Abstract
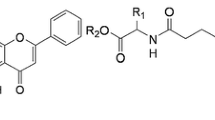
Two series of 7-O-modified chrysin derivatives were prepared from 7-O-carboxymethyl chrysin(2a), 7-O-carboxypropylchrysin(2b) and short-chain alcohols by using 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride(EDCI), N-hydroxybenzotriazole(HOBt) and 4-dimethylamiopryidine(DMAP) as coupling reagents. Taking cisplatin as a reference substance, their anti-proliferative activities in vitro against human gastric carcinoma MGC-803 cells were evaluated by the standard 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide( MTT) method. The results showed that among the compounds tested, compound hepty 4-(5-hydroxy-4-oxo-2-phenyl-4H-chromen-7-yloxy) acetate(3f) displayed the most potent growth-inhibitory effect on MGC-803 cells with half maximal inhibitory concentration(IC50) value of 3.23 μmol/L. The preliminary mechanism of inhibitory effect of compound 3f was also detected by flow cytometry(FCM), and the compound exerted anticancer activity via inducing the apoptosis of MGC-803 cells in a dose dependent manner, which suggested that compound 3f would be a potential anti-cancer agent.
Similar content being viewed by others
References
Woo K. J., Jeong Y. J., Inoue H., Park J. W., Kwon T. K., FEBS Lett., 2005, 579, 705
Lapido T., Walker M. D., Kanner J., J. Agric. Food Chem., 2002, 50, 7220
Khoo B. Y., Chua S. L., Balaram P., Int. J. Mol. Sci., 2010, 11, 2188
Cushnie T. P., Lamb A. J., Int. J. Antimicrob. Agents, 2005, 26, 343
Sirovina D., Orsolic N., Koncic M. Z., Kovacevic G., Benkovic V., Gregorovic G., Hum. Exp. Toxicol., 2013, 32, 1058
Brown E’., Hurd N. S., McCall S., Ceremuga T. E., AANA J., 2007, 75, 333
Pushpavalli G., Kalaiarasi P., Veeramani C., Pugalendi K. V., Eur. J. Pharmacol., 2010, 631, 36
Jeong H. J., Shin Y. G., Kim I. H., Pezzuto J. M., Arch. Pharm. Res., 1999, 22, 309
Yu X. M., Phan T., Patel P. N., Jaskula-Sztul R., Chen H., Cancer, 2013, 119, 774
Sun L. P., Chen A. L., Hung H. C., Chien Y. H., Huang J. S., Huang C. Y., Chen Y. W., Chen C. N., J. Agric. Food Chem., 2012, 60, 11748
Samarghandian S., Afshari J. T., Davoodi S., Clinics, 2011, 66, 1073
Woo K. J., Jeong Y. J., Park J. W., Kwon T. K., Biochem. Biophys. Res. Commun., 2004, 325, 1215
Pichichero E., Cicconi R., Mattei M., Canini A., Int. J. Oncol., 2011, 38, 473
Sawicka D., Car H., Borawska M. H., Nikliński J., Folia. Histochem. Cytobiol., 2012, 50, 25
Tsuji P. A., Winn R. N., Walle T., Chem. Biol. Interact., 2006, 164, 85
Lv P. C., Wang K. R., Li Q. S., Chen J., Sun J., Zhu H. L., Bioorg. Med. Chem., 2010, 18, 1117
Hu K., Wang W., Cheng H., Pan S. S., Ren J., Med. Chem. Res., 2011, 20, 838
Zheng X., Zhao F. F., Liu Y. M., Yao X., Zheng Z. T., Luo X., Liao D. F., Med. Chem., 2010, 6, 6
Leon I. E., Di Virgilio A. L., Porro V., Muglia C. I., Naso L. G., Williams P. A., Bollati-Fogolin M., Etcheverry S. B., Dalton. Trans., 2013, 42, 11868
Yang F., Jin H., Pi J., Jiang J. H., Liu L., Bai H. H., Yang P. H., Cai J. Y., Bioorg. Med. Chem. Lett., 2013, 23, 5544
Song X. D., He J., Ma J., Liu Y. M., Zheng X., Lei X. Y., Guo Y., Chem. J. Chinese Universities, 2014, 35(7), 1465
Veselovskaya M. V., Garazd M. M., Ogorodniichuk A. S., Garazd Ya. L., Khilya V. P., Chem. Nat. Compd., 2008, 44, 704
Mosmann T., J. Immunol. Methods, 1983, 65, 55
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the Construct Program of the Key Discipline in Hunan Province, China(No.[2011]76).
Rights and permissions
About this article
Cite this article
Liu, Y., Song, X., Ma, J. et al. Synthesis of new 7-O-modified chrysin derivatives and their anti-proliferative and apoptotic effects on human gastric carcinoma MGC-803 cells. Chem. Res. Chin. Univ. 30, 925–930 (2014). https://doi.org/10.1007/s40242-014-4269-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-014-4269-6