Abstract
Background
One significant ethnomedicinal motivation behind Pleurotus tuberregium (PTT) and Anchomanes difformis (ADT) tubers is cardiovascular-related conditions treatment. This investigation is in this way planned for deciding the impact of PTT and ADT enhanced eating routine on key enzymes linked with hypercholesterolemia in elevated cholesterol fed rodents.
Methods
Rats were isolated into control group, hypercholesterolemic-prompted untreated group, hypercholesterolemic-treated groups with dietary routine containing PTT (5% and 10%), ADT (5% and 10%), 5% PTT and 5% ADT conbination and traditional medication, atorvastatin for 28 days. Ten rodents were utilized for every one of the groups.
Results
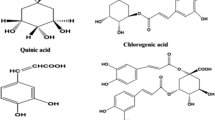
Feeding with PTT and ADT comprehensive eating regimen and their combination significantly (P < 0.05) diminished the AChE, HMG-CoA, ALT, AST, ALP, LDH and CK activities and levels of mevalonate, triglyceride (TG), total cholesterol (TC), low density lipoprotein (LDLch), atherogenic indices, MDA and ROS yet significantly increased the SOD, CAT, GPx activities, and level of HDL, GSH when contrasted with HC-initiated untreated rodents. Likewise, histopathological of liver and heart demonstrated no obsessive changes in all the treated groups when contrasted with healthy control group. HPLC fingerprinting of the PTT and ADT aqueous concentrates uncovered the nearness of ferulic acid, vanillic acid, catechin, quercetin, chlorogenic acid, ellagic acid and gallic acid. Notwithstanding, aqueous concentrate of ADT contained plentiful convergences of the polyphenolics when contrasted with PTT concentrate.
Conclusions
The tubers HMG-CoA reductase inhibitory activity could additionally support their antihypercholesterolemic use in folk medication. Accordingly, the tubers may in this way be valuable as restorative nourishment for helpful treatment of clinical conditions related hypercholesterolemia with the ADT diet holding more guarantee.








Similar content being viewed by others
Data availability
The data analyzed and materials utilized in this investigation are available from the comparing author on reasonable solicitation.
References
Venkatakrishnan K, Thangarajan S. Asian Pacific Journal of Tropical Medicine. 2012; 2012: 949–955.
Thiruchenduran M, Vijayan NA, Sawaminathan JK, Devaraj SN. Protective effect of grape seed proanthocyanidins against cholesterol cholic acid diet-induced hypercholesterolemia in rats. Cardiovasc Pathol. 2011;20:361–8.
Gu W, Xi BL, Wu QN. Cloning and distribution pattern of HMGR gene conserved fragment in Alisma orientale. J Chin Med Mater. 34(9):1335–1339.
Gu W, Geng C, Xue W. Characterization and function of the 3-hydroxy-3-methylglutaryl-CoA reductase gene in Alisma orientale (Sam.) Juz. and its relationship with protostane triterpene production. Plant Physiol Biochem. 2015;97:378–89.
Kobayashi T, Kato-Emori S, Tomita K, Ezura H. Detection of 3-hydroxy-3-methylglutaryl-coenzymeAreductase protein Cm-HMGR during fruit development in melon (Cucumis melo L.). Theor Appl Genet. 2002;104(5):779–85.
Tommer R, Rachel A, Sylvie PC, Jerry R. Impairedregulation of 3-hydroxy-3-methylglutaryl-coenzyme A reductase degradation in lovastatin-resistant cells. J Biol Chem. 1999;274(41):29341–51.
Eghdamian B, Ghose K. Mode of action and adverse effects of lipid lowering drugs. Drugs Today (Barc). 1998;34:943–56.
López C, Andrés F. Manual for the preparation and sale of fruits and vegetables. Food and Agriculture Organization of the United Nations. 2004; p. 6. ISBN 92-5-104991-2.
Nnorom IC. Lead and copper in the sclerotium of the mushroom Pleurotus tuber-regium (Osu): assessment of contribution to dietary intake in southeastern Nigeria. Toxicol Environ Chem. 2011;93(7):1359–67. https://doi.org/10.1080/02772248.2011.588603.
WHO. Trace elements in human nutrition and health. Geneva: World Health Organisation (WHO); 1996.
Akindahunsi AA, Oyetayo FL. Nutrient and anti-nutrient distribution of edible mushroom, Pleurotus tuber-regium (fries) singer. LWT Food Sci Technol (Lebensmittel-Wissenschaft and Technologie). 2006;39:548–53.
Bello OM, Jagaba SM, Bello OE. A wild edible vegetable Anchomanes difformis (Blume) Engl.: its ethnomedicinal, phytochemistry, nutritional importance and other uses. EurAsian J BioSci. 2019;13:1137–47.
Oyetayo VO. Comparative studies of the phytochemical and antimicrobial properties of the leaf, stem and tuber of Anchomanes difformis. J Pharm Toxicol. 2007;2:407–10. https://doi.org/10.3923/jpt.2007.407.410.
Burkill HM. The useful plants of west tropical Africa. Kew: Royal Botanic Gardens; 2000.
Adekunle O, Adetunji T. Antimicrobial activity of Anchomanes difformis (Blume) Engl. [family ARACEAE]. Acta SATECH. 2010;3(2):87–90.
Akinkurolere RO. Asssessment of the insecticidal properties of Anchomanes difformis (P. Beauv) powder on five beetles of stored produce. J Entomol. 2007;4(1):51–5. https://doi.org/10.3923/je.2007.51.55.
Atawodi SE, Bulus T, Ibrahim S, Ameh DA, Nok AJ, Mamman M, Galadima M. In vitro trypanocidal effect of methanolic extract of some Nigerian Savanna plants. Afr J Biotech. 2003;2:317–21. https://doi.org/10.5897/AJB2003.000-1065.
Okpo SO, Ching FP, Ayinde BA, Udi OO, Alonge PO, Eze GO. Gastroprotective effects of the ethyl acetate fraction of Anchomanes difformis (Engl). Int J Health Res. 2011;4(4):155–61.
Okpo SO, Ayinde BA, Ugwa ZI, Ching FP, Alonge PO, Udi OO. Anti-ulcer activity of the aqueous extract of Anchomanes difformis. Niger J Pharm Sci. 2012;11(1):58–65.
Okpo SO, Ozolua RI, Ching FP, Eze GO. Acute and short term toxicity studies on the aqueous extract of Anchomanes Difformis. Nig J Pharm Sci. 2013;12(2):01–10. https://doi.org/10.4314/jpb.v10i2.1.
Apetorgbor AK, Dzomeku M, Apetorgbor MM. Int Food Res J. 2013;20(6):3387–93.
Ude CM, Ezenwugo AEN, Agu RC. Composition and food value of sclerotium (Osu) and edible mushroom (Pleurotus tuber-regium). J Food Sci Technol. 2001;38:612–14.
Gregori A, Svagelj M, Pohleven J. Cultivation techniques and medicinal properties of Pleurotus spp. Food Technol Biotechnol. 2007;45(3):238–49.
Bernas E, Jaworska G, Lisiewska Z. Edible mushroom as a source of valuable nutritive constituents. Acta Sci Polonorum Technologia Alimentaria. 2006;5:5–20.
Agunloye OM, Oboh G. Hypercholesterolemia, angiotensin converting enzyme and ecto-enzymes of purinergic system: Ameliorative properties of caffeic and chlorogenic acid in hypercholesterolemic rats. J Food Biochem. 2018:2018;e12604. https://doi.org/10.1111/jfbc.12604.
Boligon AA, Piana M, Kubiça TF, Mario DN, Dalmolin TV, Bonez PC, Athayde ML. HPLC analysis and antimicrobial, antimycobacterial and antiviral activities of Tabernaemontana catharinensis A. DC. J Appl Biomed. 2015;13:7–18. https://doi.org/10.1016/j.jab.2014.01.004.
Reis EM, Neto FWS, Cattani VB, Peroza LR, Busanello A, Leal CQ, Fachinetto R. Antidepressant-like effect of Ilex paraguariensis in rats. Biomed Res Int. 2014;2014:1–9. https://doi.org/10.1155/2014/958209.
Takasaki Y. Serum lipid levels and factors affecting atherogenic index in Japanese children. J Phys Anthrop Appl Human Sci. 2005;24:511–5.
Onat A, Can G, Kaya H, Hergenc G. Atherogenic index of plasma (log10 triglyceride/high- density lipoprotein-cholesterol) predicts high blood pressure diabetes, and vascular events. J Clin Lipid. 2010;4:89–98.
Ikewuchi CJ, Ikewuchi CC. Alteration of plasma lipid profiles and atherogenic indices by Stachytarpheta jamaicensis L. (Vahl). Boikemistri. 2009;21:71–7.
Ellman GL, Courtney KD, Andres V, Featherstone RM. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol. 1961;1961(7):88–95.
Claiborne A. Catalase activity. In: Greenwald RA, editor. Handbook of methods for oxygen radical research. Boca Raton: CRC Press; 1985. pp. 283–4.
Misra HP, Fridovich I. The role of superoxide anion in the auto oxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem. 1972;247:3170–5.
Paglia DE, Valentine WN. Studies on quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med. 1967;70:158–69.
Ellman GL. Tissue sulfhydryl groups. Arch Biochem Biophys. 1959;82:70–7.
Hayashi I, Morishita Y, Imai K, Nakamura M, Nakachi K, Hayashi T. High-throughput spectrophotometric assay of reactive oxygen species in serum. Mutat Res Genet Toxicol Environ Mutagen. 2007;631(1):55–61.
Lowry OH, Rosenbrough NJ, Farr AL, Randall RJ. Protein measurement with folin phenol reagent. J Biol Chem. 1951;193:265.
Choe S, Kim H, Jeong T, Bok S, Park Y. Naringin has an antiatherogenic effect with the inhibition of intercellular adhesion molecule-1 in hypercholesterolemic rabbits. J Cardiovasc Pharmacol. 2001;38:947–55.
Kumar SA, Sudhahar V, Varalakshmi P. Attenuation of serum lipid abnormalities and cardiac oxidative stress by eicosapentaenoate-lipoate (EPA-LA) derivative in experimental hypercholesterolemia. Clin Chim Acta. 2005;355:197–204.
Girija K, Lakshman K, Udaya C, Sachi GS, Divya T. Anti-diabetic and anti-cholesterolemic activity of methanol extracts of three species of Amarathus. Asian Pac J Trop Biomed. 2011;1(2):133–8.
Johnkennedy N, Adamma E, Nnedimma NC. Hypolipidemic effects of aqueous extract of Acalypha capitata leaves in rats on high cholesterol diet. Asian Pac J Trop Biomed. 2011; 1(S2):S183–S185.
Anandhi R, Annadurai T, Anitha TS, Muralidharan AR, Najmunnisha K, Nachiappan V, Thomas PA, Geraldine P. Antihypercholesterolemic and antioxidative effects of an extract of the oyster mushroom, Pleurotus ostreatus, and its major constituent, chrysin, in Triton WR-1339-induced hypercholesterolemic rats. J Physiol Biochem. 2012. https://doi.org/10.1007/s13105-012-0215-6.
Sudhahar V, Kumar SA, Mythili Y, Varalakshmi P. Remedial effect of lupeol and its ester derivatives on hypercholesterolemia-induced oxidative and inflammatory stresses. Nutr Res. 2007;27:778–87.
Patil UK, Saraf S, Dixit VK. Hypolipidemic activity of seeds of Cassia tora Linn. J Ethnopharmacol. 2004;90:249–52.
Hokanson JE, Austin MA. Plasma triglyceride level is a risk factor for cardiovascular disease independent of high-density lipoprotein cholesterol level: a meta-analysis of population-based prospective studies. J Cardiovasc Risk. 1996;3:213–9.
Oyetayo FL. Responses of plasma lipids to edible mushroom diets in albino rats. Afr J Biotechnol. 2006;5:1263–6.
Hu SH, Liang ZC, Chia YC, Lien JL, Chen KS, Lee MY. Antihyperlipidemic and antioxidant effects of extracts from Pleurotus citrinopileatus. J Agric Food Chem. 2006;54:2103–10.
Alam N, Amin R, Khan A, Ara I, Shim MJ, Lee MW. Comparative effects of oyster mushrooms on lipid profile, liver and kidney function in hypercholesterolemic rats. Mycobiology. 2009;37:37–42.
Schneider I, Kressel G, Meyer A, Krings U, Berger R, Hahn A. Lipid lowering effects of oyster mushroom (Pleurotus ostreatus) in humans. J Funct Foods. 2011;3:17–24.
Vallianou I, Peroulis N, Pantazis P, Hadzopoulou-Cladaras M. Camphene, a plant-derived monoterpene, reduces plasma cholesterol and triglycerides in hyperlipidemic rats independently of HMG-CoA reductase activity. PLoS One. 2011;6:e20516.
Fki I, BouazizM, Sahnoun Z, Sayadi S. Hypocholesterolemic effects of phenolic-rich extracts of Chemlali olive cultivar in rats fed a cholesterol-rich diet. Bioorg Med Chem. 2005;13:5362–70.
Moss DW, Henderson AR. Enzymes. In: Burtis CA, Ashwood ER, editors. Tietz Textbook of Clinical Chemistry. 2nd ed. Philadelphia: WB Saunders; 1999. pp. 735–896.
Bukowska B, Sici´nska P, Pajak A. Oxidative stress and damage to erythrocytes in patients with chronic obstructive pulmonary disease - changes in ATPase and acetylcholinesterase activity. Int J Biochem Cell Biol. 2015;93(6):574–80.
Yamchuen P, Aimjongjun S, Limpeanchob N. Oxidized low density lipoprotein increases acetylcholinesterase activity correlating with reactive oxygen species production. Neurochem Int. 2014;78:1–6.
Dal Forno GO, Kist LW, De Azevedo MB. Intraperitoneal exposure to nano/microparticles of fullerene (C60) increases acetylcholinesterase activity and lipid peroxidation in adult zebrafish (danio rerio) Brain. BioMed Res Int. 2013;(7):1–11.
Duchnowicz P, Ziobro A, Rapacka E, Koter-Michalak M, Bukowska B. Changes in cholinesterase activity in blood of adolescent with metabolic syndrome after supplementation with extract from Aronia melanocarpa. BioMed Res Int. 2018;2018.
D’Souza T, Mengi S, Hassarajani S, Chattopadhayay S. Efficacy study of the bioactive fraction (F-3) of Acorus calamus in hyperlipidemia. Indian J Pharmacol. 2007;39(4):196–200.
Rodwell VW, Nordstrom Jl, Mitschelen JJ. Regulation of HMG-CoA reductase. Adv Lipid Res. 1976;14:1–74.
Davaro F, Forde SD, Garfield M, et al. 3-Hydroxyl-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor (statin)-induced 28-kDa interleukin-1β interferes with mature IL-1β signaling. J Biol Chem. 2014;289(23):16214–22. https://doi.org/10.1074/jbc.M114.571505.
Prasad K, Kalra J. Oxygen free radicals and hypercholesterolemic atherosclerosis: effect of vitamin E. Am Heart J. 1993;125:958–73.
Halliwell B. Lipid peroxidation, antioxidants and cardiovascular disease: how should we move forward? Cardiovasc Res. 2000;47:410–8.
Schonewille M, de Boer JF, Mele L. Statins increase hepatic cholesterol synthesis and stimulate fecal cholesterol elimination in mice. J Lipid Res. 2016;57:1455–64.
Palvai VR, Urooj A. Inhibition of 3-Hydroxy-3-methylglutaryl Coenzyme A Reductase (Ex Vivo) by Morus indica (Mulberry). Chin J Biol. 2014;2014. https://doi.org/10.1155/2014/318561.
Moron MS, Depierre JW, Mannervik B. Levels of glutathione, glutathione reductase and glutathione Stransferase activities in rat lung and liver. Biochim Biophys Acta. 1979;582:67–78.
Klaassen CD, Bracken WM, Dudley RE, Goering PL, Hazelton GA, Hjelle JJ. Role of sulfhydryls in the hepatotoxicity of organic and metallic compounds. Fundam Appl Toxicol. 1985;5:806–15.
Holmes A, Coppey LJ, Davidson EP, Yorek MA. Rat Models of Diet-Induced Obesity and High Fat/Low Dose Streptozotocin Type 2 Diabetes: Effect of Reversal of High Fat Diet Compared to Treatment with Enalapril or Menhaden Oil on Glucose Utilization and Neuropathic Endpoints. J Diabetes Res. 2015;2015. https://doi.org/10.1155/2015/307285.
Wissler RW. Theories and new horizons in the pathogenesis of atherosclerosis and the mechanisms of clinical effects. Arch Pathol Lab Med. 1992;116:1281–91.
Le Ngoc-Anh. Lipoprotein-associated oxidative stress: A new twist to the postprandial hypothesis. Int J Mol Sci. 2015;16(1):401–19.
Faleye FJ, Akinwunmi OA, Ojo OC. Investigation of the methanol extract of Anchomanes Difformis tuber extract for in vitro antioxidant, Α- amylase and Α- Glucosidase inhibitory activities. FUW Trends Sci Technol J. 2018;3(2A):342–346.
Khamis AA, Salama AF, Kenawy ME, Mohamed TM. (2017). Regulation of hepatic hydroxy methyl glutarate – CoA reductase for controlling hypercholesterolemia in rats. Biomed Pharmacother. 2017;95:1242–1250.
Jentzsch AM, Bachmann H, Fürst P, Biesalski HK. (1996). Improved analysis of malondialdehyde in human body fluids. Free Radic Biol Med. 1996;20(2):251–6.
Acknowledgements
We gratefully acknowledge Dr. Nnaemeka T. Asogwa, Central Research and Diagnostic Laboratory, Number 132, University Road, Tanke, Ilorin, Kwara State, Nigeria for his technical assistance in carrying out histopathological examination. This work was acted in partial fulfillment of the prerequisites for M.Sc of Ogundumi oluwatosin Adesola in Department of Biochemistry, Faculty of Science, Ekiti State University, Ado Ekiti, Nigeria.
Funding
This investigation was self-supported and no fund was gotten for the work.
Author information
Authors and Affiliations
Contributions
ASF and OOA carried out the analyses, analyzed and deciphered the data, and drafted the manuscript. OFL and ASF planned the investigation and participated in analysis and interpretation of data. OFL coordinated the investigation, overhauled the manuscript and approved the final form to be submitted for publication and aided in the analysis and interpretation of data. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This examination was approved by the Ethics Committee of Ekiti State University, Ado Ekiti, Nigeria.
Consent for publication
Not applicable.
Conflict of Interest
The authors declare that they have no contending interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 435 KB)
Rights and permissions
About this article
Cite this article
Oyetayo, F.L., Akomolafe, S.F. & Ogundumi, O.A. Anti-hypercholesterolemic potential of diet supplemented with Anchomanes difformis and Pleurotus tuberregium tubers in high cholesterol fed rats. J Diabetes Metab Disord 19, 1139–1155 (2020). https://doi.org/10.1007/s40200-020-00615-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40200-020-00615-z