Abstract
Background
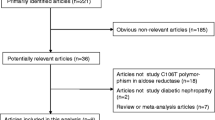
This study aimed to synthesize evidence on the association between IL-10 gene (−819 C/T, −1082 A/G, −592 A/C) polymorphisms and the risk of developing diabetic nephropathy.
Methods
A systematic literature search was done in health-related electronic databases. The search was limited to studies published in English until September 2017. We also checked the references of retrieved articles and relevant reviews for any additional studies. The methodological quality of the studies included in this review was assessed using the ‘Scales for Quality Assessment’. The I2 test was used to quantify between-study heterogeneity. A value of I2 > 50% indicated substantial heterogeneity. For the pooled analysis, summary odds ratio (OR) and its 95% confidence interval (CI) in random effect model were used.
Results
Eight case-control studies (1192 cases with diabetic nephropathy and 2399 controls) met the inclusion criteria. Three groups of people namely Africans, Asians and Caucasians were included in this review. There were significant protective effects of SNP -819 C/T in overall population (OR 0.32, 95% CI 0.26–0.4) and − 1082 A/G SNP in the Asian population (OR 0.64, 95% CI 0.47–0.86) on diabetic nephropathy in the recessive model. There was no significant effect of −592 A/C on diabetic nephropathy.
Conclusion
The findings suggest the protective effects of −1082A/G and -819G/A polymorphisms on the risk of developing diabetic nephropathy in type 2 diabetes mellitus, especially in the Asian population. Well- designed, prospective studies with sufficient number of participants are recommended to substantiate these findings.






Similar content being viewed by others
Abbreviations
- AJOL :
-
African Journals online
- CI :
-
confidence intervals
- HWE :
-
Hardy-Weinberg equilibrium
- IL :
-
interleukin
- LILACS :
-
Latin American and Caribbean health sciences literature
- MeSH :
-
medical subject heading
- OR :
-
odds ratios
- SNP :
-
single nucleotide polymorphism
- T2DM :
-
type 2 diabetes mellitus
References
Lyssenko V, Laakso M. Genetic screening for the risk of type 2 diabetes: worthless or valuable? Diabetes Care. 2013;36:120–6.
Moore KW, de Waal Malefyt R, Coffman RL, O'Garra A. Interleukin-10 and the interleukin-10 receptor. Annu Rev Immunol. 2001;19:683–765.
Cyktor JC, Turner J. Interleukin-10 and immunity against prokaryotic and eukaryotic intracellular pathogens. Infect Immun. 2011;79:2964–73.
Bijjiga E, Martino AT. Interleukin 10 (IL-10) regulatory cytokine and its clinical consequences. J Clin Cell Immunol. 2013;S1:007.
Iyer SS, Cheng G. Role of interleukin 10 transcriptional regulation in inflammation and autoimmune disease. Crit Rev Immunol. 2012;32:23–63.
Navarro-Gonzalez JF, Mora-Fernandez C. The role of inflammatory cytokines in diabetic nephropathy. J Am Soc Nephrol. 2008;19:433–42.
Montazeri S, Nalliah S, Radhakrishnan AK. Is there a genetic variation association in the IL-10 and TNF alpha promoter gene with gestational diabetes mellitus? Hereditas. 2010;147:94–102.
Eskdale J, Kube D, Tesch H, Gallagher G. Mapping of the human IL10 gene and further characterization of the 5′ flanking sequence. Immunogenetics. 1997;46:120–8.
Karhukorpi J, Laitinen T, Karttunen R, Tiilikainen AS. The functionally important IL-10 promoter polymorphism (−1082G→A) is not a major genetic regulator in recurrent spontaneous abortions. Mol Hum Reprod. 2001;7:201–3.
Kilpinen S, Huhtala H, Hurme M. The combination of the interleukin-1α (IL-1α-889) genotype and the interleukin-10 (IL-10 ATA) haplotype is associated with increased interleukin-10 (IL-10) plasma levels in healthy individuals. Eur Cytokine Netw. 2002;13:66–71.
Temple SE, Lim E, Cheong KY, Almeida CA, Price P, Ardlie KG, et al. Alleles carried at positions −819 and −592 of the IL10 promoter affect transcription following stimulation of peripheral blood cells with Streptococcus pneumoniae. Immunogenetics. 2003;55:629–32.
Seaquist ER, Goetz FC, Rich SS, Barbosa J. Familial clustering of diabetic kidney disease: evidence for genetic susceptibility to diabetic nephropathy. N Engl J Med. 1998;320:1161–5.
Rich SS. Genetics of diabetes and its complications. J Am Soc Nephrol. 2006;17:353–60.
Yaghini N, Mahmoodi M, Asadikaram GR, Hassanshahi GH, Khoramdelazad H, Arababadi M. Serum levels of interleukin 10 (IL-10) in patients with type 2 diabetic. Iran Red Crescent Med J. 2011;13:752.
Higgins JPT, Green S, ed. Cochrane handbook for systematic reviews of interventions. Version 5.1.0 (updated March 2011). The Cochrane Collaboration; 2011. Available from http://handbook.cochrane.org. Accessed 18 May 2018.
Attia J, Thakkinstian A, D'Este C. Meta-analyses of molecular association studies: methodologic lessons for genetic epidemiology. J Clin Epidemiol. 2003;56:297–303.
Thakkinstian A, McEvoy M, Minelli C, Gibson P, Hancox B, Duffy D, et al. Systematic review and meta-analysis of the association between{h}2-adrenoceptor polymorphisms and asthma: a HuGE review. Am J Epidemiol. 2005;162:201–11.
Guo SW, Thompson EA. 1992. Performing the exact test of hardy-Weinberg proportion for multiple alleles. Biometrics. 1992;48:361–72.
Lee YH. Meta- analysis of genetic association studies. Ann Lab Med. 2015;35:283–7.
Zintzaras E, Lau J. Synthesis of genetic association studies for pertinent gene-disease associations requires appropriate methodological and statistical approaches. J Clin Epidemiol. 2008;61:634–45.
Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–34.
Babel N, Gabdrakhmanova L, Hammer MH, Schoenemann C, Skrypnikov V, Poliak N, et al. Predictive value of cytokine gene polymorphisms for the development of end-stage renal disease. J Nephrol. 2006;19:802–7.
Ezzidi I, Mtiraoui N, Kacem M, Mallat SG, Mohamed MBH, Chaieb M, et al. Interleukin-10-592 C/A, -819C/T and -1082A/G promoter variants affect the susceptibility to nephropathy in Tunisian type 2 diabetes (T2DM) patients. Clin Endocrinol. 2009;70:401–7.
Kung WJ, Lin CC, Liu SH, Chaung HC. Association of interleukin-10 polymorphisms with cytokines in type 2 diabetes nephropathy. Diabetes Technol Ther. 2010;12:809–13.
Erdogan M, Cetinkalp S, Ozgen AG, Saygili F, Berdeli A, Yilmaz C. Interleukin-10 (−1082G/a) gene polymorphism in patients with type 2 diabetes with and without nephropathy. Genet Test Mol Biomarkers. 2012;16:91–4.
Arababadi MK, Reza Mirzaei M, et al. Interleukin (IL)-10 gene polymorphisms are associated with type 2 diabetes with and without nephropathy: a study of patients from the southeast region of Iran. Inflammation. 2012;35:797–802.
Yin Q, Zhai Q, Wang D, Hai J, Cao M, Wang J, et al. Investigation on the association between inerleukin-10-592C/A, 819C/T and -1082A/G gene polymorphisms and development of diabetic nephropathy. Int J Clin Exp Pathol. 2015;8:15216–21.
Ma DH, Xu QY, Liu Y, Zhai QQ, Guo MH. Association between interleukin-10 gene polymorphisms and susceptibility to diabetic nephropathy in a Chinese population. Genet Mol Res 2016;15(2). https://doi.org/10.4238/gmr.15027570.
Mahmoud AA, Sheneef A, Sayed AA, Ezat MAW, Sabet EA. Association of interleukin-10 (−592 A/C) gene polymorphism with its level in type 2 diabetes mellitus with and without nephropathy. Mol Genet Med. 2016;10:1.
Wu HC, Ling H, Na SP, Xie RJ. The research on the relationship between the polymorphism of 1082A/G, anti-inflammatory interleukin-10 gene promoter with its effect of preventing ESRD patients from microinflammation and arteriosclerosis. Zhonghua Yi Xue Za Zhi. 2005;85:2076–80.
Rodrigues KF, Pietrani NT, Sandrim VC, Vieira MAF, Fernandes AP, Bosco AA, et al. Association of a large panel of cytokine gene polymorphisms with complications and comorbidities in type 2 diabetes patients. J Diabeties Res. 2015; https://doi.org/10.1155/2015/605965.
Tsiavou A, Hatziagelaki E, Chaidaroglou A, Manginas A, Koniavitou K, Degiannis D, et al. TNF-alpha, TGF-beta1, IL-10, IL-6, gene polymorphisms in latent autoimmune diabetic of adults (LADA) and type 2 diabetes mellitus. J Clin Immunol. 2004;24:591–9.
Chang YH, Huang CN, Wu CY, Shiau MY. Association of interleukin-10 −592 C/A and −819C/T polymorphisms with type 2 diabetes mellitus. Hum Immunol. 2005;66:1258–63.
Scarpelli D, Cardellini M, Andreozzi F, Laratta E, Hribal ML, Marini MA, et al. Variants of the interleukin-10 promoter gene are associated with obesity and insulin resistance but not type 2 diabetes in Caucasian Italian subjects. Diabetes. 2006;55:1529–33.
Saxena M, Agrawal CC, Bid HK, Banerjee M. An interleukin-10 gene promoter polymorphism (−592A/C) associated with type 2 diabetes: a north Indian study. Biochem Genet. 2012;50:549–59.
Mtiraoui N, Ezzidi I, Kacem M, Ben Hadj Mohamed M, Chaieb M, Haj Jilani AB, et al. Predictive value of interleukin-10 promoter genotypes and haplotypes in determining the susceptibility to nephropathy in type 2 diabetic patients. Diabetes Metab Res Rev. 2009;25:57–63.
World Health Organization (WHO). WHO expert committee on diabetes mellitus. WHO technical report series No. 646. Geneva: WHO; 1980.
American Diabeties Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2004;27:S5–S10.
Couper KN, Blount DG, Riley EM. IL-10: the master regulator of immunity to infection. J Immunol. 2008;180:5771–7.
Zhang F, Yang Y, Lei H, Qiu J, Wang Y, Hu D, et al. A meta-analysis about the association between −1082 G/A and -819 C/T polymorphisms of IL-10 gene and risk of type 2 diabetes. Hum Immunol. 2013;74:618–26.
Mysliwska J, Zorena K, Semetkowska-Jurkiewicz E, Rachon D, Suchanek H, Mysliwski A. High levels of circulating interleukin-10 in diabetic nephropathy patients. Eur Cytokine Netw. 2005;16:117–22.
Saraheimo M, Teppo AM, Forsblom C, Fagerudd J, Groop PH. Diabetic nephropathy is associated with low-grade inflammation in type diabetic patients. Diabetologia. 2003;46:1402–7.
Acknowledgements
The authors are grateful to the participants and researchers of the primary studies included in this analysis, and to the two anonymous reviewers and the editor for providing the comments and inputs to improve the quality of manuscript. We thank the International Medical University (IMU) in Kuala Lumpur of Malaysia for allowing us to perform this study.
Author information
Authors and Affiliations
Contributions
SN: conceptualized and coordinated the study; CN, NHH, AKB: designed; CN, NHH: extracted data; AKB: checked data accuracy; CN, NHH, AKB: assessed quality of studies; CN, NHH: analyzed; SN, CN, AKB, NHH: interpreted; CN: wrote the first draft; CN, NHH, AKB, SN: revised the manuscript; All authors approved the final version of the revised manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Ethics approval and consent were waived by the Joint Research and Ethics Committee at the International Medical University in Kula Lumpur of Malaysia as this study was exclusively performed with the published data.
Consent for publication
Not applicable.
Competing interest
The authors declare that they have no competing interests.
Electronic supplementary material
Additional File 1
Scale for quality assessment of molecular association studies of diabetic nephropathy (DOC 51 kb)
Additional File 2
Leave-one- out met analysis with −1082 A/G (PDF 88 kb)
Rights and permissions
About this article
Cite this article
Naing, C., Htet, N.H., Basavaraj, A.K. et al. An association between IL-10 promoter polymorphisms and diabetic nephropathy: a meta-analysis of case-control studies. J Diabetes Metab Disord 17, 333–343 (2018). https://doi.org/10.1007/s40200-018-0349-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40200-018-0349-3