Abstract
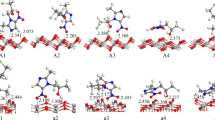
Understanding the interaction of WO3 with CO2 and H2O is vital for clarifying its role in the photocatalytic reduction of CO2. In this study, we employed density functional theory to investigate the interaction of CO2 and H2O with both perfect and defective monoclinic WO3(001) surfaces. The interactions of co-adsorbed CO2 and H2O were also studied. The major finding is that the presence of oxygen vacancies and co-adsorbed CO2 or H2O can significantly increase the stability of CO2 and H2O on the WO3(001) surface. A defective WO3(001) surface is more capable of adsorbing a single CO2 or H2O molecule than a perfect WO3(001) surface, and H2O adsorbed onto a defective WO3(001) surface spontaneously dissociates into a hydrogen atom and a hydroxy group. The presence of co-adsorbed H2O can increase the stability of CO2 on the WO3(001) surface, while the presence of the co-adsorbed CO2 can increase the stability of H2O on WO3(001) surface. The analysis of the bonding mechanisms shows that the charge redistribution between the adsorbate and the WO3(001) surface containing oxygen vacancies and co-adsorbed CO2 or H2O is stronger than that between the adsorbate and the perfect WO3(001) surface; thus, adsorption energy is higher in the former case. The results will be useful for designing WO3 photocatalysts, as well as for an atomistic-level understanding of the photocatalytic reduction of CO2.
Similar content being viewed by others
References
Cox P. M., Betts R. A., Jones C. D., Spall S. A., Nature, 2000, 408(6809), 184
Houghton J., Rep. Prog. Phys., 2005, 68(6), 1343
Karl T. R., Trenberth K. E., Science, 2003, 302(5651), 1719
Tahir M., Amin N. S., Energ. Convers. Manage., 2013, 76, 194
Navalon S. Dhakshinamoorthy A., Alvaro M., Garcia H., Chemsus-chem, 2013, 6(4), 562
Habisreutinger S. N., Schmidt-Mende L., Stolarczyk J. K., Angew. Chem. Int. Edit., 2013, 52(29), 7372
Zheng Y., Pan Z., Wang X., Chin. J. Catal., 2013, 34(3), 524
Hong J., Zhang W., Ren J., Xu R., Anal. Methods, 2013, 5(5), 1086
Mao J., Li K., Peng T., Catal. Sci. Technol., 2013, 3(10), 2481
Kukkola J., Maklin J., Halonen N., Kyllonen T., Toth G., Szabo M., Shchukarev A., Mikkola J. P., Jantunen H., Kordas K., Sensor Actuat. B: Chem., 2011, 153(2), 293
Vemuri R. S., Bharathi K. K., Gullapalli S. K., Ramana C. V., ACS Appl. Mater. Inter., 2010, 2(9), 2623
Vemuri R. S., Engelhard M. H., Ramana C. V., ACS Appl. Mater. In-ter., 2012, 4(3), 1371
Amano F., Ishinaga E., Yamakata A., J. Phys. Chem. C, 2013, 117(44), 22584
Chen X., Zhou Y., Liu Q., Li Z., Liu J., Zou Z., ACS Appl. Mater. In-ter., 2012, 4(7), 3372
Xie Y. P., Liu G., Yin L., Cheng H. M., J. Mater. Chem., 2012, 22(14), 6746
Campbell C. T., Peden C. H. F., Science, 2005, 309(5735), 713
Lee J., Sorescu D. C., Deng X., J. Am. Chem. Soc., 2011, 133(26), 10066
Indrakanti V. P., Kubicki J. D., Schobert H. H., Fuel Process. Tech-nol., 2011, 92(4), 805
He H., Zapol P., Curtiss L. A., J. Phys. Chem. C, 2010, 114(49), 21474
Pipornpong W., Wanbayor R., Ruangpornvisuti V., Appl. Surf. Sci., 2011, 257(24), 10322
Oliver P. M., Parker S. C., Egdell R. G. Jones F. H., J. Chem. Soc., Faraday Trans., 1996, 92, 2049
Lambert-Mauriat C., Oison V., Saadi L., Aguir K., Surf. Sci., 2012, 606, 40
Jones F. H., Rawlings K., Foord J. S., Cox P. A., Egdell R. G., Pethi-ca J. B., Wanklyn B. M. R., Phys. Rev. B, 1995, 52, R14392
Gholizadeh R., Yu Y. X., Appl. Surf. Sci., 2015, 357, 1187
Wu D. L., Jiang W., Liu X. Q., Qiu N. X., Xue Y., Chem. Res. Chi-nese Universities, 2016, 32(1), 118
Zhang H., Zhang H. M., Wang L. J., Shen J. Y., Chem. J. Chinese Universities, 2016, 37(9), 1660
Payne M. C., Teter M. P., Allan D. C., Arias T. A., Joannopoulos J. D., Rev. Mod. Phys., 1992, 64, 1045
Ceperley D. M., Alder B., J. Phys. Rev. Lett., 1980, 45(7), 566
Perdew J. P., Zunger A., Phys. Rev. B, 1981, 23, 5048
Segall M., Lindan P., Probert M., Pickard C., Hasnip P., Clark S., Payne M., J. Phys. Condens. Matter., 2002, 14, 2717
Vanderbilt D., Phys. Rev. B, 1990, 41, 7892
Monkhorst H. J., Pack J. D., Phys. Rev. B, 1976, 13(12), 5188
Hahn K. R., Iannuzzi M., Seitsonen A. P., Hutter, J., J. Phys. Chem. C, 2013, 117(4), 1701
Zhao Z., Li Z., Zou Z., J. Phys. Chem. C, 2012, 116(13), 7430
Loopstra B. O., Rietveld H. M., Acta Cryst., 1969, B25, 1420
Levy M., Pagnier T., Sensor. Actuat. B, 2007, 126, 204
Sun X. L., Huo R. P., Bu Y. X., Li J. L., Chem. J. Chinese Universi-ties, 2015, 36(8), 1570
Yu Y. X., ACS Appl. Mater. Inter., 2014, 6, 16267
Yu Y. X., J. Mater. Chem. A, 2014, 2, 8910
Gao H. W., Pishney S., Janik M. J., Surf. Sci., 2013, 609, 140
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China(No.11447151) and the Natural Science Foundation of Shandong Province, China(No.ZR2014BP008).
Rights and permissions
About this article
Cite this article
Liu, L., Lin, M., Liu, Z. et al. Density functional theory study of CO2 and H2O adsorption on a monoclinic WO3(001) surface. Chem. Res. Chin. Univ. 33, 255–260 (2017). https://doi.org/10.1007/s40242-017-6378-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-017-6378-5