Abstract
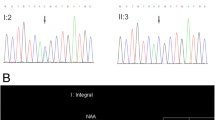
Creatine is the main source of energy for the brain. Primary creatine deficiency syndromes (PCDSs) are inborn error of metabolism of creatine synthesis. Symptoms of central nervous system involvement are the most common clinical manifestations in these disorders. We reviewed medical records of all genetically confirmed patients diagnosed by whole exome sequencing who were referred to Myelin and Neurodegenerative Disorders Clinic, Children’s Medical Center, Tehran, Iran, from May 2016 to Dec 2018. A literature review was conducted on clinical and genomic variability of PCDS to compare our patients with previously reported cases. We report two patients with creatine deficiency among a cohort of 550 registered cases out of which 200 patients had a genetically confirmed neurodegenerative disorder diagnosis. The main complain in the first patient with creatine transporter (CRTR) deficiency was seizure and genetic study in this patient identified a novel hemizygote variant of “c.92 > T; p.Pro31Leu” in the first exon of SLC6A8 gene. The second patient with guanidinoacetate methyltransferase (GAMT) deficiency had an unknown motor and speech delay as the striking manifestation and molecular assay revealed a novel homozygote variant of “c.134G > A; p.Trp45*” in the first exon of GAMT gene. PCDSs usually are associated with nonspecific neurologic symptoms. The first presented case had a mean delayed diagnosis of 5 years. Therefore, in children with unexplained neurologic features including developmental delay and/or regression, mental disability and repeated seizures without any significant findings in metabolic studies, PCDSs can be considered as a differential diagnosis and molecular analysis can be helpful for the precise diagnosis and treatment.




Similar content being viewed by others
References
Cecil KM, Salomons GS, Ball WSJ, Wong B, Chuck G, Verhoeven NM, Jakobs C, DeGrauw TJ (2001) Irreversible brain creatine deficiency with elevated serum and urine creatine: a creatine transporter defect? Ann Neurol 49:401–404
Stockler S, Holzbach U, Hanefeld F et al (1994) Creatine deficiency in the brain: a new, treatable inborn error of metabolism. Pediatr Res 36:409–413
Andres RH, Ducray AD, Schlattner U, Wallimann T, Widmer HR (2008) Functions and effects of creatine in the central nervous system. Brain Res Bull 76:329–343
Thurm A, Himelstein D, DʼSouza P, Rennert O, Jiang S, Olatunji D, Longo N, Pasquali M, Swedo S, Salomons GS, Carrillo N (2016) Creatine transporter deficiency: screening of males with neurodevelopmental disorders and neurocognitive characterization of a case. J Dev Behav Pediatr 37(4):322–326
Valle D, Simell O (2001) The hyperornithinemias. In: Scriver CR, Beaudet A, Sly WS, Valle D (eds) The metabolic and molecular bases of inherited disease. McGraw Hill, New York, pp 1857–1896
van de Kamp M, Betsalel OT, Mercimek-Mahmutoglu S, Abulhoul L, Grünewald S, Anselm I, Azzouz H, Bratkovic D et al (2013) Phenotype and genotype in 101 males with X-linked creatine transporter deficiency. J Med Genet 50(7):463–472
Stockler-Ipsiroglu S, Mercimek-Mahmutoglu S, Solomons G (2012) Creatine deficiency syndromes. In: Saudubray JM, van den Berghe G, Walter JH (eds) Inborn metabolic diseases, diagnosis and treatment, 5th edn. Springer, New York, pp 239–247
Mercimek-Mahmutoglu S, Connolly MB, Poskitt KJ, Horvath GA, Lowry N, Salomons GS, Casey B, Sinclair G, Davis C, Jakobs C, Stockler-Ipsiroglu S (2010) Treatment of intractable epilepsy in a female with SLC6A8 deficiency. Mol Genet Metab 101(4):409–412
Schulze A, Battini R (2007) Pre-symptomatic treatment of creatine biosynthesis defects. Subcell Biochem 46:167–181
Aydin HI (2018) Creatine transporter deficiency in two brothers with autism spectrum disorder. Indian Pediatr 55(1):67–68
Chun-Hui Hu, Fan Yu-Ying, Wang Long-Fei, Tao Yu, Wang Xiao-Ming, Wang Hua (2015) Diagnosis of X-linked creatine transporter deficiency in a patient from Northeast China. Neurol Asia 20(2):197–201
Heussinger N, Saake M, Mennecke A, Dörr HG, Trollmann R (2017) Variable white matter atrophy and intellectual development in a family with X-linked creatine transporter deficiency despite genotypic homogeneity. Pediatr Neurol 67:45–52
van de Kamp JM, Errami A, Howidi M, Anselm I, Winter S, Phalin-Roque J, Osaka H, van Dooren SJM, Mancini GM, Steinberg SJ, Salomons GS (2014) Genotype–phenotype correlation of contiguous gene deletions of SLC6A8, BCAP31 and ABCD1. Clin Genet 87(2):141–147
Kato H, Miyake F, Shimbo H, Ohya M, Sugawara H, Aida N, Anzai R, Takagi M, Okuda M, Takano K, Wada T, Iai M, Yamashita S, Osaka H (2014) Urine screening for patients with developmental disabilities detected a patient with creatine transporter deficiency due to a novel missense mutation in SLC6A8. Brain Dev 36(7):630–633
Nozaki F, Kumada T, Shibata M, Fujii T, Wada T, Osaka H (2015) A family with creatine transporter deficiency diagnosed with urinary creatine/creatinine ratio and the family history: the third Japanese familial case. No to Hattatsu 47(1):49–52
Ardon O, Procter M, Maoab R, Longo N, Landau YE, ShilonHadass A, Gabis LV, Hoffmann C, Tzadok M, Heimerd G, Sadac S, Ben-Zeev B, Aniksterd Y (2016) Creatine transporter deficiency: novel mutations and functional studies. Mol Genet Metab Rep 30(8):20–23
Dreha-Kulaczewski S, Kalscheuer V, Tzschach A, Hu H, Helms G, Brockmann K, Weddige A (2014) A novel SLC6A8 mutation in a large family with x-linked intellectual disability: clinical and proton magnetic resonance spectroscopy data of both hemizygous males and heterozygous females. JIMD Rep 13:91–99
Nash SR, Giros B, Kingsmore SF et al (1994) Cloning, pharmacological characterization, and genomic localization of the human creatine transporter. Recept Channels 2(2):165–174
Aydin Halil Ibrahim, Mujgan Sonmez F (2017) A novel mutation in guanidinoacetate methyltransferase (GAMT) deficiency in two patients associated with epilepsy, developmental delay, hyperactivity, autistic behavior. Eur J Paediatr Neurol 21:123–140
Pacheva I, Ivanov I, Penkov M, Kancheva D, Jordanova A, Ivanova M (2016) Creatine deficiency syndrome could be missed easily: a case report of guanidinoacetate methyltransferase deficiency presented with neurodevelopmental delay, seizures, and behavioral changes, but normal structural MRI. Ann Clin Lab Sci 46(5):557–561
Stern WM, Winston JS, Murphy E, Cross JH, Sander JW (2017) Guanidinoacetate methyltransferase (GAMT) deficiency: rare but treatable epilepsy. Pract Neurol 17(3):207–211
Stockler-Ipsiroglu S, van Karnebeek C, Longo N, Korenke GC, Mercimek-Mahmutoglu S, Marquart I, Barshop B et al (2014) Guanidinoacetate methyltransferase (GAMT) deficiency: outcomes in 48 individuals and recommendations for diagnosis, treatment and monitoring. Mol Genet Metab J 111(1):16–25
Sun W, Wang Y, Zu Z, Jiang Y, Lu W, Wang H, Wu B, Zhang P, Peng X, Zhou H (2017) First reported Chinese case of guanidinoacetate methyltransferase deficiency in a 4-year old child. Clin Chim Acta 470:42–45
Khaikin Yannay, Sidky Sarah, Anastasi Jose Abdenur Arnaud, Ballhausen Diana et al (2018) Treatment outcome of twenty-two patients with guanidinoacetate methyltransferase deficiency: an international retrospective cohort study. Eur J Pediatr Neurol 22(3):369–379
Braissant O, Henry H (2008) AGAT, GAMT and SLC6A8 distribution in the central nervous system, in relation to creatine deficiency syndromes: a review. J Inherit Metab Dis 31:230–239
Nouioua S, Cheillan D, Zaouidi S, Salomons GS, Amedjout N, Kessaci F, Boulahdour N, Hamadouche T, Tazir M (2013) Creatine deficiency syndrome. A treatable myopathy due to arginine-glycine amidinotransferase (AGAT) deficiency. Neuromuscul Disord 23:670–674
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rostami, P., Hosseinpour, S., Ashrafi, M.R. et al. Primary creatine deficiency syndrome as a potential missed diagnosis in children with psychomotor delay and seizure: case presentation with two novel variants and literature review. Acta Neurol Belg 120, 511–516 (2020). https://doi.org/10.1007/s13760-019-01168-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13760-019-01168-6