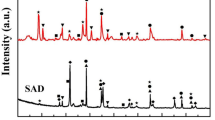
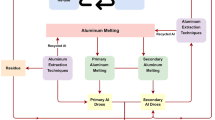
Abstract
Aluminum dross (AD) from aluminum alloy production is harmful for the environment, while it also contains valuable metal and oxides. Therefore, its comprehensive utilization in an environmental-friendly route is of great importance for the sustainable development of the aluminum industry. Therefore, a green route to comprehensively reuse AD was proposed in this paper. Effects of different processing conditions on extraction behaviors of aluminum, soluble salt and aluminum nitride (AlN) were studied. The extraction rate of aluminum can reach 96.5% with processing temperature of 750 °C, processing time of 40 min, and refining agent addition of 10 wt%. With the liquid–solid mass ratio of 10, the leaching time of 8 h, the leaching temperature of 90 °C and stirring speed of 300 rad/min, the maximum leaching rate of chlorine salt, fluorine salt and hydrolysis of AlN can reach 98.4%, 93.4% and 87.6%, respectively. The tailings after leaching treatment can be used in a variety of applications, such as the production of cement, ceramic and refractory which can be used in the chemical and metallurgical industry.






Similar content being viewed by others
References
Abdulkadir A, Ajayi A, Hassan MI (2015) Evaluating the chemical composition and the molar heat capacities of a white aluminum dross. Energy Procedia 75:2099–2105. https://doi.org/10.1016/j.egypro.2015.07.326
Adeosun SO, Usman MA, Ayoola WA, Sekunowo IO (2012) Evaluation of the mechanical properties of polypropylene-aluminum-dross composite. ISRN Polym Sci 2012:1–6. https://doi.org/10.5402/2012/282515
Bruckard WJ, Woodcock JT (2009) Recovery of valuable materials from aluminium salt cakes. Int J Miner Process 93:1–5. https://doi.org/10.1016/j.minpro.2009.05.002
Bruckard WJ, Woodcock JT (2007) Characterisation and treatment of Australian salt cakes by aqueous leaching. Miner Eng 20:1376–1390. https://doi.org/10.1016/j.mineng.2007.08.020
Dash B, Das BR, Tripathy BC et al (2008) Acid dissolution of alumina from waste aluminium dross. Hydrometallurgy 92:48–53. https://doi.org/10.1016/j.hydromet.2008.01.006
Davies M, Smith P, Bruckard WJ, Woodcock JT (2008) Treatment of salt cakes by aqueous leaching and Bayer-type digestion. Miner Eng 21:605–612. https://doi.org/10.1016/j.mineng.2007.12.001
Ewais EMM, Khalil NM, Amin MS et al (2009) Utilization of aluminum sludge and aluminum slag (dross) for the manufacture of calcium aluminate cement. Ceram Int 35:3381–3388. https://doi.org/10.1016/j.ceramint.2009.06.008
Hashishin T, Kodera Y, Yamamoto T et al (2004) Synthesis of (Mg, Si)Al2O4 spinel from aluminum dross. J Am Ceram Soc 87:496–499
Hassankhani-Majd Z, Anbia M (2021) Recovery of valuable materials from phosphorus slag using nitric acid leaching followed by precipitation method. Resour Conserv Recycl 169:105547. https://doi.org/10.1016/j.resconrec.2021.105547
Hiraki T, Miki T, Nakajima K et al (2014) Thermodynamic analysis for the refining ability of salt flux for aluminum recycling. Mater (basel) 7:5543–5553. https://doi.org/10.3390/ma7085543
Hiraki T, Nagasaka T (2015) An easier upgrading process of aluminum dross residue by screening technique. J Mater Cycles Waste Manag 17:566–573. https://doi.org/10.1007/s10163-014-0283-5
Hong JP, Wang J, Chen HY et al (2010) Process of aluminum dross recycling and life cycle assessment for Al-Si alloys and brown fused alumina. Trans Nonferrous Met Soc China (engl Ed) 20:2155–2161. https://doi.org/10.1016/S1003-6326(09)60435-0
Huang XL, El BA, Arambewela M et al (2014) Characterization of salt cake from secondary aluminum production. J Hazard Mater 273:192–199. https://doi.org/10.1016/j.jhazmat.2014.02.035
Kim J, Biswas K, Jhon KW et al (2009) Synthesis of AlPO4-5 and CrAPO-5 using aluminum dross. J Hazard Mater 169:919–925. https://doi.org/10.1016/j.jhazmat.2009.04.035
Kudyba A, Akhtar S, Johansen I, Safarian J (2021) Aluminum recovery from white aluminum dross by a mechanically activated phase separation and remelting process. JOM 73:2625–2634. https://doi.org/10.1007/s11837-021-04730-x
Li A, Zhang H, Yang H (2014) Evaluation of aluminum dross as raw material for high-alumina refractory. Ceram Int 40:12585–12590. https://doi.org/10.1016/j.ceramint.2014.04.069
Li J, Wang J, Chen H, Sun B (2012) Microstructure observation of β-sialon-15R ceramics synthesized from aluminum dross. Ceram Int 38:3075–3080. https://doi.org/10.1016/j.ceramint.2011.12.006
Mahinroosta M, Allahverdi A (2018) Hazardous aluminum dross characterization and recycling strategies: a critical review. J Environ Manag 223:452–468. https://doi.org/10.1016/j.jenvman.2018.06.068
Manfredi O, Wuth W, Bohlinger I (1997) Characterizing the physical and chemical properties of aluminum dross. Jom 49:48–51. https://doi.org/10.1007/s11837-997-0012-9
Murayama N, Maekawa I, Ushiro H et al (2012) Synthesis of various layered double hydroxides using aluminum dross generated in aluminum recycling process. Int J Miner Process 110–111:46–52. https://doi.org/10.1016/j.minpro.2012.03.011
Ramaswamy P, Gomes SA, Ravichander NP (2019) Utilization of aluminum dross: refractories from industrial waste. IOP Conf Ser Mater Sci Eng. https://doi.org/10.1088/1757-899X/577/1/012101
Roy RR, Sahai Y (1997) Interfacial tension between aluminum alloy and molten salt flux. Mater Trans JIM 38:546–552. https://doi.org/10.2320/matertrans1989.38.546
Saravanakumar P, Bhoopashram J, Kavin Prasath M, Jaycharan M (2017) Role of salt fluxes in aluminium refining: a review. Int J Latest Eng Manag Res 2:45–51
Sarker MSR, Alam MZ, Qadir MR et al (2015) Extraction and characterization of alumina nanopowders from aluminum dross by acid dissolution process. Int J Miner Metall Mater 22:429–436. https://doi.org/10.1007/s12613-015-1090-2
Shi J, Qiu Y, Yu B et al (2021a) Titanium extractive from titania-bearing blast furnace slag: a review. JOM. https://doi.org/10.1007/s11837-021-05049-3
Shi J, Qiu Y, Yu B et al (2021b) Equilibrium phase relations of the CaO-SiO2-Ti3O5 system at 1400 °C and a p(O2 of 10–16 atm. JOM. https://doi.org/10.1007/s11837-021-05049-3
Shi M, Li Y, Shi J (2021c) Fabrication of periclase and magnesium aluminate spinel refractory from washed residue of secondary aluminum dross. Ceram Int. https://doi.org/10.1016/j.ceramint.2021.11.312
Shinzato MC, Hypolito R (2005) Solid waste from aluminum recycling process: characterization and reuse of its economically valuable constituents. Waste Manag 25:37–46. https://doi.org/10.1016/j.wasman.2004.08.005
Tsakiridis PE (2012) Aluminium salt slag characterization and utilization—a review. J Hazard Mater 217–218:1–10. https://doi.org/10.1016/j.jhazmat.2012.03.052
Tsakiridis PE, Oustadakis P, Agatzini-Leonardou S (2013) Aluminum recovery during black dross hydrothermal treatment. J Environ Chem Eng 1:23–32. https://doi.org/10.1016/j.jece.2013.03.004
Ünlü N, Drouet MG (2002) Comparison of salt-free aluminum dross treatment processes. Resour Conserv Recycl 36:61–72. https://doi.org/10.1016/S0921-3449(02)00010-1
Utigard TA, Roy RR, Friesen K (2001) Properties of fluxes used in molten aluminium processing. High Temp Mater Process 20:303–307. https://doi.org/10.1515/HTMP.2001.20.3-4.303
Wan B, Li W, Liu F et al (2020) Determination of fluoride component in the multifunctional refining flux used for recycling aluminum scrap. J Mater Res Technol 9:3447–3459. https://doi.org/10.1016/j.jmrt.2020.01.082
Xiao Y, Reuter MA, Boin U (2005) Aluminium recycling and environmental issues of salt slag treatment. J Environ Sci Heal 40:1861–1875. https://doi.org/10.1080/10934520500183824
Yang Q, Li Q, Zhang G et al (2019) Investigation of leaching kinetics of aluminum extraction from secondary aluminum dross with use of hydrochloric acid. Hydrometallurgy 187:158–167. https://doi.org/10.1016/j.hydromet.2019.05.017
Yoshimura HN, Abreu AP, Molisani AL et al (2008) Evaluation of aluminum dross waste as raw material for refractories. Ceram Int 34:581–591. https://doi.org/10.1016/j.ceramint.2006.12.007
Zhang Y, Guo Z, Han Z, Xiao X (2018) Effect of rare earth oxides doping on MgAl2O4 spinel obtained by sintering of secondary aluminium dross. J Alloys Compd 735:2597–2603. https://doi.org/10.1016/j.jallcom.2017.11.356
Zhang Y, Guo ZH, Xiao XY et al (2019) Feasibility of aluminum recovery and MgAl2O4 spinel synthesis from secondary aluminum dross. Int J Miner Metall Mater 26:309–318. https://doi.org/10.1007/s12613-019-1739-3
Acknowledgements
The authors acknowledge the financial support from the National Natural Science Foundation of China (51834004, 51774076, 51474057 and 51704062).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Consent for publication
The manuscript is original. It has not been published previously by any of the author and even not under the consideration in any other journal at the time of submission.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Editorial responsibility: Maryam Shabani.
Rights and permissions
About this article
Cite this article
Shi, M., Li, Y. & Ni, P. Recycling valuable elements from aluminum dross. Int. J. Environ. Sci. Technol. 19, 12069–12078 (2022). https://doi.org/10.1007/s13762-022-03925-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-022-03925-2