Abstract
Purpose of Review
Spinal muscular atrophy is a devastating neurodegenerative disorder. With the recent approval of the first treatment, nusinersen (Spinraza), the natural course of the disease will change. It poses new challenges in administration and highlights the importance of early diagnosis.
Recent Findings
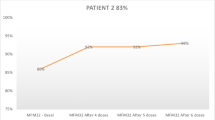
Nusinersen, an antisense oligonucleotide, helps to ameliorate disease severity in patients with spinal muscular atrophy (SMA)1 and SMA2 early in their disease course, helping to decrease the risk of death and respiratory support, as well as enhance motor outcomes in all patients with SMA. Treatment earlier in the course of disease is associated with better outcomes. Overall, treatment is well tolerated, but repeated intrathecal administration can be both technically difficult as well as expensive.
Summary
Nusinersen is an important step forward in the treatment of spinal muscular atrophy. With the availability of a treatment, we have to change our approach to diagnosis, focusing on carrier testing and/or newborn screening as well as a low threshold for clinical testing.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Ogino S, Leonard DGB, Rennert H, Wilson RB. Spinal muscular atrophy genetic testing experience at an academic medical center. J Mol Diagn. 2002;4:53–8.
Majumdar R, Rehana Z, Al Jumah M, Fetaini N. Spinal muscular atrophy carrier screening by multiplex polymerase chain reaction using dried blood spot on filter paper. Ann Hum Genet. 2005;69:216–21.
• Macdonald WK, Hamilton D, Kuhle S. SMA carrier testing: a meta-analysis of differences in test performance by ethnic group. Prenat Diagn. 2014;34:1219–26. Describes the limitations of carrier testing among different ethnic groups.
Darras BT. Spinal muscular atrophies. Pediatr Clin N Am. 2015;62:743–66.
Finkel RS, McDermott MP, Kaufmann P, et al. Observational study of spinal muscular atrophy type I and implications for clinical trials. Neurology. 2014;83:810–7.
Kaufmann P, McDermott MP, Darras BT, et al. Prospective cohort study of spinal muscular atrophy types 2 and 3. Neurology. 2012;79:1889–97.
Kaufmann P, McDermott MP, Darras BT, et al. Observational study of spinal muscular atrophy type 2 and 3: functional outcomes over 1 year. Arch Neurol. 2011;68:779–86.
Rudnik-Schöneborn S, Lützenrath S, Borkowska J, Karwanska A, Hausmanowa-Petrusewicz I, Zerres K. Analysis of creatine kinase activity in 504 patients with proximal spinal muscular atrophy types I-III from the point of view of progression and severity. Eur Neurol. 1998;39:154–62.
Lefebvre S, Bürglen L, Reboullet S, Clermont O, Burlet P, Viollet L, et al. Identification and characterization of a spinal muscular atrophy-determining gene. Cell. 1995;80:155–65.
Ogino S, Leonard DGB, Rennert H, Wilson RB. Spinal muscular atrophy genetic testing experience at an academic medical center. J Mol Diagn. 2002;4:53–8.
Burghes AHM, Beattie CE. The NIH public access policy. Brain Behav Immun. 2008;22:4109.
Schrank B, Götz R, Gunnersen JM, Ure JM, Toyka KV, Smith AG, et al. Inactivation of the survival motor neuron gene, a candidate gene for human spinal muscular atrophy, leads to massive cell death in early mouse embryos. Proc Natl Acad Sci U S A. 1997;94:9920–5.
Lorson CL, Hahnen E, Androphy EJ, Wirth B. A single nucleotide in the SMN gene regulates splicing and is responsible for spinal muscular atrophy. Proc Natl Acad Sci U S A. 1999;96:6307–11.
Singh RN, Howell MD, Ottesen EW, Singh NN. Diverse role of survival motor neuron protein. Biochim Biophys Acta - Gene Regul Mech. 2017;1860:299–315.
Feldkötter M, Schwarzer V, Wirth R, Wienker TF, Wirth B. Quantitative analyses of SMN1 and SMN2 based on real-time lightCycler PCR: fast and highly reliable carrier testing and prediction of severity of spinal muscular atrophy. Am J Hum Genet. 2002;70:358–68.
Singh NK, Singh NK, Singh RN, Singh RN. Splicing of a critical exon of human. Society. 2006;26:1333–46.
Hua Y, Vickers TA, Baker BF, Bennett CF, Krainer AR. Enhancement of SMN2 exon 7 inclusion by antisense oligonucleotides targeting the exon. PLoS Biol. 2007;5:729–44.
Hua Y, Sahashi K, Hung G, Rigo F, Passini MA, Bennett CF, et al. Antisense correction of SMN2 splicing in the CNS rescues necrosis in a type III SMA mouse model. Genes Dev. 2010;24:1634–44.
Passini MA, Bu J, Richards AM, et al. Ameliorate symptoms of severe spinal muscular atrophy. 2011. https://doi.org/10.1126/scitranslmed.3001777.Antisense.
Rigo F, Chun SJ, Norris DA, et al. Pharmacology of a central nervous system delivered 2’-O-methoxyethyl-modified survival of motor neuron splicing oligonucleotide in mice and nonhuman primates. J Pharmacol Exp Ther. 2014;350:46–55.
Chiriboga CA, Swoboda KJ, Darras BT, Iannaccone ST, Montes J, De Vivo DC, et al. Results from a phase 1 study of nusinersen (ISIS-SMNRx) in children with spinal muscular atrophy. Neurology. 2016;86:890–7.
•• Finkel RS, Chiriboga CA, Vajsar J, et al. Treatment of infantile-onset spinal muscular atrophy with nusinersen: a phase 2, open-label, dose-escalation study. Lancet. 2016;388:3017–26. Phase 2 study of nursinersen, demonstrating efficacy of the drug throughout the brain after intrathecal administration.
•• Finkel RS, Mercuri E, Darras BT, et al. Nusinersen versus sham control in infantile-onset spinal muscular atrophy. N Engl J Med. 2017;377:1723–32. Phase 3 study of nusinersen, demonstrating efficacy in SMA type 1 patients.
Kuntz NL, Mercuri E, Finkel RS, Kirschner J, Chiriboga CA, Sun P, et al. No Title. In: Efficacy and safety of nusinersen in later-onset spinal muscular atrophy (SMA) End of Study results from the phase 3 CHERSH study. 2017. p S264.
De Vivo DC, Bertini E, Hwu WL, Foster R, Gheuens S, Farwell W, et al. No title. In: Child Neurol. Soc. One year outcomes following treatment with Nusinersen: Interim results from the NURTURE Study of presymptomatic infants with genetically diagnosed spinal muscular atrophy (SMA). 2018. pp S265–6.
Foust KD, Wang X, McGovern VL, et al. Rescue of the spinal muscular atrophy phenotype in a mouse model by early postnatal delivery of SMN. Nat Biotechnol. 2010;28:271–4.
Chiriboga CA. Nusinersen for the treatment of spinal muscular atrophy. Expert Rev Neurother. 2017;17:955–62.
Haché M, Swoboda KJ, Sethna N, Farrow-Gillespie A, Khandji A, Xia S, et al. Intrathecal injections in children with spinal muscular atrophy. J Child Neurol. 2016;31:899–906.
Engelhardt JA. Comparative renal toxicopathologyof antisense oligonucleotides. Nucleic Acid Ther. 2016;26:199–209.
Wood SL, Brewer F, Ellison R, Biggio JR, Edwards RK. Prenatal Carrier Screening for Spinal Muscular Atrophy. Am J Perinatol. 2016;33(12):1211–7. https://doi.org/10.1055/s-0036-1593347.
Dobrowolski SF, Pham HT, Downes FP, Prior TW, Naylor EW, Swoboda KJ. Newborn screening for spinal muscular atrophy by calibrated short-amplicon melt profiling. Clin Chem. 2012;58:1033–9.
• Lin C-W, Kalb SJ, Yeh W-S. Delay in diagnosis of spinal muscular atrophy: asystematic literature review. Pediatr Neurol. 2015;53:293–300. Describes what leads to delay inclinical diagnosis across different types of spinal muscular atrophy.
•• Mendell JR, Al-Zaidy S, Shell R, et al. Single-dose gene-replacement therapy for spinal muscular atrophy. N Engl J Med. 2017;377:1713–22. The first report of gene therapy in SMA, demonstrating impressive motor gains andclinical outcomes
Harrington EA, Sloan JL, Manoli I, Chandler RJ, Schneider M, McGuire PJ, et al. Neutralizing antibodies against adeno-associated viral capsids in patients with mutmethylmalonic acidemia. Hum Gene Ther. 2016;27:345–53.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Sabrina W. Yum declares no conflicts of interest. Elizabeth A Kichula was an advisor for Avexis Pharmaceuticals. John Brandsema reports grants and personal fees from Biogen, grants and personal fees from AveXis, grants and personal fees from Sarepta, personal fees from Alexion, personal fees from Marathon, and grants and personal fees from PTC Therapeutics.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Neurology
Rights and permissions
About this article
Cite this article
Kichula, E.A., Yum, S.W. & Brandsema, J. Spinal Muscular Atrophy: Entering the Treatment Age. Curr Pediatr Rep 6, 9–15 (2018). https://doi.org/10.1007/s40124-018-0150-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40124-018-0150-2