Abstract
Objectives
Oncogenic osteomalacia is a rare paraneoplastic metabolic syndrome that is characterised by severe hypophosphataemia, hyperphosphaturia and osteomalacia secondary to renal loss of phosphate. It is commonly caused by overproduction of fibroblast growth factor-23 (FGF23) from benign tumours of mesenchymal origin. Currently, there is no clear evidence on the management of oncogenic osteomalacia in patients with metastatic solid tumours.
Methods
We report a case of breast cancer-induced oncogenic osteomalacia and discuss its diagnosis and management.
Results
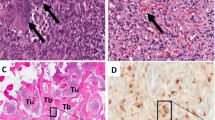
A 71-year-old woman with advanced breast cancer developed symptomatic oncogenic osteomalacia with raised FGF23, severe hypophosphataemia and hypocalcaemia. The electrolytic disturbances were exacerbated after the administration of bisphosphonates in the context of her oncological treatment. Systemic chemotherapy and maintenance endocrine treatment along with phosphate and calcium supplementation reduced the activity of oncogenic osteomalacia and resolved the electrolytic imbalances.
Conclusions
To our knowledge, this is the first reported case of oncogenic osteomalacia in a patient with breast cancer. Oncogenic osteomalacia constitutes a diagnostic and therapeutic challenge. Pre-clinical and clinical evidence suggest that a possible underlying mechanism is the presence of molecular alterations in the FGF/FGFR signalling pathway leading to overexpression of FGF23. In metastatic setting, anticancer treatment can potentially lead to the normalisation of the electrolytic disturbances and reduction of the activity of oncogenic osteomalacia. The use of antiresorptive therapy in patients with bone metastases can potentially trigger FGF23 overexpression. Its use should be guided by the patients’ risk of skeletal-related events and electrolytic disturbances as well as the degree of activity of oncogenic osteomalacia.
Similar content being viewed by others
References
Alonso G, Varsavsky M. Tumour-induced osteomalacia: an emergent paraneoplastic syndrome. Endocrinol Nutr. 2016;63(4):181–6.
Huang X, Jiang Y, Xia W. FGF23 and phosphate wasting disorders. Bone Res. 2013;1(2):120–32.
Florenzano P, Gafni RI, Collins MT. Tumor-induced osteomalacia. Bone Rep. 2017;7:90–7.
Hu F, et al. Quantitative ELISA-like immunohistochemistry of fibroblast growth factor 23 in diagnosis of tumor-induced Osteomalacia and clinical characteristics of the disease. Dis Markers. 2016;2016:3176978.
Lee EK, Martinez MCR, Blakely K, Santos KD, Hoang VC, Chow A, et al. FGF23: mediator of poor prognosis in a sizeable subgroup of patients with castration-resistant prostate cancer presenting with severe hypophosphatemia? Med Hypotheses. 2014;83(4):482–7.
Jiang Y, Xia WB, Xing XP, Silva BC, Li M, Wang O, et al. Tumor-induced osteomalacia: an important cause of adult-onset hypophosphatemic osteomalacia in China: report of 39 cases and review of the literature. J Bone Miner Res. 2012;27(9):1967–75.
Larsson T, Marsell R, Schipani E, Ohlsson C, Ljunggren Ö, Tenenhouse HS, et al. Transgenic mice expressing fibroblast growth factor 23 under the control of the alpha1(I) collagen promoter exhibit growth retardation, osteomalacia, and disturbed phosphate homeostasis. Endocrinology. 2004;145(7):3087–94.
Shimada T, Hasegawa H, Yamazaki Y, Muto T, Hino R, Takeuchi Y, et al. FGF-23 is a potent regulator of vitamin D metabolism and phosphate homeostasis. J Bone Miner Res. 2004;19(3):429–35.
Guo YC, Yuan Q. Fibroblast growth factor 23 and bone mineralisation. Int J Oral Sci. 2015;7(1):8–13.
Minisola S, Peacock M, Fukumoto S, Cipriani C, Pepe J, Tella SH, et al. Tumour-induced osteomalacia. Nat Rev Dis Primers. 2017;3:17044.
Hannan FM, Athanasou NA, Teh J, Gibbons CLMH, Shine B, Thakker RV. Oncogenic hypophosphataemic osteomalacia: biomarker roles of fibroblast growth factor 23, 1,25-dihydroxyvitamin D3 and lymphatic vessel endothelial hyaluronan receptor 1. Eur J Endocrinol. 2008;158(2):265–71.
Shimada T, Yamazaki Y, Takahashi M, Hasegawa H, Urakawa I, Oshima T, et al. Vitamin D receptor-independent FGF23 actions in regulating phosphate and vitamin D metabolism. Am J Physiol Ren Physiol. 2005;289(5):F1088–95.
Dey B, Gochhait D, Subramanian H, Ponnusamy M. Oncogenic Osteomalacia: an approach to diagnosis with a case report. J Clin Diagn Res. 2017;11(4):Ed05–ed07.
Reinert RB, Bixby D, Koenig RJ. Fibroblast growth factor 23-induced hypophosphatemia in acute leukemia. J Endocr Soc. 2018;2(5):437–43.
Okamiya T, Takahashi K, Kamada H, Hirato J, Motoi T, Fukumoto S, et al. Oncogenic osteomalacia caused by an occult paranasal sinus tumor. Auris Nasus Larynx. 2015;42(2):167–9.
Elderman JH, Wabbijn M, de Jongh F. Hypophosphataemia due to FGF-23 producing B cell non-Hodgkin's lymphoma. BMJ Case Rep. 2016;2016:bcr2015213954. https://doi.org/10.1136/bcr-2015-213954.
Rodriguez-Velver KV, Endocrinology Division, University Hospital “Dr. Jose E. Gonzalez”, Medical School, Autonomous University of Nuevo Leon, Monterrey, Mexico, Zapata-Rivera MA, Endocrinology Division, University Hospital “Dr. Jose E. Gonzalez”, Medical School, Autonomous University of Nuevo Leon, Monterrey, Mexico, Montes-Villarreal J, Endocrinology Division, University Hospital “Dr. Jose E. Gonzalez”, Medical School, Autonomous University of Nuevo Leon, Monterrey, Mexico, et al. Tumour-induced Osteomalacia secondary to a sarcoma. Eur Endocrinol. 2016;12(2):104–6.
Abate EG, Bernet V, Cortese C, Garner HW. Tumor induced osteomalacia secondary to anaplastic thyroid carcinoma: a case report and review of the literature. Bone Rep. 2016;5:81–5.
Sauder A, Wiernek S, Dai X, Pereira R, Yudd M, Patel C, et al. FGF23-associated tumor-induced Osteomalacia in a patient with small cell carcinoma: a case report and regulatory mechanism study. Int J Surg Pathol. 2016;24(2):116–20.
Mak MP, da Costa e Silva VT, Martin RM, Lerario AM, Yu L, Hoff PMG, et al. Advanced prostate cancer as a cause of oncogenic osteomalacia: an underdiagnosed condition. Support Care Cancer. 2012;20(9):2195–7.
Leaf DE, Pereira RC, Bazari H, Jüppner H. Oncogenic osteomalacia due to FGF23-expressing colon adenocarcinoma. J Clin Endocrinol Metab. 2013;98(3):887–91.
Tanner Y, Grose RP. Dysregulated FGF signalling in neoplastic disorders. Semin Cell Dev Biol. 2016;53:126–35.
Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature. 2012;490(7418):61–70. https://doi.org/10.1038/nature11412.
Touat M, Ileana E, Postel-Vinay S, Andre F, Soria JC. Targeting FGFR signaling in Cancer. Clin Cancer Res. 2015;21(12):2684–94.
Martin A, David V, Quarles LD. Regulation and function of the FGF23/klotho endocrine pathways. Physiol Rev. 2012;92(1):131–55.
Sharpe R, Pearson A, Herrera-Abreu MT, Johnson D, Mackay A, Welti JC, et al. FGFR signaling promotes the growth of triple-negative and basal-like breast cancer cell lines both in vitro and in vivo. Clin Cancer Res. 2011;17(16):5275–86.
Xiao L, Naganawa T, Lorenzo J, Carpenter TO, Coffin JD, Hurley MM. Nuclear isoforms of fibroblast growth factor 2 are novel inducers of hypophosphatemia via modulation of FGF23 and KLOTHO. J Biol Chem. 2010;285(4):2834–46.
Sequist LV, et al. Abstract CT326: phase I study of BGJ398, a selective pan-FGFR inhibitor in genetically preselected advanced solid tumors. Cancer Res. 2014;74(19 Suppl):CT326.
Miller CB, et al. Response of tumor-induced osteomalacia (TIO) to the FGFR inhibitor BGJ398. J Clin Oncol 2016;34(15_suppl):e22500–e22500
Miller RP, Tadagavadi RK, Ramesh G, Reeves WB. Mechanisms of cisplatin nephrotoxicity. Toxins. 2010;2(11):2490–518.
Chong WH, Molinolo AA, Chen CC, Collins MT. Tumor-induced osteomalacia. Endocr Relat Cancer. 2011;18(3):R53–77.
Martin M, Bell R, Bourgeois H, Brufsky A, Diel I, Eniu A, et al. Bone-related complications and quality of life in advanced breast cancer: results from a randomized phase III trial of denosumab versus zoledronic acid. Clin Cancer Res. 2012;18(17):4841–9.
Body JJ, Bone HG, de Boer RH, Stopeck A, van Poznak C, Damião R, et al. Hypocalcaemia in patients with metastatic bone disease treated with denosumab. Eur J Cancer. 2015;51(13):1812–21.
Liamis G, Milionis HJ, Elisaf M. Medication-induced hypophosphatemia: a review. QJM-Int J Med. 2010;103(7):449–59.
Vervloet MG, van Ittersum FJ, Buttler RM, Heijboer AC, Blankenstein MA, ter Wee PM. Effects of dietary phosphate and calcium intake on fibroblast growth factor-23. Clin J Am Soc Nephrol: CJASN. 2011;6(2):383–9.
Acknowledgements
We appreciate the patient and her family for their cooperation in this case report.
Author information
Authors and Affiliations
Contributions
CS and JA managed the patient and wrote the manuscript. SM and KC supervised the management of the case, revised critically and approved the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
The authors declare that they did not have any industrial links of affiliations.
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
Written informed consent was obtained from the patient for publication of this case report.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Savva, C., Adhikaree, J., Madhusudan, S. et al. Oncogenic osteomalacia and metastatic breast cancer: a case report and review of the literature. J Diabetes Metab Disord 18, 267–272 (2019). https://doi.org/10.1007/s40200-019-00398-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40200-019-00398-y