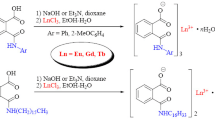
Abstract
The lowest triplet energy levels of the six ligands(T) were determined to be 22989 cm−1[1,3-bis-(1′-phenyl-3′-methyl-5′-pyrazolon-4′)-1,3-propanedione, BPMPTD], 23148 cm−1[1,4-bis-(1′-phenyl-3′-methyl-5′-pyrazolon-4′)-1,4-butanedione, BPMPBD], 23419 cm−1[1,5-bis-(1′-phenyl-3′-methyl-5′-pyrazolon-4′)-1,5-pentane-dione, BPMPPD], 23310 cm−1[1,6-bis-(1′-phenyl-3′-methyl-5′-pyrazolon-4′)-1,6-hexanedione, BPMPHD], 21978 cm−1[1,9-bis-(1′-phenyl-3′-methyl-5′-pyrazolon-4′)-1,9-nonanedione, BPMPND] and 21930 cm−1[1,10-bis-(1′-phenyl-3′-methyl-5′-pyrazolon-4′)-1,10-decanedione, BPMPDD], respectively. It was explained satisfactorily that the six ligands are more efficient for sensitizing the luminescence of Tb3+ than that of Eu3+ at room temperature, and the order of the luminescent intensities for the Tb3+ complexes is explained by the relative energy gap between T and 5 D J of Tb3+ or Eu3+. As a conclusion, when 2700 cm−1<ΔE(T-5 D 4)<3000 cm−1, the luminescent intensity of the Tb3+ complex is the strongest. This means that the lowest triplet energy level of the ligand is a chief factor to dominate RE3+ luminescence.
Similar content being viewed by others
References
Wu W. N., Yuan W. B., Tang N., Yang R. D., Yan L., Xu Z. H., Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 2006, 65, 912
Shin M. G., Thangaraju K., Kim S., Park J. W., Kim Y. H., Kwon S. K., Organic Electronics, 2011, 12, 785
Wang X., Yan Q., Chu P. C., Luo Y. H., Zhang Z. S., Wu S., Wang L. J., Zhang Q. J., Journal of Luminescence, 2011, 131, 1719
Zhuravlev K. P., Tsaryuk V. I., Pekareva I. S., Sokolnicki J., Klemenkova Z. S., Journal of Photochemistry & Photobiology, A: Chemistry, 2011, 219, 139
Sato S., Wada M., Bull. Soc. Jpn., 1970, 43, 1955
Lu Y., Zhang D., Xu Y., Chem. Res. Chinese Universites, 2013, 29(5), 831
Gou R. H., Yang R. D., Yan L., Journal of Rare Earths, 2009, 27, 790
Wang Y. L., Gou R. H., Yang R. D., Yan L., Journal of Sichuan University(Natural Science Edition), 2012, 49, 86
Ding Y. F., Yu X. B., Xiong J., Chin. Rare Earth, 2003, 24, 18
Li W. X., Tian G. X., Gao J. F., Chem. Reagent, 2003, 25, 8
Wu S. L., Yang Y. S., J. Alloys Comp., 1992, 180, 399
Yu Y. L., Wang L. M., Xu S. K., Chem. J. Chinese Universities, 2013, 34(7), 1617
Cai Z. H., Tan M. Y., Journal of Rare Earths, 2002, 20, 382
Pylewski L. L., Mikulski C. M., Coord. Chem. Rev., 1973, 11, 93
Speca A. N., Gelfand L. S., Inorg. Chem., 1976, 15, 1493
Ma L., Yang R. D., Yan L., Synth. React. Inorg. Met. Org. Chem., 1998, 28, 1343
Liu S. F., Yan L., Yang R. D., Journal of Rare Earths, 2000, 18, 81
Yan L., Yang R. D., Song F. L., Chinese Journal of Applied Chemistry, 1999, 16, 59
Boekelheide V., Linn W. J., J. Am. Chem. Soc., 1954, 76, 1286
Jerzy S., Miroslaw S., Roczniki Chem., 1964, 38, 1793
Abraham R. J., Loftus P., Proton and Carbon-13 NMR Spectroscopy, Wiley, New York, 1983, 212
Nakamato K., Infrared and Raman Spectra of Inorganic and Coordination Compounds, John Wiley, New York, 1978, 227
Geary W. Y., Coord. Chem. Rev., 1971, 7, 81
Qiang S., Rare Earth Chemistry, Henan Technology Publication, Zhengzhou, 1993, 304
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China(No.29571013).
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Gou, R., Wang, Y., Yang, R. et al. Luminescent properties of terbium(III) and europium(III) complexes with six 4-acetyl-bispyrazolones. Chem. Res. Chin. Univ. 30, 190–193 (2014). https://doi.org/10.1007/s40242-014-3380-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-014-3380-z