Abstract
Purpose of Review
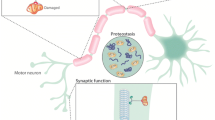
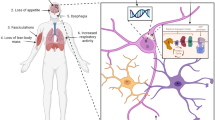
Amyotrophic lateral sclerosis (ALS) is a progressive neurodegenerative disease for which there is no cure and treatments are at best palliative. Several genes have been linked to ALS, which highlight defects in multiple cellular processes including RNA processing, proteostasis, and metabolism. Clinical observations have identified glucose intolerance and dyslipidemia as key features of ALS; however, the causes of these metabolic alterations remain elusive.
Recent Findings
Recent studies reveal that motor neurons and muscle cells may undergo cell type-specific metabolic changes that lead to utilization of alternate fuels. For example, ALS patients’ muscles exhibit reduced glycolysis and increased reliance on fatty acids. In contrast, ALS motor neurons contain damaged mitochondria and exhibit impaired lipid beta oxidation, potentially leading to increased glycolysis as a compensatory mechanism.
Summary
These findings highlight the complexities of metabolic alterations in ALS and provide new opportunities for designing therapeutic strategies based on restoring cellular energetics.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Robberecht W, Philips T. The changing scene of amyotrophic lateral sclerosis. Nat Rev Neurosci. 2013;14(4):248–64. doi:10.1038/nrn3430.
Al-Chalabi A, Jones A, Troakes C, King A, Al-Sarraj S, van den Berg LH. The genetics and neuropathology of amyotrophic lateral sclerosis. Acta Neuropathol. 2012;124(3):339–52. doi:10.1007/s00401-012-1022-4.
Rosen DR, Siddique T, Patterson D, Figlewicz DA, Sapp P, Hentati A, et al. Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature. 1993;362(6415):59–62. doi:10.1038/362059a0.
Bruijn LI, Houseweart MK, Kato S, Anderson KL, Anderson SD, Ohama E, et al. Aggregation and motor neuron toxicity of an ALS-linked SOD1 mutant independent from wild-type SOD1. Science. 1998;281(5384):1851–4.
Taylor JP, Brown Jr RH, Cleveland DW. Decoding ALS: from genes to mechanism. Nature. 2016;539(7628):197–206. doi:10.1038/nature20413.
Ling SC, Polymenidou M, Cleveland DW. Converging mechanisms in ALS and FTD: disrupted RNA and protein homeostasis. Neuron. 2013;79(3):416–38. doi:10.1016/j.neuron.2013.07.033.
DeJesus-Hernandez M, Mackenzie IR, Boeve BF, Boxer AL, Baker M, Rutherford NJ, et al. Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS. Neuron. 2011;72(2):245–56. doi:10.1016/j.neuron.2011.09.011.
Renton AE, Majounie E, Waite A, Simon-Sanchez J, Rollinson S, Gibbs JR, et al. A hexanucleotide repeat expansion in C9ORF72 is the cause of chromosome 9p21-linked ALS-FTD. Neuron. 2011;72(2):257–68. doi:10.1016/j.neuron.2011.09.010.
Peters OM, Ghasemi M, Brown Jr RH. Emerging mechanisms of molecular pathology in ALS. J Clin Invest. 2015;125(5):1767–79. doi:10.1172/JCI71601.
Dupuis L, Pradat PF, Ludolph AC, Loeffler JP. Energy metabolism in amyotrophic lateral sclerosis. Lancet Neurol. 2011;10(1):75–82. doi:10.1016/S1474-4422(10)70224-6.
Wills AM, Hubbard J, Macklin EA, Glass J, Tandan R, Simpson EP, et al. Hypercaloric enteral nutrition in patients with amyotrophic lateral sclerosis: a randomised, double-blind, placebo-controlled phase 2 trial. Lancet. 2014;383(9934):2065–72. doi:10.1016/S0140-6736(14)60222-1.
Lawton KA, Cudkowicz ME, Brown MV, Alexander D, Caffrey R, Wulff JE, et al. Biochemical alterations associated with ALS. Amyotrophic lateral sclerosis : official publication of the World Federation of Neurology Research Group on Motor Neuron Diseases. 2012;13(1):110–8. doi:10.3109/17482968.2011.619197.
Lawton KA, Brown MV, Alexander D, Li Z, Wulff JE, Lawson R, et al. Plasma metabolomic biomarker panel to distinguish patients with amyotrophic lateral sclerosis from disease mimics. Amyotrophic lateral sclerosis & frontotemporal degeneration. 2014;15(5–6):362–70. doi:10.3109/21678421.2014.908311.
Bouteloup C, Desport JC, Clavelou P, Guy N, Derumeaux-Burel H, Ferrier A, et al. Hypermetabolism in ALS patients: an early and persistent phenomenon. J Neurol. 2009;256(8):1236–42. doi:10.1007/s00415-009-5100-z.
Wuolikainen A, Jonsson P, Ahnlund M, Antti H, Marklund SL, Moritz T, et al. Multi-platform mass spectrometry analysis of the CSF and plasma metabolomes of rigorously matched amyotrophic lateral sclerosis, Parkinson’s disease and control subjects. Mol BioSyst. 2016;12(4):1287–98. doi:10.1039/c5mb00711a.
Sun S, Sun Y, Ling SC, Ferraiuolo L, McAlonis-Downes M, Zou Y, et al. Translational profiling identifies a cascade of damage initiated in motor neurons and spreading to glia in mutant SOD1-mediated ALS. Proc Natl Acad Sci U S A. 2015;112(50):E6993–7002. doi:10.1073/pnas.1520639112.
Okamoto K, Kihira T, Kondo T, Kobashi G, Washio M, Sasaki S, et al. Nutritional status and risk of amyotrophic lateral sclerosis in Japan. Amyotroph Lateral Scler. 2007;8(5):300–4. doi:10.1080/17482960701472249.
Gallo V, Wark PA, Jenab M, Pearce N, Brayne C, Vermeulen R, et al. Prediagnostic body fat and risk of death from amyotrophic lateral sclerosis: the EPIC cohort. Neurology. 2013;80(9):829–38. doi:10.1212/WNL.0b013e3182840689.
Dupuis L, Corcia P, Fergani A, Gonzalez De Aguilar JL, Bonnefont-Rousselot D, Bittar R, et al. Dyslipidemia is a protective factor in amyotrophic lateral sclerosis. Neurology. 2008;70(13):1004–9. doi:10.1212/01.wnl.0000285080.70324.27.
Hamadeh MJ, Rodriguez MC, Kaczor JJ, Tarnopolsky MA. Caloric restriction transiently improves motor performance but hastens clinical onset of disease in the Cu/Zn-superoxide dismutase mutant G93A mouse. Muscle Nerve. 2005;31(2):214–20. doi:10.1002/mus.20255.
Dupuis L, Oudart H, Rene F, Gonzalez de Aguilar JL, Loeffler JP. Evidence for defective energy homeostasis in amyotrophic lateral sclerosis: benefit of a high-energy diet in a transgenic mouse model. Proc Natl Acad Sci U S A. 2004;101(30):11159–64. doi:10.1073/pnas.0402026101.
Tefera TW, Wong Y, Barkl-Luke ME, Ngo ST, Thomas NK, McDonald TS, et al. Triheptanoin protects motor neurons and delays the onset of motor symptoms in a mouse model of amyotrophic lateral sclerosis. PLoS One. 2016;11(8):e0161816. doi:10.1371/journal.pone.0161816.
Tefera TW, Tan KN, McDonald TS, Borges K. Alternative fuels in epilepsy and amyotrophic lateral sclerosis. Neurochem Res. 2016; doi:10.1007/s11064-016-2106-7.
Joardar A, Menzl J, Podolsky TC, Manzo E, Estes PS, Ashford S, et al. PPAR gamma activation is neuroprotective in a Drosophila model of ALS based on TDP-43. Hum Mol Genet. 2014; doi:10.1093/hmg/ddu587.
Reyes ET, Perurena OH, Festoff BW, Jorgensen R, Moore WV. Insulin resistance in amyotrophic lateral sclerosis. J Neurol Sci. 1984;63(3):317–24.
Pradat PF, Bruneteau G, Gordon PH, Dupuis L, Bonnefont-Rousselot D, Simon D, et al. Impaired glucose tolerance in patients with amyotrophic lateral sclerosis. Amyotroph Lateral Scler. 2010;11(1–2):166–71. doi:10.3109/17482960902822960.
Chio A, Calvo A, Ilardi A, Cavallo E, Moglia C, Mutani R, et al. Lower serum lipid levels are related to respiratory impairment in patients with ALS. Neurology. 2009;73(20):1681–5. doi:10.1212/WNL.0b013e3181c1df1e.
Rafiq MK, Lee E, Bradburn M, McDermott CJ, Shaw PJ. Effect of lipid profile on prognosis in the patients with amyotrophic lateral sclerosis: insights from the olesoxime clinical trial. Amyotrophic lateral sclerosis & frontotemporal degeneration. 2015;16(7–8):478–84. doi:10.3109/21678421.2015.1062517.
Jawaid A, Salamone AR, Strutt AM, Murthy SB, Wheaton M, McDowell EJ, et al. ALS disease onset may occur later in patients with pre-morbid diabetes mellitus. Eur J Neurol. 2010;17(5):733–9. doi:10.1111/j.1468-1331.2009.02923.x.
Schutz B, Reimann J, Dumitrescu-Ozimek L, Kappes-Horn K, Landreth GE, Schurmann B, et al. The oral antidiabetic pioglitazone protects from neurodegeneration and amyotrophic lateral sclerosis-like symptoms in superoxide dismutase-G93A transgenic mice. J Neurosci. 2005;25(34):7805–12. doi:10.1523/JNEUROSCI.2038-05.2005.
Dupuis L, Dengler R, Heneka MT, Meyer T, Zierz S, Kassubek J, et al. A randomized, double blind, placebo-controlled trial of pioglitazone in combination with riluzole in amyotrophic lateral sclerosis. PLoS One. 2012;7(6):e37885. doi:10.1371/journal.pone.0037885.
Ionasescu V, Luca N. Studies on carbohydrate metabolism in amyotrophic lateral sclerosis and hereditary proximal spinal muscular atrophy. Acta Neurol Scand. 1964;40:47–57.
Nagano Y, Tsubaki T, Chase TN. Endocrinologic regulation of carbohydrate metabolism. Amyotrophic lateral sclerosis and Parkinsonism-dementia on Guam. Arch Neurol. 1979;36(4):217–20.
Harno K, Rissanen A, Palo J. Glucose tolerance in amyotrophic lateral sclerosis. Acta Neurol Scand. 1984;70(6):451–5.
Harris MD, Davidson MB, Rosenberg CS. Insulin antagonism is not a primary abnormality of amyotrophic lateral sclerois but is related to disease severity. J Clin Endocrinol Metab. 1986;63(1):41–6. doi:10.1210/jcem-63-1-41.
Ludolph AC, Langen KJ, Regard M, Herzog H, Kemper B, Kuwert T, et al. Frontal lobe function in amyotrophic lateral sclerosis: a neuropsychologic and positron emission tomography study. Acta Neurol Scand. 1992;85(2):81–9.
Karpati G, Klassen G, Tanser P. The effects of partial chronic denervation on forearm metabolism. Can J Neurol Sci. 1979;6(2):105–12.
Poulton KR, Rossi ML. Peripheral nerve protein glycation and muscle fructolysis: evidence of abnormal carbohydrate metabolism in ALS. Funct Neurol. 1993;8(1):33–42.
Chiang PM, Ling J, Jeong YH, Price DL, Aja SM, Wong PC. Deletion of TDP-43 down-regulates Tbc1d1, a gene linked to obesity, and alters body fat metabolism. Proc Natl Acad Sci U S A. 2010;107(37):16320–4. doi:10.1073/pnas.1002176107.
Stallings NR, Puttaparthi K, Dowling KJ, Luther CM, Burns DK, Davis K, et al. TDP-43, an ALS linked protein, regulates fat deposition and glucose homeostasis. PLoS One. 2013;8(8):e71793. doi:10.1371/journal.pone.0071793.
• Valbuena GN, Rizzardini M, Cimini S, Siskos AP, Bendotti C, Cantoni L, et al. Metabolomic analysis reveals increased aerobic glycolysis and amino acid deficit in a cellular model of amyotrophic lateral sclerosis. Mol Neurobiol. 2016;53(4):2222–40. doi:10.1007/s12035-015-9165-7. NSC-34 cell lines expressing a SOD1G93A variant show metabolic changes in stress response when deprived of serum. Specifically, G93A-NSCs show an increase in Pyruvate dehydrogenase kinase 1 (PDK1) and Lactate dehydrogenase A (LDHA), which promote lactate production.
Fergani A, Oudart H, Gonzalez De Aguilar JL, Fricker B, Rene F, Hocquette JF, et al. Increased peripheral lipid clearance in an animal model of amyotrophic lateral sclerosis. J Lipid Res. 2007;48(7):1571–80. doi:10.1194/jlr.M700017-JLR200.
• Palamiuc L, Schlagowski A, Ngo ST, Vernay A, Dirrig-Grosch S, Henriques A, et al. A metabolic switch toward lipid use in glycolytic muscle is an early pathologic event in a mouse model of amyotrophic lateral sclerosis. EMBO Mol Med. 2015;7(5):526–46. doi:10.15252/emmm.201404433. Pre-symptomatic SOD1G86R mice show reduced glucose processing and a switch to lipid dependency in glycolytic muscle.
Henriques A, Croixmarie V, Priestman DA, Rosenbohm A, Dirrig-Grosch S, D’Ambra E, et al. Amyotrophic lateral sclerosis and denervation alter sphingolipids and up-regulate glucosylceramide synthase. Hum Mol Genet. 2015;24(25):7390–405. doi:10.1093/hmg/ddv439.
Palomo GM, Manfredi G. Exploring new pathways of neurodegeneration in ALS: the role of mitochondria quality control. Brain Res. 2015;1607:36–46. doi:10.1016/j.brainres.2014.09.065.
•• Wang W, Wang L, Lu J, Siedlak SL, Fujioka H, Liang J, et al. The inhibition of TDP-43 mitochondrial localization blocks its neuronal toxicity. Nat Med. 2016;22(8):869–78. doi:10.1038/nm.4130. TDP-43 localizes to the inner mitochondrial membrane where it inhibits the translation of mRNA crucial to respiratory complex I function.
•• Lopez-Gonzalez R, Lu Y, Gendron TF, Karydas A, Tran H, Yang D, et al. Poly(GR) in C9ORF72-related ALS/FTD compromises mitochondrial function and increases oxidative stress and DNA damage in iPSC-derived motor neurons. Neuron. 2016;(2):92, 383–391. doi:10.1016/j.neuron.2016.09.015. GR(80) dipeptides produced by C9orf72 G4C2 expansion repeats cause mitochondrial dysfunction and increase oxidative stress in induced pluripotent stem cells.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Archi Joardar and Ernesto Manzo declare that they have no conflict of interest.
Daniela C. Zarnescu has a patent pending.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Neurogenetics and Psychiatric Genetics
Rights and permissions
About this article
Cite this article
Joardar, A., Manzo, E. & Zarnescu, D.C. Metabolic Dysregulation in Amyotrophic Lateral Sclerosis: Challenges and Opportunities. Curr Genet Med Rep 5, 108–114 (2017). https://doi.org/10.1007/s40142-017-0123-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40142-017-0123-8