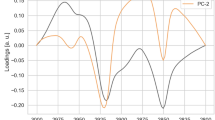
Abstract
The conformation of lens protein α crystallin was investigated using different spectroscopic techniques under normal and UV-C-irradiated condition. The structural elucidation of commercially available lens protein α crystallin under the effects of UV-C irradiation has never been reported earlier. To study the effects of irradiation on the lens protein, we used UV–visible spectroscopy, CD spectroscopy, and steady-state and time-resolved fluorescence measurements along with FTIR study, under increasing doses of UV-C irradiation. Using the secondary and tertiary structural changes as parameters for detecting conformational perturbation, we investigated the structural paradigm shift in the lens protein α crystallin. Increasing doses of UV-C radiation resulted in decreasing β sheet content of α crystallin from 30 to 10%. The fluorescence profile confirmed the formation of ROS species in the protein upon extensive exposure to UV-C irradiation. These results inferred UV-C irradiation may induce alteration of secondary structure of the lens protein leading to impaired biological functioning.





Similar content being viewed by others
References
Ikehata H, Ono T (2011) The mechanisms of UV mutagenesis. J Radiat Res 52(2):115–125
Protić-Sabljić M, Tuteja N, Munson PJ, Hauser J, Kraemer KH, Dixon K (1986) UV light-induced cyclobutane pyrimidine dimers are mutagenic in mammalian cells. Mol Cell Biol 6(10):3349–3356
Birben E, Sahiner UM, Sackesen C, Erzurum S, Kalayci O (2012) Oxidative stress and antioxidant defense. World Allergy Organ J 5(1):9
Wu JWR, Kao CY, Lin LTW, Wen WS, Lai JT, Wang SSS (2013) Human γD-crystallin aggregation induced by ultraviolet C irradiation is suppressed by resveratrol. Biochem Eng J 78:189–197
Horwit J, Bova MP, Ding L, Haley DA, Stewart PL (1999) Lens α-crystallin: function and structure. Eye 13:403–408
Peschek J, Braun N, Franzmann TM, Georgalis Y, Haslbeck M, Weinkauf S, Buchner J (2009) The eye lens chaperone α-crystallin forms defined globular assemblies. PNAS 106(32):13272–13277
Bhagyalaxmi SG, Srinivas PNBS, Barton KA, Kumar KR, Vidyavathi M, Petrash JM, Padma T (2009) A novel mutation (F71L) in αA-crystallin associated with age-related cataract results in defective chaperone-like function despite unaltered structure. Biochim Biophys Acta 1792(10):974–981
Horwitz J (2003) Alpha-crystallin. Exp Eye Res 76(2):145–153
Reddy GB, Kumar PA, Kumar MS (2006) Chaperone-like activity and hydrophobicity of α-crystallin. IUBMB Life 58(11):632–641
Posner M (2003) A comparative view of alpha-crystallins: the contribution of comparative studies to understanding function. Integr Comp Biol 43(4):481–491
Kingsley CN, Bierma JC, Pham V, Martin RW (2014) γS-crystallin proteins from the antarctic nototheniidtoothfish: a model system for investigating differential resistance to chemical and thermal denaturation. J Phys Chem B 118(47):13544–13553
Horwitz J, Bova MP, Ding LL, Haley DA, Stewart PL (1999) Lens α-crystallin: function and structure. Eye 13:403–408
Bloemendal H, de Jon W, Jaenicke R, Lubsen NH, Slingsby C, Tardieu A (2004) Ageing and vision: structure, stability and function of lens crystallins. Prog Biophys Mol Biol 86(3):407–485
Welch WJ (1992) Mammalian stress response: cell physiology, structure/function of stress proteins, and implications for medicine and disease. Physiol Rev 72(4):1063–1081
Santhoshkumar P, Udupa P, Murugesan R, Sharma KK (2008) Significance of interactions of low molecular weight crystallin fragments in lens aging and cataract formation. J Biol Chem 283(13):8477–8485
Moreau KL, King JA (2012) Protein misfolding and aggregation in cataract disease and prospects for prevention. Trends Mol Med 18(5):273–282
Kumar AP, Reddy GB (2009) Modulation of a-crystallin chaperone activity: a target to prevent or delay cataract? IUBMB Life 61(5):485–495
Borkman RF, Douhal A, Yoshihara K (1993) Picosecond fluorescence decay in photolyzed lens protein α crystallin. Biochemistry 32(18):4787–4792
Kong J, Shaoning YU (2007) Fourier transform infrared spectroscopic analysis of protein secondary structures. Acta Biochim Biophys Sin 39(8):549–559
Haiss W, Thanh NT, Aveyard J, Fernig DG (2007) Determination of size and concentration of gold nanoparticles from UV–Vis spectra. Anal Chem 79(11):4215–4221
Griebenow K, Laureano YD, Santos AM, Clemente IM, Rodríguez L, Vidal MW, Barletta G (1999) Improved enzyme activity and enantioselectivity in organic solvents by methyl-β-cyclodextrin. J Am Chem Soc 121(36):8157–8163
Cohen FB, Baillet G, Ayguavives TD, Garcia PO, Krutmann J, García PP, Reme C, Wolffsohn JS (2014) Ultraviolet damage to the eye revisited: eye-sun protection factor (E-SPF®), a new ultraviolet protection label for eyewear. Clin Ophthalmol 8:87–104
Schmid FX (2001) Biological macromolecules: UV–visible spectrophotometry. Wiley, New York
Greenfield NJ (2006) Using circular dichroism spectra to estimate protein secondary structure. Nat Protoc 1(6):2876–2890
Basak P, Pattanayak R, Bhattacharyya M (2015) Transition metal induced conformational change of heme proteins. Spectrosc Lett 48(5):324–330
Gabellieri E, Strambini GB (2006) ANS fluorescence detects widespread perturbations of protein tertiary structure in ice. Biophys J 90(9):3239–3245
Mafia K, Gupta R, Kirk M, Wilson L, Srivastava OP, Barnes S (2008) UV-A-induced structural and functional changes in human lens deamidated αB-crystallin. Mol Vis 14:234–248
Walrant P, Santus R (1974) N-formyl-kynurenine, a tryptophan photooxidationproduct, as a photodynamic sensitizer. Photochem Photobiol 19(6):411–417
Borkman RF (1977) Ultraviolet action spectrum for tryptophan destruction in aqueous solution. Photo chem Photobiol 26(2):163–166
Wang SSS, Wen WS (2010) Examining the influence of ultraviolet C irradiation on recombinant human γD-crystallin. Mol Vis 16:2777–2790
Ghisaidoobe AB, Chung SJ (2014) Intrinsic tryptophan fluorescence in the detection and analysis of proteins: a focus on Förster resonance energy transfer techniques. Int J Mol Sci 15(12):22518–22538
Baldassarre M, Li C, Eremina N, Goormaghtigh E, Barth A (2015) Simultaneous fitting of absorption spectra and their second derivatives for an improved analysis of protein infrared spectra. Molecules 20(7):12599–12622
Vedantham G, Sparks HG, Sane SU, Tzannis S, Przybycien TM (2000) A holistic approach for protein secondary structure estimation from infrared spectra in H2O solutions. Anal Biochem 285(1):33–49
Acknowledgments
We are grateful to World Bank-ICZMP (54-ICZMP/3P), DST (FIST), UGC-CAS, UGC-UPE, and DBT-IPLS, Government of India for providing the instrumental facility in the Department of Biochemistry, Calcutta University. Turban Kar thanks to UGC-DAE Consortium for Scientific Research, New Delhi, India, for providing the financial assistance in the form of grants and fellowships.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ghosh, R.K., Kar, T., Dutta, B. et al. Aberration in the structural paradigm of lens protein α crystallin by UV-C irradiation. Appl Biol Chem 61, 281–287 (2018). https://doi.org/10.1007/s13765-018-0351-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13765-018-0351-y