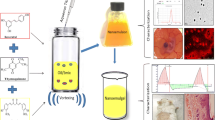
Abstract
Background
Combination of curcumin with anti-inflammatory drug like caffeine shows augmented antipsoriatic action compared to curcumin alone and reduce the time taken for treatment of Psoriasis.
Objective
The objective of the present study was to develop nanosponge (NS) based topical gel of curcumin (CUR) and caffeine (CFN) combination that acts as a potential system for the treatment of psoriasis.
Methods
NS composed of dimethyl carbonate (DMC) as crosslinker and beta-cyclodextrin (β-CD) as polymer were prepared by hot melt method and incorporated in topical gels. Factorial design (32) was constructed in a fully randomized manner to study all nine possible experimental runs. The gels were prepared by varying the content of carbopol-934 (gelling agent) (X1) and guar gum (polymer) (X2). The effect of these two independent variables on viscosity (Y1) and in vitro percent drug release (Y2) of prepared gels was evaluated. Other evaluation studies for NS and nanogels were conducted. In vivo animal studies were carried out for optimized formulation using mouse model of imiquimod-induced psoriasis.
Results
The physical and chemical characteristics exhibited by the prepared NS and gels (F1-F9) were found to be optimal. The optimization resulted in achieving formulation N10 with 69.72% in vitro drug release and 12,329.78cp viscosity. Histopathology studies revealed that prepared nanogel has promising anti-psoriatic activity. The results concluded that CUR and CFN combination has reduced the time required for showing anti-psoriatic activity to 10 days when compared to CUR alone that took around 20 days. Moreover, the nanogel has depicted sustained drug release till 12 h.
Conclusions
From the experimental findings it has been concluded that CUR and CFN combination significantly augmented the anti-psoriatic efficacy with respect to individual components and also reduced the time required for onset of effect. Thus, the proposed nanogel would be an imperative drug delivery system for more effective anti-psoriatic therapy.

Graphical abstract










Similar content being viewed by others
References
Michelle AL, Anne MB, James GK. Pathogenesis and therapy of psoriasis. Nature. 2007;445(22):866–72.
Rendon A, Schäkel K. Psoriasis Pathogenesis and Treatment. Int J Mol Sci. 2019;20(6):1475. Published 2019 Mar 23. https://doi.org/10.3390/ijms20061475.
Boehncke WH. Etiology and pathogenesis of psoriasis. Rheum Dis Clin N Am. 2015;41(4):665–75. https://doi.org/10.1016/j.rdc.2015.07.013.
Khanna NM. Turmeric - Nature’s precious gift. Curr Sci. 1999;76:1351–6.
Golnaz S, Minoo A, Parvin M, Jinous A, Kosar R, Mehdi R. Topical turmeric Microemulgel in the Management of Plaque Psoriasis; a clinical evaluation. Iran J Pharm Res. 2015;14(3):865–76.
Yunus P, Omid F, Stephen LA, Muhammed M, Alexandra EB, Thomas PJ, et al. Evidence of curcumin and curcumin analogue effects in skin diseases: a narrative review. J Cell Physiol. 2018;234(2):1165–78.
Vollono L, Falconi M, Gaziano R, et al. Potential of Curcumin in Skin Disorders. Nutrients. 2019;11(9):2169. Published 2019 Sep 10. https://doi.org/10.3390/nu11092169.
Nardo VD, Gianfaldoni S, Tchernev G, et al. Use of Curcumin in Psoriasis. Open Access Maced. J Med Sci. 2018;6(1):218–20. Published 2018 Jan 21. https://doi.org/10.3889/oamjms.2018.055.
Varma SR, Sivaprakasam TO, Mishra A, Prabhu S, Rangesh RM. Imiquimod-induced psoriasis-like inflammation in differentiated Human keratinocytes: Its evaluation using curcumin. Eur J Pharmacol. 2017;813:33–41. https://doi.org/10.1016/j.ejphar.2017.07.040.
Baden HP, Gifford AM. Isometric contraction of epidermis and stratum corneum with heating. J Investig Dermatol. 1970;54(4):298–3.
Duvoix A, Blasius R, Delhalle S, Schnekenburger M, Morceau F, Henry E, et al. Chemopreventive and therapeutic effects of curcumin. Cancer Lett. 2005;223:181–90.
Boushey HA. Bronchodilators and other agents used in asthma. In: Katzung BG, editor. Basic and clinical pharmacology. 7th ed. Los Altos (CA): Appleton & Lange; 1998. p. 330–7.
Wilken R, Veena MS, Wang MB, Srivatsan ES. Curcumin: a review of anti-cancer properties and therapeutic activity in head and neck squamous cell carcinoma. Mol Cancer.10; 2011.
Vali A, Asilian A, Khalesi E, Khoddami L, Shahtalebi M, Mohammady M. Evaluation of the efficacy of topical caffeine in the treatment of psoriasis vulgaris. J Dermatolog Treat. 2005;16(4):234–7. https://doi.org/10.1080/09546630510011801.
Yang HB, Song W, Chen LY. Differential expression and regulation of prohibition during curcumin-induced apoptosis of immortalized human epidermal HaCaT cells. Int J Mol Med. 2014;33(3):507–14.
Alashqar M, Goldstein N. Caffeine in the treatment of atopic dermatitis and psoriasis: a review. From Gene to Clinic International Congress, London: Psoriasis; 2017.
Osmani RAM, Hani U, Bhosale RR, Kulkarni PK, Shanmughanathan S. Nanosponge carriers- an archetype swing in cancer therapy: a comprehensive review. Curr Drug Targets. 2017;18(1):108–18.
Osmani RAM, Kulkarni PK, Shanmuganathan S, Hani U, Srivastava A, Prerana M, et al. A 32 full factorial design for development and characterization of a nanosponge-based intravaginal in situ gelling system for vulvovaginal candidiasis. RSC Adv. 2016;6:18737–50.
Sai VC, Priti PP, Kisan RJ, Gajendra G, Vilasrao JK. Cyclodextrin-based nanosponges: a propitious platform for enhancing drug delivery. Expert Opin Drug Deliv. 2014;11:111–20.
Venkatalakshmi R, Jason Y. Development and evaluation of mouth dissolving tablets using natural super Disintegrants. J Young Pharm. 2017;9(3):332–5.
Sarfaraz A. Formulation and evaluation of clobetasopropionate loaded nanoemulsion gel containing tea tree oil. World J Pharm Pharm Sci. 2016;5(10):616–28.
Darandale SS, Vavia PR. Cyclodextrin-based nanosponges of curcumin: formulation and physicochemical characterization. J Incl Phenom Macrocycl Chem. 2013;75:315–22.
Osmani RAM, Aloorkar NH, Ingale D, Kulkarni PK, Hani U, Bhosale RR. Microsponges based novel drug delivery system for augmented arthritis therapy. Saudi Pharm J. 2015;23(5):562–72.
Osmani RAM, Bhosale RR, Hani U, Vaghela R, Kulkarni PK. Cyclodextrin based nanosponges: impending carters in drug delivery and nanotherapeutics. Curr Drug Ther. 2015;10(1):3–19.
Kent S, Kristen MS, Marina RY, Kevin MY, Liselotte J, Gil Y. Mouse model of imiquimod-induced psoriatic itch. Pain. 2016;157:2536–43.
William RS, Kellie AM, Andrew JS, Doina D, Yi F, Xianying X. Imiquimod has strain-dependent effects in mice and does not uniquely model human psoriasis. Genome Med. 2017;9(24):1–21.
Honary S, Zahir F. Effect of zeta potential on properties of Nano-drug delivery systems- a review. Trop J Pharm Res. 2013;12:255–64.
Chatterjee S, Hui PC, Kan CW, Wang W. Dual-responsive (pH/temperature) Pluronic F-127 hydrogel drug delivery system for textile-based transdermal therapy. Sci Rep. 2019;9(1):1–3.
Kang D, Luo L, Jiang W, Lu Q, Rong M, Lai R. Curcumin shows excellent therapeutic effect on psoriasis in mouse model. Biochimie. 2016;123:73–80.
Acknowledgements
The author(s) express deep sense of gratitude towards JSS Academy of Higher Education and Research (JSSAHER), Mysuru, and University of Mysore, Mysuru, for provision of obligatory facilities to carry out present research work and Genespy Pvt. Ltd., Mysuru, for their valuable support in conducting histopathological analysis.
Funding
This study did not receive any funds.
Author information
Authors and Affiliations
Contributions
All the authors have contributed equally in reparation of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Iriventi, P., Gupta, N.V., Osmani, R.A.M. et al. Design & development of nanosponge loaded topical gel of curcumin and caffeine mixture for augmented treatment of psoriasis. DARU J Pharm Sci 28, 489–506 (2020). https://doi.org/10.1007/s40199-020-00352-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40199-020-00352-x