Abstract
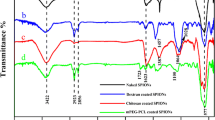
Mn-TCPP-CS n (n=6, 11, 20) as a type of potential magnetic resonance imaging(MRI) contrast agents were synthesized via manganese(II) meso-tetra(4-carboxyphenyl) porphyrin(Mn-TCPP) modified with chitosan oligosaccharides(CS n ). Experimental data of infared(IR), UV-Vis, MS, inductively coupled plasma-atomic emission spectrometer(ICP-AES) and size exclusion chromatography evidenced the formation of Mn-TCPP-CS n . The stability results show that Mn-TCPP-CS n in aqueous solution was stable enough to prevent Mn(II) ions from leaking. The magnetic properties in vitro indicate that Mn-TCPP-CS20 possesses higher longitudinal relaxivity(r 1=10.38 L·mmol−1·s−1) in aqueous solution than unmodified porphyrin Mn-TCPPNa4[manganese(II) meso-tetra(4-carboxyphenyl) porphyrin, tetrasodium salt](r 1=5.10 L·mmol−1·s−1) and the commercial contrast agent Gd-DTPA(r 1=4.05 L·mmol−1·s−1). The preliminary T 1-weighted flash image studies in vitro show that the contrast and the imaging signal of Mn-TCPP-CS n were superior to those of Mn-TCPPNa4 and Gd-DTPA under the same conditions. The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide(MTT) assay shows that Mn-TCPP-CS n has a good biocompatibility. In addition, the thermodynamical parameters(ΔH<0, ΔS<0, ΔG<0) of Mn-TCPP-CS n bound to bovine serum albumin(BSA) show that Mn-TCPP-CS n could bind to BSA spontaneously, where the binding complex was stabilized mainly by van der Waals interactions and hydrogen bonds. These results suggest that Mn-TCPP-CS n have the advantage of becoming a potential MRI contrast agent.
Similar content being viewed by others
References
Lauffer R. B., Chem. Rev., 1987, 87, 901
Caravan P., Ellison J. J., McMurry T. J., Lauffer R. B., Chem. Rev., 1999, 99, 2293
Yan G. P., Zhuo R. X., Chin. Sci. Bull., 2001, 46, 1233
Weinmann H. J., Ebert W., Misselwitz B., Schmitt-Willich H., Eur. J. Radiol., 2003, 46, 33
Huh Y. M., Jun Y. W., Song H. T., Kim S., Choi J. S., Lee J. H., Yoon S., Kim K. S., Shin J. S., Suh J. S., Cheon J., J. Am. Chem. Soc., 2005, 127, 12387
Feng J. H., Sun G. Y., Pei F. K., Liu M. L., Bioorg. Med. Chem., 2003, 11, 3359
Patronas N. J., Cohen J. S., Knop R. H., Dwyer A. J., Colcher D., Lundy J., Mornex F., Hambright P., Sohn M., Myers C. E., Cancer Treatm. Rep., 1986, 70, 391
Lyon R. C., Faustino P. J., Cohen J. S., Katz A., Mornex F., Colcher D., Baglin C., Koenig S. H., Hambright P., Magn. Reson. Med., 1987, 4, 24
Lew S., Brooks M. V., J. Nurse Pract., 2009, 5, 344
Place D. A., Faustino P. J., Berghmanns K. K., van Zijl P. C. M., Chesnick A. S., Cohen J. S., Magn. Reson. Imaging, 1992, 10, 919
Wang S., Westmoreland T. D., Inorg. Chem., 2009, 48, 719
Chen C. Y., Zhang P. Q., Lu X. L., Hou X. L., Chai Z. F., Fresenius J. Anal. Chem., 1999, 363, 512
Zhang Z., He R., Yan K., Guo Q. N., Lu Y. G., Wang X. X., Lei H., Li Z. Y., Bioorg. Med. Chem. Lett., 2009, 19, 6675
Bradshaw J. E., Gillogly K. A., Wilson L. J., Kumar K., Wan X. M., Tweedle M. F., Hernandez C., Bryant R. G., Inorg. Chim. Acta, 1998, 275/276, 106
Yang X. L., Yang X. W., Huang Y., Zhang Q., Wu J. B., Cao B. N., Zhang X. Y., Chem. J. Chinese Universities, 2012, 33(2), 426
Huang Y., Cao B. N., Yang X. L., Zhang Q., Han X. J., Guo Z. Y., Magn. Reson. Imaging, 2013, 31, 604
Rinaudo M., Prog. Polym. Sci., 2006, 31, 603
Tsai G. J., Zhang S. L., Shieh P. L., J. Food Prot., 2004, 67, 396
Datta-Gupta N., Bardos T. J., J. Heterocyclic Chem., 1966, 3, 495
Yan G. P., Bischa D., Bottle S. E., Free Rad. Boil. Med., 2007, 43, 111
Matsumoto K., Yakumaru H., Narazaki M., Nakagawa H., Anzai K., Ikehira H., Ikota N., Magn. Reson. Imaging, 2008, 26, 117
Aine S., Anelli P. L., Botta M., Brocchetta M., Canton S., Fedeli F., Gianolio E., Terreno E., J. Biol. Inorg. Chem., 2002, 7, 58
Sun G. Y., Feng J. H., Jing F. Y., Pei F. K., Chem. J. Chinese Universities, 2002, 23(10), 1837
Lindman S., Lynch I., Thulin E., Nilsson H., Dawson K. A., Linse S., Nano Lett., 2007, 7, 914
Yan G. P., Li Z., Xu W., Zhou C. K., Yang L., Zhang Q., Li L., Liu F., Han L., Ge Y. X., Guo J. F., Int. J. Pharm., 2011, 407, 119
Guo X. M., Shi T. S., J. Mol. Struct., 2006, 789, 8
Sun E., Shi Y. H., Zhang P., Zhou M., Zhang Y., Tang X., Shi T., J. Mol. Struct., 2008, 889, 28
Xiao J. B., Chen J. W., Cao H., Ren F. L., Yang C. S., Chen Y., Xu M., J. Photochem. Photobiol. A: Chem., 2007, 191, 222
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China(Nos.21261008, 21302071, 21171076), and the Cooperation Project of Hainan International Science and Technology, China(No.KJHZ2014-05).
Rights and permissions
About this article
Cite this article
Liu, Y., Huang, Y., Boamah, P.O. et al. Synthesis and in vitro evaluation of manganese(II)-porphyrin modified with chitosan oligosaccharides as potential MRI contrast agents. Chem. Res. Chin. Univ. 30, 549–555 (2014). https://doi.org/10.1007/s40242-014-4015-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-014-4015-0