Abstract
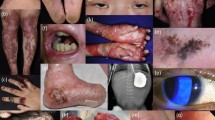
Epidermolysis bullosa (EB) represents a group of inherited blistering skin diseases, some forms of which are associated with considerable morbidity and increased mortality. Notably, in recessive dystrophic EB there can be extensive muco-cutaneous fragility and disease complications such as scars, contractures, anemia, malnutrition, and malignancy. Currently, there is no effective therapy or cure for EB. Over the last decade, however, a number of important advances have been made that are bringing new treatments closer to the clinic, including gene therapy, protein replacement therapy, cell therapies [allogeneic fibroblasts, mesenchymal stromal cells (MSCs), bone marrow stem cell transplantation, culturing/grafting revertant mosaic keratinocytes], gene editing/engineering, and clinical application of inducible pluripotent stem cells. Although a cure for EB still remains elusive, recent data on animal models and initial human clinical trials have raised the expectations of patients, clinicians, and researchers that disease modification and improved quality of life are feasible goals. Furthermore, the lessons learned in treating EB are likely to have significant implications for improving the management of other genetic diseases.



Similar content being viewed by others
References
Bruckner-Tuderman L, McGrath JA, Robinson EC, et al. Progress in Epidermolysis Bullosa Research: Summary of DEBRA International Research Conference 2012. J Invest Dermatol. 2013;133:2121–6.
Fine JD, Eady RA, Bauer EA, et al. The classification of inherited epidermolysis bullosa (EB): report of the Third International Consensus Meeting on Diagnosis and Classification of EB. J Am Acad Dermatol. 2008;58:931–50.
Fine JD, Mellerio JE. Extracutaneous manifestations and complications of inherited epidermolysis bullosa: part II. Other organs. J Am Acad Dermatol. 2009;61:387–402.
Fine JD, Mellerio JE. Extracutaneous manifestations and complications of inherited epidermolysis bullosa: part I. Epithelial associated tissues. J Am Acad Dermatol. 2009;61:367–84.
Varki R, Sadowski S, Uitto J, et al. Epidermolysis bullosa. II. Type VII collagen mutations and phenotype-genotype correlations in the dystrophic subtypes. J Med Genet. 2007;44:181–92.
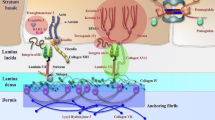
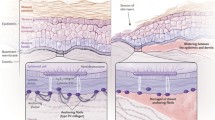
McGrath JA, Ishida-Yamamoto A, O’Grady A, et al. Structural variations in anchoring fibrils in dystrophic epidermolysis bullosa: correlation with type VII collagen expression. J Invest Dermatol. 1993;100:366–72.
Chung HJ, Uitto J. Type VII collagen: the anchoring fibril protein at fault in dystrophic epidermolysis bullosa. Dermatol Clin. 2010;28:93–105.
Mavilio F, Pellegrini G, Ferrari S, et al. Correction of junctional epidermolysis bullosa by transplantation of genetically modified epidermal stem cells. Nat Med. 2006;12:1397–402.
U.S. National Institutes of Health. Gene transfer for recessive dystrophic epidermolysis bullosa. http://clinicaltrials.gov/ct2/show/NCT01263379?term=NCT01263379&rank=1. Accessed Oct 2013.
Urnov FD, Rebar EJ, Holmes MC, et al. Genome editing with engineered zinc finger nucleases. Nat Rev Genet. 2010;11:636–46.
Carroll D. Genome engineering with zinc-finger nucleases. Genetics. 2011;188:773–82.
Osborn MJ, Starker CG, McElroy AN, et al. TALEN-based gene correction for epidermolysis bullosa. Mol Ther. 2013;21:1151–9.
Woodley DT, Keene DR, Atha T, et al. Injection of recombinant human type VII collagen restores collagen function in dystrophic epidermolysis bullosa. Nat Med. 2004;10:693–5.
Woodley DT, Wang X, Amir M, et al. Intravenously injected recombinant human type VII collagen homes to skin wounds and restores skin integrity of dystrophic epidermolysis bullosa. J Invest Dermatol. 2013;133:1910–3.
Palazzi X, Marchal T, Chabanne L, et al. Inherited dystrophic epidermolysis bullosa in inbred dogs: a spontaneous animal model for somatic gene therapy. J Invest Dermatol. 2000;115:135–7.
Cogan J, Wang XY, Hou YP, Khilili M, Woodley DT, Chen M. Epitope mapping of anti-type VII collagen antibodies in the patients with recessive dystrophic epidermolysis bullosa. J Invest Dermatol. 2013;133:S45.
Wong T, Gammon L, Liu L, et al. Potential of fibroblast cell therapy for recessive dystrophic epidermolysis bullosa. J Invest Dermatol. 2008;128:2179–89.
Nagy N, Almaani N, Tanaka A, et al. HB-EGF induces COL7A1 expression in keratinocytes and fibroblasts: possible mechanism underlying allogeneic fibroblast therapy in recessive dystrophic epidermolysis Bullosa. J Invest Dermatol. 2011;131:1771–4.
Kern JS, Loeckermann S, Fritsch A, et al. Mechanisms of fibroblast cell therapy for dystrophic epidermolysis bullosa: high stability of collagen VII favors long-term skin integrity. Mol Ther. 2009;17:1605–15.
Fritsch A, Loeckermann S, Kern JS, et al. A hypomorphic mouse model of dystrophic epidermolysis bullosa reveals mechanisms of disease and response to fibroblast therapy. J Clin Invest. 2008;118:1669–79.
Venugopal SS, Yan W, Frew JW, et al. A phase II randomized vehicle-controlled trial of intradermal allogeneic fibroblasts for recessive dystrophic epidermolysis bullosa. J Am Acad Dermatol. 2013;69:898–908.
Petrof G, Martinez-Quiepo M, Mellerio JE, et al. Fibroblast cell therapy enhances initial healing in recessive dystrophic epidermolysis bullosa wounds: results of a randomised, vehicle-controlled trial. Br J Dermatol. 2013;169:1025–33.
Conget P, Rodriguez F, Kramer S, et al. Replenishment of type VII collagen and re-epithelialization of chronically ulcerated skin after intradermal administration of allogeneic mesenchymal stromal cells in two patients with recessive dystrophic epidermolysis bullosa. Cytotherapy. 2010;12:429–31.
Tamai K, Yamazaki T, Chino T, et al. PDGFRalpha-positive cells in bone marrow are mobilized by high mobility group box 1 (HMGB1) to regenerate injured epithelia. Proc Natl Acad Sci U S A. 2011;108:6609–14.
Wagner JE, Ishida-Yamamoto A, McGrath JA, et al. Bone marrow transplantation for recessive dystrophic epidermolysis bullosa. N Engl J Med. 2010;363:629–39.
Almaani N, Nagy N, Liu L, Dopping-Hepenstal PJ, et al. Revertant mosaicism in recessive dystrophic epidermolysis bullosa. J Invest Dermatol. 2010;130:1937–40.
Jonkman MF, Scheffer H, Stulp R, et al. Revertant mosaicism in epidermolysis bullosa caused by mitotic gene conversion. Cell. 1997;88:543–51.
Lai-Cheong JE, McGrath JA. Revertant mosaicism in the skin. G Ital Dermatol Venereol. 2013;148:73–82.
Pasmooij AMG, Jonkman MF, Uitto J. Revertant mosaicism in heritable skin diseases: mechanisms of natural gene therapy. Discov Med. 2012;14:167–79.
Pasmooij AM, Nijenhuis M, Brander R, et al. Natural gene therapy may occur in all patients with generalized non-Herlitz junctional epidermolysis bullosa with COL17A1 mutations. J Invest Dermatol. 2012;132:1374–83.
Gostyński A, Llames S, García M, et al. Long-term survival of type XVII collagen revertant cells in an animal model of revertant cell therapy. J Invest Dermatol. 2013. doi: 10.1038/jid.2013.308
Gostynski A, Deviaene FC, Pasmooij AM, et al. Adhesive stripping to remove epidermis in junctional epidermolysis bullosa for revertant cell therapy. Br J Dermatol. 2009;161:444–7.
Gostynski A, Bremer J, Huizinga J, et al. Successful therapeutic transplantation of revertant skin in a patient with epidermolysis bullosa. J Invest Dermatol. 2013;133:S132.
Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–76.
Tolar J, Xia L, Riddle MJ, Lees CJ, et al. Induced pluripotent stem cells from individuals with recessive dystrophic epidermolysis bullosa. J Invest Dermatol. 2011;131:848–56.
Itoh M, Kiuru M, Cairo MS, et al. Generation of keratinocytes from normal and recessive dystrophic epidermolysis bullosa-induced pluripotent stem cells. Proc Natl Acad Sci U S A. 2011;108:8797–802.
Umegaki N, Guo Z, Itoh M, et al. Generation of induced pluripotent stem cells from revertant mosaic keratinocytes: a novel strategy for natural gene therapy for epidermolysis bullosa. J Invest Dermatol. 2013;133:S243–6.
Acknowledgments
No sources of funding were used to prepare this review. The authors have no conflicts of interest that are directly relevant to the content of this review. Original studies on cell therapy for RDEB in the McGrath laboratory were supported by the UK National Institute for Health Research (NIHR) Biomedical Research Centre based at Guy’s and St Thomas’ NHS Foundation Trust and King’s College London. The views expressed are those of the authors and not necessarily those of the UK National Health Service, the NIHR, or the UK Department of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hsu, CK., Wang, SP., Lee, J.YY. et al. Treatment of Hereditary Epidermolysis Bullosa: Updates and Future Prospects. Am J Clin Dermatol 15, 1–6 (2014). https://doi.org/10.1007/s40257-013-0059-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40257-013-0059-z