Abstract
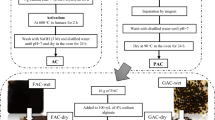
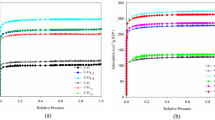
Hydrofluoric acid is used in large volumes during solar panels manufacturing for cleaning purpose. It results in the generation of wastewater containing high concentrations of fluoride. Chemical precipitation is commonly used to remove most of fluoride from the wastewater. It reduces fluoride concentration to ~ 20 mg/L, whereas the discharge standard is set to 2 mg/L. Adsorption is preferable when dealing with diluted wastewater. This study evaluated the adsorption of fluoride using two low-cost adsorbents: granular palm shell activated carbon and its modification with the prepared chitosan of prawn shells. The effects of pH, dosage, contact time and initial fluoride concentration were studied in batch mode adsorption experiments. The highest removal efficiencies of fluoride by two adsorbents comprised 46% and 55%, respectively, at pH 2 and pH 7. The contrasting behavior of the two adsorbents in relation to pH is attributed by the adsorbents surface charge associated with the surface functional groups identified by FTIR. The adsorption isotherm modeling showed better fit to the Langmuir model for both adsorbents. Adsorption kinetics results fitted well into the pseudo-second-order kinetics model suggesting chemisorption mechanism of fluoride removal.






Similar content being viewed by others
References
Abri A, Tajbakhsh M, Sadeghi A (2018) Adsorption of fluoride on a chitosan-based magnetic nanocomposite: equilibrium and kinetics studies. Water Supply 19:40–51. https://doi.org/10.2166/ws.2018.050
Akbari H, Jorfi S, Mahvi AH, Yousefi M, Balarak D (2018) Adsorption of fluoride on chitosan in aqueous solutions: determination of adsorption kinetics. Res Rep Fluoride 51:319–327
Alagumuthu G, Rajan M (2010) Equilibrium and kinetics of adsorption of fluoride onto zirconium impregnated cashew nut shell carbon. Chem Eng J 158:451–457
Amalraj A, Pius A (2017) Removal of Fluoride from drinking water using aluminum hydroxide coated activated carbon prepared from bark of Morinda tinctoria. Appl Water Sci 7:2653–2665. https://doi.org/10.1007/s13201-016-0479-z
Araga R, Soni S, Sharma S (2017) Fluoride adsorption from aqueous solution using activated carbon obtained from KOH-treated jamun (Syzygium cumimi) seed. J Environ Chem Eng 6:5608–5616. https://doi.org/10.1016/j.jece.2017.10.023
Awalludin M, Sulaiman O, Hashim R, Aidawati W (2015) An overview of the oil palm industry in Malaysia and its waste utilization through thermochemical conversion specifically via liquefaction. Renew Sustain Energy Rev 50:1469–1484. https://doi.org/10.1016/j.rser.2015.05.085
Bhantnagar A, Kumar E, Silanpaa M (2011) Fluoride removal from water by adsorption—A review. Chem Eng J 171:811–840. https://doi.org/10.1016/j.cej.2011.05.028
Bhaumik R, Mondal NK (2015) Adsorption of fluoride from aqueous solution by a new low-cost adsorbent: thermally and chemically activated coconut fibre dust. Clean Technol Environ Policy 17:2157–2172. https://doi.org/10.1007/s10098-015-0937-6
Choong CE, Wong KT, Jang SB, Nah IW, Choi J, Ibrahim S, Yoon Y, Jang M (2020) Fluoride removal by palm shell waste based powdered activated carbon versus functionalized carbon with magnesium silicate: implications for their application in water treatment. Chemosphere 239:124765
Christe K, Schneider S, Gutmann V (2019) Halogen. Encyclopædia Britannica. https://www.britannica.com/science/halogen. Accessed 2 Feb 2020
Daifullah AA, Yakout SM, Elreefy SA (2007) Adsorption of fluoride in aqueous solution using KMnO4-modified activated carbon derived from stream pyrolysis of rice straw. J Hazard Mater 147:633–643. https://doi.org/10.1016/j.jhazmat.2007.01.062
DOE (2010) Environmental requirement: a guide for investors. Department of Environment Malaysia. http://www.doe.gov.my/eia/wp-content/uploads/2012/03/A-Guide-For-Investors1.pdf. Accessed 5 Feb 2020
Dhawan R, Goyal M, Bhasin KK (2016) Influence of impregnants on adsorption of dimethylsulfide vapors by activated carbon. Mater Today Proc 4:10515–10519. https://doi.org/10.1016/j.matpr.2017.06.411
Elieh-Ali-Komi D, Hamblin MR (2016) Chitin and chitosan: production and application of versatile biomedical nanomaterials. Int J Adv Res (Indore) 4(3):411–427
Foungchuen J, Pairin N, Phalakornkul C (2016) Impregnation of chitosan onto activated carbon for adsorption of carbon dioxide: biohydrogen purification. KMUTNB Int J Appl Sci Technol 9:197–206
Freundlich H (1906) Adsorpt Solut Phys Chem 57:384–410
Getachew T, Hussen A, Rao VM (2015) Defluoridation of water by activated carbon prepared from banana (Musa paradisiaca) peel and coffee (Coffea arabica) husk. Int J Environ Sci Technol 12:1857–1866. https://doi.org/10.1007/s13762-014-0545-8
Habuda-Stanić M, Ravančić M, Flanagan A (2014) A review on adsorption of fluoride from aqueous solution. Materials 7(9):6317–6366. https://doi.org/10.3390/ma7096317
Hegde RM, Rego RM, Potla KM, Kurkuri MD, Kigga M (2020) Bio-inspired materials for defluoridation of water: a review. Chemosphere 253:126657
Heidari F, Rasoulianboroujeni M, Bahrololoom ME, Tayebi L (2016) Preparation of chitosan from natural shrimp shells with different deacetylation degree. Mater Res Innovation 1:1–5
Herawan SG, Hadi MS, Ayob MR, Putra A (2013) Characterization of activated carbon from oil-palm shell by CO2 activation with no holding carbonization temperature. Sci World J. https://doi.org/10.1155/2013/624865
Ho Y, McKay G (1998) A comparison of chemisorption kinetic models applied to pollutant removal on various sorbents. Process Saf Environ Prot 76:332–340. https://doi.org/10.1205/095758298529696
Huang CJ, Liu JC (1999) Precipitate flotation of fluoride-containing wastewater from a semiconductor manufacturer. Water Res 33(16):3404–3412
Issabayeva G, Aroua MK, Chen SK (2012) Biomodification of palm shell activated carbon using Aspergillus niger and Bacillus subtilis and its effect on the adsorption of lead ions from aqueous solutions. African J Biotechnol 11(82):14812–14821. https://doi.org/10.5897/AJB12.1901
Jagtap S, Thakre D, Wanjari S, Kamble S, Labhsetwar N, Rayalu S (2008) New modified chitosan-based adsorbent for defluoridation of water. J Colloid Interface Sci 332(2):280–290. https://doi.org/10.1016/j.jcis.2008.11.080
Jha S, Singh R, Damodaran T, Mishra V, Sharma D, Rai D (2013) Fluoride in groundwater: toxicological exposure and remedies. J Toxicol Environ Health Part B 16(1):52–66. https://doi.org/10.1080/10937404.2013.769420
Lagergren SY (1898) Zur Theorie der sogenannten Adsorption gelöster Stoffe, Kungliga Svenska Vetenskapsakad. Handlingar 24:1–39
Langmuir I (1918) Adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 40:1361–1403. https://doi.org/10.1021/ja02242a004
Loganathan P, Vigneswaran S, Kandasamy J, Naidu R (2013) Defluoridation of drinking water using adsorption processes. J Hazard Mater 248–249:1–19. https://doi.org/10.1016/j.jhazmat.2012.12.043
Mouelhi M, Giraudet S, Amrane A, Hamrouni B (2016) Competitive adsorption between fluoride and natural organic matter onto activated alumina. Environ Technol 37(18):2326–2336. https://doi.org/10.1080/09593330.2016.1149521
Okoroigwe EC, Saffron CM, Kamdem PD (2014) Characterization of palm kernel shell for materials reinforcement and water treatment. J Chem Eng Mater Sci 5:1–6. https://doi.org/10.5897/JCEMS2014.0172
Pokhrel S, Lach R, Grellmann W, Wutzler A, Lebek W, Godehardt R, Yadav PN, Adhikari R (2016) Synthesis of chitosan from prawn shell and characterization of its structural and antimicrobial properties. Nepal J Sci Technol 17(1):5–9. https://doi.org/10.3126/njst.v17i1.25056
Pongener C, Bhomick PC, Supong A, Baruah M, Sinha UB, Sinha D (2018) Adsorption of fluoride onto activated carbon synthesized from Mahinot esculenta biomass—Equilibrium, kinetic and thermodynamics studies. J Environ Chem Eng 6(2):2383–2389. https://doi.org/10.1016/j.jece.2018.02.045
Singh J, Singh P, Singh A (2016) Fluoride ions vs removal technologies: a study. Arabian J Chem 9(6):815–824. https://doi.org/10.1016/j.arabjc.2014.06.005
Swain SK, Dey RK, Islam M, Patel RK, Jha U, Patnaik T, Airoldi C (2009) Removal of fluoride from aqueous solution using aluminum-impregnated chitosan biopolymer. Sep Sci and Technol 44(9):2096–2116. https://doi.org/10.1080/01496390902881212
Tahaikt M, Habbani RE, Haddou AA, Achary I, Amor Z, Taky M, Alami A, Boughriba A, Hafsi M, Elmidaoui A (2007) Fluoride removal from groundwater by nanofiltration. Desalination 1:46–53
Vithanage M, Bhattacharya P (2015) Fluoride in the environment: sources, distribution and defluoridation. Environ Chem Letters 13(2):1–17. https://doi.org/10.1007/s10311-015-0496-4
VMinteq (2013) Visual MINTEQ freeware chemical equilibrium model. https://vminteq.lwr.kth.se/. Accessed 16 Mar 2020
Waghmare SS, Arfin T (2015) Fluoride removal from water by various technique: review. Int J Innovative Sci Eng Technol 2(9):560–571
WHO (2004) Fluoride in drinking water. World Health Organization. https://www.who.int/water_sanitation_health/dwq/chemicals/fluoride.pdf. Accessed 10 Feb 2020
Xiang Q, Liang Y, Chen L, Wang C, Chen B, Chen X, Zhou M (2003) Effects of fluoride in drinking water on children’s intelligence. Fluoride 36:84–94
Zhang J, Chen N, Tang Z, Yu Y, Hu QL, Feng CP (2015) Adsorption of fluoride from aqueous solution onto Fe-impregnated chitosan and mechanism. Phys Chem Chem Phys 17:12041–12050. https://doi.org/10.1039/C5CP00817D
Acknowledgements
We would like to express our gratitude to the UTARRF Grant IPSR/RMC/UTARRF/2019-C2/G05 and CPAMR Research Centre for the financial support of our research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial responsibility: G. Ravindran.
Rights and permissions
About this article
Cite this article
Issabayeva, G., Wong, S.H., Pang, C.Y. et al. Fluoride removal by low-cost palm shell activated carbon modified with prawn shell chitosan adsorbents. Int. J. Environ. Sci. Technol. 19, 3731–3740 (2022). https://doi.org/10.1007/s13762-021-03448-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-021-03448-2