Abstract
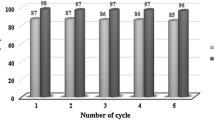
Nano-tungsten trioxide-supported sulfonic acid was found to be an impressive heterogeneous catalyst for the one-pot condensation of aromatic aldehydes, with β-naphthol to afford 14-Aryl-14H-dibenzo[a,j]xanthene derivatives in good to excellent yields. The major advantages of the present method are short reaction times, simple workup, ease in purification and environmentally benign approach. The catalyst can be recovered easily and reused five times without noticeable loss of activity.




Similar content being viewed by others
References
Reddi Mohan Naidu K, Satheesh Krishna B, Anil Kumar M et al (2012) Design, synthesis and antiviral potential of 14-aryl/heteroaryl-14H-dibenzo[a,j]xanthenes using an efficient polymer-supported catalyst. Molecules (Basel, Switzerland) 17:7543–7555
Wang H, Lu L, Zhu S et al (2006) The phototoxicity of xanthene derivatives against Escherichia coli, Staphylococcus aureus, and Saccharomyces cerevisiae. Curr Microbiol 52:1–5
Spatafora C, Barresi V, Bhusainahalli VM et al (2014) Bio-inspired benzo[k,l]xanthene lignans: synthesis, DNA-interaction and antiproliferative properties. Org Biomol Chem 12:2686–2701
Lee SH, Nam DH, Park CB (2009) Screening xanthene dyes for visible light-driven nicotinamide adenine dinucleotide regeneration and photoenzymatic synthesis. Adv Synth Catal 351:2589–2594
Pellosi DS, Batistela VR, Souza VR et al (2013) Evaluation of the photodynamic activity of xanthene dyes on Artemia salina described by chemometric approaches. An Acad Bras Cienc 85:1267–1274
Wu H, Chen X-M, Wan Y et al (2009) Synthesis and luminescence of 14-Aryl- or Alkyl-14H-dibenzo[a,j]xanthenes catalyzed by 2-1′-methylimidazolium-3-yl-1-ethyl sulfate. Synth Commun 39:3762–3771
Heravi MM, Bamoharram FF, Tavakoli-Hoseini N (2011) Generation of Brønsted acidic ionic liquid by Keggin Heteropoly acid and its application in the synthesis of 14-Aryl-14H-dibenzo[a,j]xanthenes. Synth React Inorg Met-Org Nano-Metal Chem 41:616–620
Ghassamipour S, Sardarian AR (2012) Facile catalyzed preparation of 14-Aryl- or Alkyl-14-H-dibenzo[a,j]xanthenes by Dodecylphosphonic acid and dodecylsulfamic acid: environmentally benign methods. J Heterocycl Chem 49:669–674
Esmaeilpour M, Javidi J, Dehghani F et al (2014) Fe3O4@SiO2-imid-PMAn magnetic porous nanospheres as recyclable catalysts for the one-pot synthesis of 14-aryl- or alkyl-14H-dibenzo[a,j]xanthenes and 1,8-dioxooctahydroxanthene derivatives under various conditions. New J Chem 38:5453–5461
Rivera TS, Sosa A, Romanelli GP et al (2012) Tungstophosphoric acid/zirconia composites prepared by the sol–gel method: an efficient and recyclable green catalyst for the one-pot synthesis of 14-aryl-14H-dibenzo[a,j]xanthenes. Appl Catal A Gen 443–444:207–213
Kumar R, Nandi GC, Verma RK et al (2010) A facile approach for the synthesis of 14-aryl- or alkyl-14H-dibenzo[a,j]xanthenes under solvent-free condition. Tetrahedron Lett 51:442–445
Venu Madhav J, Thirupathi Reddy Y, Narsimha Reddy P et al (2009) Cellulose sulfuric acid: an efficient biodegradable and recyclable solid acid catalyst for the one-pot synthesis of aryl-14H-dibenzo[a.j]xanthenes under solvent-free conditions. J Mol Catal A: Chem 304:85–87
Amini MM, Seyyedhamzeh M, Bazgir A (2007) Heteropolyacid: an efficient and eco-friendly catalyst for the synthesis of 14-aryl-14H-dibenzo[a,j]xanthene. Appl Catal A Gen 323:242–245
Lal J, Sharma M, Sahu PK et al (2013) Multi-component one-pot synthesis of 4-Aryl substituted dihydropyrimidinones and mechanistic study under solvent-free conditions using NiO2 as heterogeneous recyclable green catalyst. Proc Natl A Sci India A 83:187–193
Choudhary VR (2013) Highly efficient catalyst derived from Ni–Fe-hydrotalcite for solvent-free O- or S-acetylation of alcohols, phenols and thiols at room temperature. Proc Natl A Sci India A 83:15–19
Dandia A, Singh R, Gupta SL et al (2014) ZnS nanoparticle catalysed four component syntheses of novel spiropolyhydroquinoline derivatives in aqueous medium under ultrasonic irradiation. Proc Natl A Sci India A 85:19–27
Kumar RS, Idhayadhulla A, Nasser AJA et al (2010) Synthesis and anticonvulsant activity of a new series of 1,4-dihydropyridine derivatives. Indian J Pharm Sci 72:719–725
Min Y, Akbulut M, Kristiansen K et al (2008) The role of interparticle and external forces in nanoparticle assembly. Nat Mater 7:527–538
Polshettiwar V, Varma RS (2010) Green chemistry by nano-catalysis. Green Chem 12:743–754
Heveling J (2012) Heterogeneous catalytic chemistry by example of industrial applications. J Chem Educ 89:1530–1536
Amoozadeh A, Rahmani S (2015) Nano-WO3-supported sulfonic acid: new, efficient and high reusable heterogeneous nano catalyst. J Mol Catal A Chem 396:96–107
Bitaraf M, Amoozadeh A, Otokesh S (2016) A simple and efficient one-pot synthesis of 1, 4-dihydropyridines using nano-WO3-supported sulfonic acid as an heterogeneous catalyst under solvent-free conditions. J Chin Chem Soc 63:336–344
Mirjalili BBF, Bamoniri A, Akbari A (2008) BF3 SiO2: an efficient alternative for the synthesis of 14-aryl or alkyl-14H-dibenzo[a,j]xanthenes. Tetrahedron Lett 49:6454–6456
Mohammadi Ziarani G, Badiei AR, Azizi M (2011) The one-pot synthesis of 14-aryl-14H-dibenzo[a,j]xanthene derivatives using sulfonic acid functionalized silica (-Pr-) under solvent free conditions. Sci Iran 18:453–457
Zolfigol MA, Khakyzadeh V, Moosavi-Zare AR et al (2012) Preparation of various xanthene derivatives over sulfonic acid functionalized imidazolium salts (SAFIS) as novel, highly efficient and reusable catalysts. C R Chim 15:719–736
Bhattacharya AK, Rana KC, Mujahid M et al (2009) Synthesis and in vitro study of 14-aryl-14H-dibenzo[a.j]xanthenes as cytotoxic agents. Bioorg Med Chem Lett 19:5590–5593
Kumar S, Goyal A, Sohal HS, Kumar S (2013) A facile, one pot, solvent free synthesis of 14-alkyl or aryl-14H-dibenzo[a,j]xanthenes and 12-aryl/alkyl-8,9,10,12-tetrahydrobenzo[a]xanthen-11-one derivatives. Chem Sci Trans 4:1459–1465
Fareghi-Alamdari R, Golestanzadeh M, Agend F et al (2013) Application of highly sulfonated single-walled carbon nanotubes: an efficient heterogeneous catalyst for the one-pot synthesis of 14-aryl-14H-dibenzo[a,j]xanthenes under solvent-free conditions. C R Chim 16:878–887
Khaligh NG, Shirini F (2015) N-Sulfonic acid poly(4-vinylpyridinium) hydrogen sulfate as an efficient and reusable solid acid catalyst for one-pot synthesis of xanthene derivatives in dry media under ultrasound irradiation. Ultrason Sonochem 22:397–403
Bigdeli MA, Heravi MM, Hossein Mahdavinia G (2007) Wet cyanuric chloride catalyzed simple and efficient synthesis of 14-aryl or alkyl-14-H-dibenzo[a,j]xanthenes. Catal Commun 8:1595–1598
Kumar A, Sharma S, Maurya RA et al (2009) diversity oriented synthesis of benzoxanthene and benzochromene libraries via one-pot, three-component reactions and their anti-proliferative activity. J Comb Chem 12:20–24
Fu G-Y, Huang Y-X, Chen X-G et al (2009) An efficient synthesis of 14-aryl or alkyl-14H-dibenzo[a. j]xanthenes using reusable HBF4-SiO2 catalyst under thermal and solvent-free conditions. J Chin Chem Soc 56:381–385
de Andrade Bartolomeu A, de Menezes M, da Silva Filho L (2014) Efficient one-pot synthesis of 14-aryl-14H-dibenzo[a,j]xanthene derivatives promoted by niobium pentachloride. Chem Pap 68:1593–1600
Dharma Rao GB, Kaushik MP, Halve AK (2012) An efficient synthesis of naphtha[1,2-e]oxazinone and 14-substituted-14H-dibenzo[a,j]xanthene derivatives promoted by zinc oxide nanoparticle under thermal and solvent-free conditions. Tetrahedron Lett 53:2741–2744
Acknowledgements
Authors thank the Faculty of Chemistry of Semnan University for supporting this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bitaraf, M., Amoozadeh, A. & Otokesh, S. Nano-WO3-Supported Sulfonic Acid: A Versatile Catalyst for the One-Pot Synthesis of 14-Aryl-14H-dibenzo[a,j]xanthene Derivatives Under Solvent-Free Conditions. Proc. Natl. Acad. Sci., India, Sect. A Phys. Sci. 89, 437–443 (2019). https://doi.org/10.1007/s40010-018-0498-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40010-018-0498-2