/label/2010/021923s008s009lbl.pdf. Accessed 29 May 2016.
Sunitinib malate, SUTENT [prescribing information]. http://www.accessdata.fda.gov/drugsatfda_docs/label/2011/021938s13s17s18lbl.pdf. Accessed 29 May 2016.
US FDA. Vandetanib, CAPRELSA. Risk evaluation and mitigation strategy. http://www.fda.gov/downloads/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/UCM253441.pdf. Accessed 29 May 2016.
Woosley R, Romero K. QT drugs list. https://crediblemeds.org/. Accessed 22 May 2016.
Ghatalia P, Je Y, Kaymakcalan MD, Sonpavde G, Choueiri TK. QTc interval prolongation with vascular endothelial growth factor receptor tyrosine kinase inhibitors. Br J Cancer. 2015;112(2):296–305. doi:10.1038/bjc.2014.564.
Lu Z, Wu C-YC, Jiang Y-P, Ballou LM, Clausen C, Cohen IS, et al. Suppression of phosphoinositide 3-kinase signaling and alteration of multiple ion currents in drug-induced long QT syndrome. Sci Transl Med. 2012;4(131):131ra50. doi:10.1126/scitranslmed.3003623.
Shah RR, Morganroth J, Shah DR. Cardiovascular safety of tyrosine kinase inhibitors: with a special focus on cardiac repolarisation (QT Interval). Drug Saf. 2013;36(5):295–316. doi:10.1007/s40264-013-0047-5.
Jeong W, Doroshow JH, Kummar S. United States Food and Drug Administration approved oral kinase inhibitors for the treatment of malignancies. Curr Probl Cancer. 2013;37(3):110–44. doi:10.1016/j.currproblcancer.2013.06.001.
Fraczek J, Vanhaecke T, Rogiers V. Toxicological and metabolic considerations for histone deacetylase inhibitors. Expert Opin Drug Metab Toxicol. 2013;9(4):441–57. doi:10.1517/17425255.2013.754011.
Noonan AM, Eisch RA, Liewehr DJ, Sissung TM, Venzon DJ, Flagg TP, et al. Electrocardiographic studies of romidepsin demonstrate its safety and identify a potential role for KATP channel. Clin Cancer Res. 2013;19(11):3095–104. doi:10.1158/1078-0432.CCR-13-0109.
Munster PN, Rubin EH, Van Belle S, Friedman E, Patterson JK, Van Dyck K, et al. A single supratherapeutic dose of vorinostat does not prolong the QTc interval in patients with advanced cancer. Clin Cancer Res. 2009;15(22):7077–84. doi:10.1158/1078-0432.CCR-09-1214.
Siemann DW, Chaplin DJ. An update on the clinical development of drugs to disable tumor vasculature. Expert Opin Drug Discov. 2007;2(10):1357–67. doi:10.1517/17460441.2.10.1357.
McKeage MJ, Baguley BC. Disrupting established tumor blood vessels: an emerging therapeutic strategy for cancer. Cancer. 2010;116(8):1859–71. doi:10.1002/cncr.24975.
Milan A, Puglisi E, Ferrari L, Bruno G, Losano I, Veglio F. Arterial hypertension and cancer. Int J Cancer. 2014;134(10):2269–77. doi:10.1002/ijc.28334.
Dowlati A, Robertson K, Cooney M, Petros WP, Stratford M, Jesberger J, et al. A phase I pharmacokinetic and translational study of the novel vascular targeting agent combretastatin A-4 phosphate on a single-dose intravenous schedule in patients with advanced cancer. Cancer Res. 2002;62(12):3408–16.
Ibrahim MA, Do DV, Sepah YJ, Shah SM, Van Anden E, Hafiz G, et al. Vascular disrupting agent for neovascular age related macular degeneration: a pilot study of the safety and efficacy of intravenous combretastatin A-4 phosphate. BMC Pharmacol Toxicol. 2013;14(1):7. doi:10.1186/2050-6511-14-7.
Bos JL. Ras oncogenes in human cancer: a review. Cancer Res. 1989;49(17):4682–9.
Mazieres J, Pradines A, Favre G. Perspectives on farnesyl transferase inhibitors in cancer therapy. Cancer Lett. 2004;206(2):159–67. doi:10.1016/j.canlet.2003.08.033.
Roffey J, Rosse C, Linch M, Hibbert A, McDonald NQ, Parker PJ. Protein kinase C intervention-the state of play. Curr Opin Cell Biol. 2009;21(2):268–79. doi:10.1016/j.ceb.2009.01.019.
Murray NR, Kalari KR, Fields AP. Protein kinase C?? expression and oncogenic signaling mechanisms in cancer. J Cell Physiol. 2011;226(4):879–87. doi:10.1002/jcp.22463.
Kreisl TN, Kim L, Moore K, Duic P, Kotliarova S, Walling J, et al. A phase I trial of enzastaurin in patients with recurrent gliomas. Clin Cancer Res. 2009;15(10):3617–23. doi:10.1158/1078-0432.CCR-08-3071.
Seruga B, Sterling L, Wang L, Tannock IF. Reporting of serious adverse drug reactions of targeted anticancer agents in pivotal phase III clinical trials. J Clin Oncol. 2011;29(2):174–85. doi:10.1200/JCO.2010.31.9624.
World Health Organization Collaborating Centre for International Drug Monitoring. VigiAccess™. http://www.vigiaccess.org. Accessed 2 Jun 2016.
Schwartz PJ, Woosley RL. Predicting the unpredictable: drug-induced QT prolongation and Torsades de Pointes. J Am Coll Cardiol. 2016;67(13):1639–50. doi:10.1016/j.jacc.2015.12.063.
Shah RR, Morganroth J. Update on cardiovascular safety of tyrosine kinase inhibitors: with a special focus on QT interval, left ventricular dysfunction and overall risk/benefit. Drug Saf. 2015;38(8):693–710. doi:10.1007/s40264-015-0300-1.
Tuccori M, Montagnani S, Capogrosso-Sansone A, Mantarro S, Antonioli L, Fornai M, et al. Adverse reactions to oncologic drugs: spontaneous reporting and signal detection. Expert Rev Clin Pharmacol. 2015;8(1):61–75.
Antman KH. Introduction: the history of arsenic trioxide in cancer therapy. Oncologist. 2001;6(Suppl 2):1–2.
Barbey JT, Pezzullo JC, Soignet SL. Effect of arsenic trioxide on QT interval in patients with advanced malignancies. J Clin Oncol. 2003;21(19):3609–15. doi:10.1200/JCO.2003.10.009.
Abo-Salem E, Fowler JC, Attari M, Cox CD, Perez-Verdia A. Antibiotic-induced cardiac arrhythmias. Cardiovasc Ther. 2014;32(1):19–25. doi:10.1111/1755-5922.12054.
Shaffer D, Singer S, Korvick J, Honig P. Concomitant risk factors in reports of Torsades de Pointes associated with macrolide use: review of the United States Food and Drug Administration Adverse Event Reporting System. Clin Infect Dis. 2002;35(2):197–200.
Frothingham R. Rates of Torsades de Pointes associated with ciprofloxacin, ofloxacin, levofloxacin, gatifloxacin, and moxifloxacin. Pharmacotherapy. 2001;21(12):1468–72.
Niedrig D, Maechler S, Hoppe L, Corti N, Kovari H. Drug safety of macrolide and quinolone antibiotics in a tertiary care hospital: administration of interacting co-medication and QT prolongation. Eur J Clin Pharmacol. 2016;72(7):859–67. doi:10.1007/s00228-016-2043-z.
FDA. FDA drug safety communication: revised recommentations for Celexa (citalopram hydrobromide) related to a potential risk of abnormal heart rhythms with high doses. http://www.fda.gov/Drugs/DrugSafety/ucm297391.htm. Accessed 22 May 2016.
Barni S, Petrelli F, Cabiddu M. Cardiotoxicity of antiemetic drugs in oncology: an overview of the current state of the art. Crit Rev Oncol Hematol. 2016;102:125–34. doi:10.1016/j.critrevonc.2016.04.012.
Maurea N, Spallarossa P, Cadeddu C, Madonna R, Mele D, Monte I, et al. A recommended practical approach to the management of target therapy and angiogenesis inhibitors cardiotoxicity: an opinion paper of the working group on drug cardiotoxicity and cardioprotection, Italian Society of Cardiology. J Cardiovasc Med (Hagerstown). 2016;17(Suppl 1):S93–104. doi:10.2459/JCM.0000000000000383.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No source of funding was used to assist in the preparation of this manuscript.
Conflicts of interest
Rezarta Cuni, Iris Parrini, Riccardo Asteggiano and Maria Rosa Conte have no conflicts of interest to declare regarding the presented work.
Rights and permissions
About this article
Cite this article
Cuni, R., Parrini, I., Asteggiano, R. et al. Targeted Cancer Therapies and QT Interval Prolongation: Unveiling the Mechanisms Underlying Arrhythmic Complications and the Need for Risk Stratification Strategies. Clin Drug Investig 37, 121–134 (2017). https://doi.org/10.1007/s40261-016-0460-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-016-0460-5