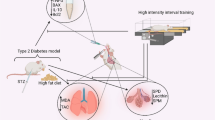
Abstract
Background
Despite the vulnerability of pulmonary tissue to diabetic conditions, there are few reports related to the detrimental effects of hyperglycemia and therapeutic modalities on lung parenchyma. Here, the apoptotic changes were monitored in the diabetic pulmonary tissue of mice (DM1) subjected to a four‒week swimming plan.
Methods
The mice were randomly allocated into Control; Control + Swimming (S); Diabetic group (D); and Diabetic + Swimming (D + S) groups (each in 8 mice). In the D and D + S groups, mice received intraperitoneally 50 mg/kg of streptozotocin (STZ). After 14 days, swimming exercise was done for four weeks. The expression of il-1β, bcl-2, bax, and caspase-3 was investigated using real-time PCR analysis. A histological examination was performed using H&E staining.
Results
DM1 significantly upregulated il-1β, bax, and caspase-3, and down-regulated bcl-2 compared to the non-diabetic mice (p < 0.05). We noted that swimming exercises reversed the expression pattern of all genes in the diabetic mice and closed to basal levels (p < 0.05). Data indicated that swimming exercise could diminish emphysematous changes, and interstitial pneumonitis induced by STZ. Along with these changes, swimming exercise had protective effects to reduce the thickness of the inter-alveolar septum and mean alveolar area in diabetic mice.
Conclusion
These data demonstrated that swimming exercises could decrease DM1-related pathologies in mouse lungs by regulating apoptosis and inflammatory response.




Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Association AD. Standards of medical care in diabetes—2013 Diabetes care, 2013. 36(Supplement 1): p. S11-S66.
Agbaje I, et al. Insulin dependant diabetes mellitus: implications for male reproductive function. Hum Reprod. 2007;22(7):1871–7.
Azimi-Nezhad M, et al. Prevalence of type 2 diabetes mellitus in Iran and its relationship with gender, urbanisation, education, marital status and occupation. Singapore Med J. 2008;49(7):571.
Ghalavand A et al. The effect of resistance training on cardio-metabolic factors in males with type 2 diabetes.Jundishapur Journal of Chronic Disease Care, 2014. 3(4).
Gardner DG, Shoback D, Muñoz BR. Greenspan. Endocrinología básica y clínica. McGraw-Hill Interamericana Editores; 2012.
Zolali E, et al. Metformin had potential to increase endocan levels in STZ-Induced Diabetic mice. Pharm Sci. 2020;26(2):133–41.
Irfan M, et al. Pulmonary functions in patients with diabetes mellitus. Lung India: official organ of Indian Chest Society. 2011;28(2):89.
Pitocco D, et al. The diabetic lung-a new target organ? Rev Diabet studies: RDS. 2012;9(1):23.
Lecube A, et al. Pulmonary function and sleep breathing: two new targets for type 2 diabetes care. Endocr Rev. 2017;38(6):550–73.
Samarghandian S, et al. Safranal treatment improves hyperglycemia, hyperlipidemia and oxidative stress in streptozotocin-induced diabetic rats. J Pharm Pharm Sci. 2013;16(2):352–62.
Dennis RJ, et al. Inadequate glucose control in type 2 diabetes is associated with impaired lung function and systemic inflammation: a cross-sectional study. BMC Pulm Med. 2010;10(1):1–7.
Talakatta G, et al. Diabetes induces fibrotic changes in the lung through the activation of TGF-β signaling pathways. Sci Rep. 2018;8(1):1–15.
Zarafshan E, Rahbarghazi R. Type 2 Diabetes Mellitus Provokes Rat Immune Cells Recruitment into the Pulmonary Niche by Up-regulation of Endothelial Adhesion Molecules 2022. 12(1): p. 176–182.
Association AD. Diagnosis and classification of diabetes mellitus Diabetes care, 2010. 33(Supplement 1): p. S62-S69.
Daghigh F, et al. Swimming training modulates lung injury induced by ovariectomy in diabetic rats: involvement of inflammatory and fibrotic biomarkers. Arch Physiol Biochem. 2022;128(2):514–20.
Rahbarghazi A, et al. Melatonin and prolonged physical activity attenuated the detrimental effects of diabetic condition on murine cardiac tissue. Tissue Cell. 2021;69:101486.
Ömercioğlu G, et al. Effects of aerobic exercise on lipopolysaccharide-induced experimental acute lung injury in the animal model of type 1 diabetes mellitus. Exp Physiol. 2022;107(1):42–57.
Paula FM, et al. Exercise training protects human and rodent β cells against endoplasmic reticulum stress and apoptosis. FASEB J. 2018;32(3):1524–36.
Samadian Z, et al. Moderate-intensity exercise training in sole and simultaneous forms with insulin ameliorates the experimental type 1 diabetes-induced intrinsic apoptosis in testicular tissue. Int J Sports Med. 2019;40(14):909–20.
Moradi A, Hosseini SA, Nikbakht M. Effect of Swimming Training and Crocin Consumption on Intrinsic Apoptosis Pathway in Muscle Tissue of High-Fat Diet-induced Obese Rats.Middle East Journal of Rehabilitation and Health Studies, 2019. 6(3).
Campos E, et al. Effect of swimming training on stress-related metabolic parameters of diabetic and non-diabetic rats. Revista Brasileira de Atividade Física & Saúde. 2014;19(2):195–5.
Zar A, et al. Effects of ginger together with swimming training on blood fat profiles in adult diabetic rats with streptozotocin. Iran J Nutr Sci Food Technol. 2016;11(2):65–74.
Khosro JD, Sajad A, Reza SG. The Effect of 6 weeks endurance swimming training on blood glucose and Cardiac tissue Antioxidants in Diabetic rats. Int Med J. 2014;21(4):379–83.
Zacarias AC, et al. Swimming training induces liver adaptations to oxidative stress and insulin sensitivity in rats submitted to high-fat diet. Redox Rep. 2017;22(6):515–23.
Oskuye ZZ, et al. Troxerutin affects the male fertility in prepubertal type 1 diabetic male rats. Iran J Basic Med Sci. 2019;22(2):197.
Qadiri A, et al. Administration of troxerutin improves testicular function and structure in type-1 diabetic adult rats by reduction of apoptosis. Avicenna J phytomedicine. 2019;9(4):374.
Ahmadi M et al. Bone marrow mesenchymal stem cells modified pathological changes and immunological responses in ovalbumin-induced asthmatic rats possibly by the modulation of miRNA155 and miRNA133 General physiology and biophysics, 2018. 37(3): p. 263–274.
Dolka I, Król M, Sapierzyński R. Evaluation of apoptosis-associated protein (Bcl-2, bax, cleaved caspase-3 and p53) expression in canine mammary tumors: an immunohistochemical and prognostic study. Res Vet Sci. 2016;105:124–33.
Muyal JP, et al. Systematic comparison of RNA extraction techniques from frozen and fresh lung tissues: checkpoint towards gene expression studies. Diagn Pathol. 2009;4(1):1–8.
Ahmadian S, et al. Intra-ovarian injection of platelet-rich plasma into ovarian tissue promoted rejuvenation in the rat model of premature ovarian insufficiency and restored ovulation rate via angiogenesis modulation. Reproductive Biology and Endocrinology. 2020;18(1):1–13.
Zolali E, et al. Metformin effect on endocan biogenesis in human endothelial cells under diabetic condition. Arch Med Res. 2019;50(5):304–14.
Krijnen PA, Simsek S, Niessen HW. Apoptosis in diabetes Apoptosis. 2009;14(12):1387–8.
Alipour MR et al. Swimming training attenuates pancreatic apoptosis through miR-34a/Sirtu in1/P53 Axis in high-fat diet and Streptozotocin-induced Type-2 diabetic rats.Journal of Diabetes & Metabolic Disorders, 2020: p.1–8.
Kumar P, et al. Hyperglycemia-induced oxidative stress induces apoptosis by inhibiting PI3-kinase/Akt and ERK1/2 MAPK mediated signaling pathway causing downregulation of 8-oxoG-DNA glycosylase levels in glial cells. Int J Biochem Cell Biol. 2014;53:302–19.
Onk D, et al. Effect of melatonin on antioxidant capacity, ınflammation and apoptotic cell death in lung tissue of diabetic rats. Acta cirurgica brasileira. 2018;33(4):375–85.
Sun C-K, et al. Early combined treatment with cilostazol and bone marrow-derived endothelial progenitor cells markedly attenuates pulmonary arterial hypertension in rats. J Pharmacol Exp Ther. 2009;330(3):718–26.
Zhang F, et al. Curcumin alleviates lung injury in diabetic rats by inhibiting nuclear factor-κB pathway. Clin Exp Pharmacol Physiol. 2015;42(9):956–63.
Kanter M, et al. Effects of low intensity exercise against apoptosis and oxidative stress in streptozotocin-induced diabetic rat heart. Exp Clin Endocrinol Diabetes. 2017;125(09):583–91.
Kermanshah I. The effect of combination of regular continuous exercise and resveratrol supplementation on some regulatory and executive factors of hepatocytic apoptosis in male diabetic rats Feyz, Journal of Kashan University of Medical Sciences, 2020. 23(6): p. 605–614.
Alimanesh Z, et al. The effect of continued training with crocin on apoptosis markers in liver tissue of high Fat Diet Induced Diabetic rats. Volume 17. Molecular & Cellular Biomechanics; 2020. p. 155. 4.
Kim DY, et al. Treadmill exercise ameliorates apoptotic cell death in the retinas of diabetic rats. Mol Med Rep. 2013;7(6):1745–50.
Cheng S-M, et al. Exercise training enhances cardiac IGFI-R/PI3K/Akt and Bcl-2 family associated pro-survival pathways in streptozotocin-induced diabetic rats. Int J Cardiol. 2013;167(2):478–85.
Acknowledgements
The authors thank the personnel of Molecular Medicine Research Center for help and guidance.
Funding
This work was supported by a grant from Molecular Medicine Research Center of Tabriz University of Medical Sciences (IR.TBZMED.VCR.REC.1399.005).
Author information
Authors and Affiliations
Contributions
N.A., A. R., F.M.B., R.R., A.G.N., J.R., and A.D. performed all analyses and prepared draft. M. A. supervised the study.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the Local Ethics Committee of Tabriz University of Medical Sciences (IR.TBZMED.VCR.REC.1399.005).
Consent for publication
None declared.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Azizi, N., Rahbarghazi, A., Bavil, F.M. et al. Swimming training reduced inflammation and apoptotic changes in pulmonary tissue in type 1 diabetic mice. J Diabetes Metab Disord 22, 793–800 (2023). https://doi.org/10.1007/s40200-023-01202-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40200-023-01202-8