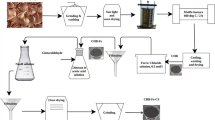
Abstract
The removal of tetracycline (TC) and sulfamethazine (SMZ) by adsorption was carried out using ferromagnetic carbon prepared from coffee residue (FAC).The aim of this study was the highlight of some parameters effect (equilibrium time, initial concentration of the antibiotic, (adsorbent dosage) and pH of the solution) on the adsorption capacity of the carbon. The retention efficiency of both substances is high for the initial concentration of the antibiotic less than 5 mg/L, as 97% and 62% were the yields obtained at a concentration of 5 mg/L. Both systems reached equilibrium at about 120 min and 0.5 g of carbon was the optimal mass. TC was preferentially adsorbed to the carbon than (SMZ) due to its zwitterionic form under the operating conditions used. The removal of (SMZ) was not affected by the medium pH compared to TC which showed better adsorption for acidic pH (3 ≤ pH ≤ 7). The kinetic study for both molecules showed that the kinetic data followed the pseudo-second-order model. Chemical regeneration of two spent carbons using a sodium hydroxide solution at pH = 12 provided significant adsorption capacities after 5successive adsorption–desorption cycles for the first carbon (CMC) and 4 cycles for the second (SMC…).













Similar content being viewed by others
References
Adams C, Wang Y, Loftin K, Meyer M (2002) Removal of antibiotics from surface and distilled water in conventional water treatment processes. J Environ Eng 128:253–260. https://doi.org/10.1061/(ASCE)0733-9372(2002)128:3(253)
Aksu Z, Tunç Ö (2005) Application of biosorption for penicillin G removal: comparison with activated carbon. Pro Bioch 40:831–847. https://doi.org/10.1016/j.procbio.2004.02.014
Berenguer R, Marco LJ, Quijada C, Cazorla AD, Morallon E (2010) Electrochemical regeneration and porosity recovery of phenol-saturated granular activated carbon in an alkaline medium. Carbon 48:2734–2745. https://doi.org/10.1016/j.carbon.2010.03.071
Cazetta AL, Vargas AM, Nogami EM, Kunita MH, Guilherme MR, Martins AC, Silva TL, Moraes JC, Almeida VC (2011a) NaOH-activated carbon of high surface area produced from coconut shell: kinetics and equilibrium studies from the methylene blue adsorption. Chem Eng J 174:117–125. https://doi.org/10.1016/j.cej.2011.08.058
Cazetta A, Vargas AMM, Nogami E, Kunita MH, Guilherme MR, Martins AC, Silva TL, Moraes JCG, Almeida VC (2011b) NaOH- activated carbon of high surface area produced from coconut shell: Kinetics and equilibrium studies from the methylene blue adsorption. Chem Eng J 174(117):125. https://doi.org/10.1088/1757-899X/334/1/012051
Chen S, Zhang J, Zhang C, Yue Q, Li Y, Li C (2010) Equilibriumand kinetic studies of methyl orange and methyl violet adsorption on activated carbon derived from Phragmitesaustralis. Desalination 252:149–156. https://doi.org/10.1016/j.desal.2009.10.010
Choi KJ, Kim SG, Kim SH (2008) Removal of tetracycline and sulfonamide classes of antibiotic compound by powdered activated carbon. Environ Technol 29:333–342. https://doi.org/10.1080/09593330802102223
Dealda MJ, Diazcruz S, Petrovic M, Barcelo D (2003) Liquid chromatography –tandem mass spectrometry of selected emerging pollutants (steroid sex hormones. drugs. and alkylphenolic surfactants in the environment. J Chromatogr A 1000:503–526. https://doi.org/10.1016/s0021-9673(03)00509-0
Dinesh M, Ankur S, Singh VK, Alexandre-Franco M, Pittman CU Jr (2011) Development of magnetic activated carbon from almond shells for trinitrophenol removal from water. Chem Eng J 172:1111–1125. https://doi.org/10.1016/j.cej.2011.06.054
Fan HT, Shi LQ, Shen H, Chen X, Xie KP (2016) Equilibrium, isotherm, kinetic and thermodynamic studies for removal of tetracycline antibiotics by adsorption onto hazelnut shell derived activated carbons from aqueous media. RSC Adv 6:109983–109991. https://doi.org/10.1039/C6RA23346E
Foo KY, Hameed BH (2012) Textural porosity surface chemistry and adsorptive properties of durian shell derived activated carbon prepared by microwave assisted NaOH activation. Chem Eng J 187:53–62. https://doi.org/10.1016/j.cej.2012.01.079
Giles CH, Smith D, Huitson A (1974) A general treatment and classification of the solute adsorption isotherm. I. Theorical. J Colloid Interface Sci 47:755–765. https://doi.org/10.1016/0021-9797(74)90252-5
He J, Dai J, Zhang T, Sun J, Xie A, Tian S, Yan Y, Huo P (2016) Preparation of highly porous carbon from sustainable α-cellulose for superior removal performance of tetracycline and sulfamethazine from water. RSC Adv 6:28023–28033. https://doi.org/10.1039/C6RA00277C
Heberer T (2002) Occurrence, fate and removal of pharmaceutical residues in the aquatic environment: a review of recent research data. Toxicol Lett 131:5–17. https://doi.org/10.1016/s0378-4274(02)00041-3
Hu XJ, Wang JS, Liu YG, Li X, Zeng GM, Bao ZL, Zeng X, X., Chen, AW F, (2011a) Adsorption of chromium(VI) by ethylenediamine-modified cross-linked magnetic chitosan resin: Isotherms. kinetics and thermodynamics. J Hazard Mater 185:306–314. https://doi.org/10.1016/j.jhazmat.2010.09.034
Hu XJ, Wang JS, Liu YG, Li X, Zeng GM, Bao ZL, Zeng XX, Chen AWF (2011b) Adsorption of chromium(VI) by ethylenediamine-modified cross-linked magnetic chitosan resin: isotherms, kinetics and thermodynamics. J Hazard Mater 185:306–314. https://doi.org/10.1016/j.jhazmat.2010.09.034
Hu X, Zhao Y, Wang H, Tan X, Yang Y, Liu Y (2017) Efficient removal of tetracycline from aqueous media with a Fe3O4 Nanoparticles @graphene oxide nanosheets assembly. Int J Environ Res Public Health 14:1480–1495. https://doi.org/10.3390/ijerph14121495
Ijaz H, Yang L, Junwen Q, Jiansheng L, Xiuyun S, Jinyou Sh, Weiqing H, Lianjun W (2018) Synthesis of magnetic yolk-shell mesoporous carbon architecture for the effective adsorption of sulfamethazine drug. Microporous Mesoporous Mater 255:110–118. https://doi.org/10.1016/j.micromeso.2017.07.027
Jing Ch, Zhen Sh, Xiude H, Sch E, Chunyue C, Qingjie G, Hongjing T (2020) Adsorption of tetracycline by shrimp shell waste from aqueous solutions: adsorption isotherm, kinetics modeling, and mechanism. ACS Omega 5:3467–3477. https://doi.org/10.1021/acsomega.9b03781
Jodeh S, Abdelwahab F, Jaradat N, Warad I, Jodeh W (2016) Adsorption of diclofenac from aqueous solution using Cyclamen persicum tubers based activated carbon (CTAC). J Assoc Arab Univ Basic Appl Sci 20:32–38. https://doi.org/10.1016/j.jaubas.2014.11.002
Joss A, Keller E, Alder AC, Göbel A, McArdell Ch, Ternes T, Siegrist H (2005) Removal of pharmaceuticals and fragrances in biological wastewater treatment Wat. Resear 39:3139–3152. https://doi.org/10.1016/j.watres.2005.05.031
Kim S, Shon H, Ngo HH (2010) Adsorption characteristics of antibiotics trimethoprim on powdered and granular activated carbon. J Ind Eng Chem 16:344–349. https://doi.org/10.1016/j.jiec.2009.09.061
Lee S, Lee JS, Song MK, Ryu JC, An B, Lee CG, Park C, Lee SH, Choi JW (2015) Effective regeneration of an adsorbent for the removal of organic contaminants developed based on UV radiation and toxicity evaluation. React Funct Polym 95:62–70. https://doi.org/10.1016/j.reactfunctpolym.2015.08.008
Leyva RR, Ocampo PRJ, Mendoza B (2012) External mass transfer and hindered diffusion of organic compounds in the adsorption on activated carbon cloth. Chem Eng J 183:141–151. https://doi.org/10.1016/j.cej.2011.12.046
Liu Y, Liu X, Dong W, Zhang L, Kong Q, Wang W (2017) Efficient adsorption of sulfamethazine onto modified activated carbon: a plausible adsorption mechanism. Sci Rep. https://doi.org/10.1038/s41598-017-12805-6
Mehrjouei M, Müller S, Möller D (2014) Energy consumption of three different advanced oxidation methods for water treatment: a cost-effectiveness study. J Clean Prod 65:178–183. https://doi.org/10.1016/j.jclepro.2013.07.036
Mehta D, Mazumdar S, Singh SK (2015) Magnetic adsorbents for the treatment of water/wastewater—a review. J Water pro Eng 7:244–265. https://doi.org/10.1016/j.jwpe.2015.07.001
Oliveira LCA, Rios RVRA, Fabris JD (2002) Activated carbon/iron oxide magnetic composites for the adsorption of contaminants in water. Carbon 40:2177–2183. https://doi.org/10.1016/S0008-6223(02)00076-3
Paxéus N (2004) Removal of selected non-steroidal anti-inflammatory drugs (NSAIDs). gemfibrozil. carbamazepine.β-blockers trimethoprin and triclosan in conventional wastewater treatment plants in five EU countries and their discharge to the aquatic environment. Water Sci Technol 50:253–260. https://doi.org/10.2166/wst.2004.0335
Peiris C, Gunatilake SR, Mlsna TE, Mohan D, Vithanage M (2017) Biochar based removal of antibiotic sulfonamides and tetracyclines in aquatic environments: a critical review. Biores Technol 246:150–159. https://doi.org/10.1016/j.biortech.2017.07.150
Pérez S, Barceló D (2007) Application of advanced MS techniques to analysis and identification of human and microbial metabolites of pharmaceuticals in the aquatic environment. Trends Analyt Chem 50:494–514. https://doi.org/10.1016/j.trac.2007.05.004
Pouretedal H, Sadegh N (2014) Effective removal of amoxicillin, cephalexin, tetracycline and penicillin G from aqueous solutions using activated carbon nanoparticles prepared from vine wood. J Water pro Eng 1:64–73. https://doi.org/10.1016/j.jwpe.2014.03.006
Rostamian R, Behnejad H (2018) A comprehensive adsorption study and modeling of antibiotics as a pharmaceutical waste by graphene oxide nanosheets. Ecotoxicol Environ Saf 147:117–123. https://doi.org/10.1016/j.ecoenv.2017.08.019
Song Q, Fang Y, Liu Z, Li L, Wang Y, Liang J, Huang Y, Lin J, Hu L, Zhang J, Tang C (2017) The performance of porous hexagonal BN in high adsorption capacity towards antibiotics pollutants from aqueous solution. Chem Eng J 325:71–79. https://doi.org/10.1016/j.cej.2017.05.057
Sun P, Xu L, Li J, Zhai P, Zhang H, Zhang Z, Zhu W (2018) Hydrothermal synthesis of mesoporous Mg3Si2O5(OH)4 microspheres as high-performance adsorbents for dye removal. Chem Eng J 334:377–388. https://doi.org/10.1016/j.cej.2017.09.120
Tang Y, Guo H, Xiao L, Yu S, Gao N, Wang Y (2013) Synthesis of reduced grapheme oxide/magnetite composites and investigation of their adsorption performance of fluoroquinolone antibiotics. Colloids Surf Physicochem Eng Asp 424:74–80. https://doi.org/10.1016/j.colsurfa.2013.02.030
Torres PJ, Gérente C, Andrès Y (2012) Sustainable activated carbons from agricultural residues dedicated to antibiotic removal by adsorption. Chin J Chem Eng 20:524–529. https://doi.org/10.1016/S1004-9541(11)60214-0
Viegas RMC, Campinas M, Costa H, Rosa MJ (2014) How do the HSDM and Boyd’s model compare for estimating intraparticle diffusion coefficients in adsorption processes. Adsorption 20:737–746. https://doi.org/10.1007/s10450-014-9617-9
Wang DW, Li F, Lu M, Cheng HM (2008) Synthesis and dye separation performance of ferromagnetic hierarchical porous carbon. Carbon 46:1593–1599. https://doi.org/10.1016/j.carbon.2008.06.052
Zhang J, Sun W, Bergman L, Rosenholm JM, Lindén M, Wu G, Xu H, Hong-C G (2012) Magnetic mesoporous silica nanospheres as DNA/drug carrier. Mater Lett 67:379–382. https://doi.org/10.1016/j.matlet.2011.09.086
Zhang BB, Xu JC, Xin PH, Han YB, Hong B, Jin HX, Jin DF, Peng XL, Li J, Gong J, Ge HL, Zhu ZW, Wang XQ (2015) Magnetic properties and adsorptive performance of manganese–zinc ferrites/activated carbon nanocomposites. J Solid State Chem 221:302–305. https://doi.org/10.1016/j.jssc.2014.10.020
Zhang X, Guo W, Ngo HH, Wen H, Li N, Wu W (2016) Performance evaluation of powdered activated carbon for removing 28 types of antibiotics from water. J Environ Manag 172:193–200. https://doi.org/10.1016/j.jenvman.2016.02.038
Zhou Q, Li Z, Shuang C, Li A, Zhang M, Wang M (2012) Efficient removal of tetracycline by reusable magnetic microspheres with a high surface area. Chem Eng J 210:350–356. https://doi.org/10.1016/j.cej.2012.08.081
Acknowledgements
The authors are thankful to Miss BRADAI and Miss BENAMOR for providing facilities to realise all the solutions analysis. The authors are also grateful to Mr BENIDER and Mr MAMAS to have performed the structural and thermal analyses for the different prepared adsorbents.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Editorial responsibility: Nour Sh. El-Gendy.
Rights and permissions
About this article
Cite this article
Khenniche, L., Chemache, Z., Saidou-Souleymane, M. et al. Elimination of antibiotics by adsorption on ferromagnetic carbon from aqueous media: regeneration of the spent carbon. Int. J. Environ. Sci. Technol. 19, 9571–9586 (2022). https://doi.org/10.1007/s13762-021-03808-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-021-03808-y