Abstract
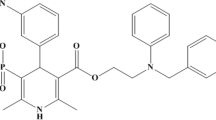
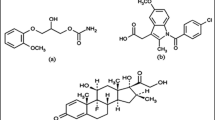
In this study, design of experiment approach was used for the development and optimization of a reversed phase HPLC method for the simultaneous determination of hydrochlorothiazide, amlodipine along with three angiotensin II receptor blockers olmesartan, losartan and valsartan in their laboratory-prepared mixtures and in three different commercial pharmaceutical single-pill combinations. The interactions between multiple chromatographic factors were studied using central composite design aiming to optimize both the resolution between the studied drugs and the run time. A total of 20 experiments were carried out and the coefficients of the second-order polynomial regression models were calculated to predict the optimal values of the HPLC method parameters. The significance of the models was evaluated by ANOVA, and the interactions between the three variables were illustrated by three-dimensional response surface plots. In this experimental design, the three studied key factors used for the optimization were A: pH, B: flow rate and C: acetonitrile (organic modifier) %. The five experimental responses observed were the four resolution factors and the run time. Obtained results were analyzed and the optimum chromatographic conditions predicted were 0.05 M potassium dihydrogen phosphate buffer (pH adjusted to 2.66 with ortho-phosphoric acid) and acetonitrile in the ratio (75.9:24.1, %v/v) as the mobile phase at flow rate 1.5 ml/min. Phenomenex Luna® 5 µm CN 100 Å LC column (250 × 4.6 mm) was used for the separation and quantification with photo diode array detection at 250 nm. Under these predicted optimum conditions, baseline separation with minimum resolution of 2.0 and run time less than 12 min were achieved, and the obtained experimental results showed good agreement with the predicted values. The optimized chromatographic separation was validated according to the International Conference on Harmonization guidelines to confirm linearity, specificity, accuracy, precision and robustness. The optimized RP-HPLC method has a wide range of application that can be used in quality control and routine analysis of the studied drugs in their different pharmaceutical single-pill combinations and laboratory mixtures aiming to reduce laboratory wastes, analysis time, cost and effort.







Similar content being viewed by others
References
N.S. Lakka, C. Kuppan, Principles of chromatography method development. Biochem. Anal. Tools Methods Bio-Molecules Stud. (2020). https://doi.org/10.5772/intechopen.89501
B. Nikolin, B. Imamović, S. Medanhodzić-Vuk, M. Sober, High perfomance liquid chromatography in pharmaceutical analyses. Bosn J. Basic Med. Sci. 4, 5–9 (2004). https://doi.org/10.17305/bjbms.2004.3405
D.B. Hibbert, Experimental design in chromatography: a tutorial review. J. Chromatogr. B (2012). https://doi.org/10.1016/j.jchromb.2012.01.020
R. Calfee, D. Piontkowski, Design and analysis of experiments. Handb Read. Res. (2016). https://doi.org/10.2307/2983009
M.A. Bezerra, R.E. Santelli, E.P. Oliveira, L.S. Villar, L.A. Escaleira, Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 76, 965–977 (2008). https://doi.org/10.1016/j.talanta.2008.05.019
A. Morshedi, M. Akbarian, Application of response surface methodology: design of experiments and optimization: a mini review. Indian J. Fundam. Appl. Life Sci. 4, 2231–6345 (2014)
B. Debrus, P. Lebrun, J.M. Kindenge, F. Lecomte, A. Ceccato, G. Caliaro, J.M.T. Mbay, B. Boulanger, R.D. Marini, E. Rozet, Innovative high-performance liquid chromatography method development for the screening of 19 antimalarial drugs based on a generic approach, using design of experiments, independent component analysis and design space. J. Chromatogr. A. 1218, 5205–5215 (2011)
B. Debrus, P. Lebrun, A. Ceccato, G. Caliaro, E. Rozet, I. Nistor, R. Oprean, F.J. Rupérez, C. Barbas, B. Boulanger, Application of new methodologies based on design of experiments, independent component analysis and design space for robust optimization in liquid chromatography. Anal. Chim. Acta. 691, 33–42 (2011)
M. Shamsipur, B. Hemmateenejad, N.J. Jahani, K.M. Majd, Liquid chromatographic–mass spectrometric monitoring of photodegradation of diphenylamine using experimental design methodology. J. Photochem. Photobiol. A Chem. 299, 210–217 (2015)
E. Landagaray, C. Vaccher, S. Yous, E. Lipka, Design of experiments for enantiomeric separation in supercritical fluid chromatography. J. Pharm. Biomed. Anal. 120, 297–305 (2016)
B. Andri, A. Dispas, R.D. Marini, P. Hubert, P. Sassiat, R. Al Bakain, D. Thiébaut, J. Vial, Combination of partial least squares regression and design of experiments to model the retention of pharmaceutical compounds in supercritical fluid chromatography. J. Chromatogr. A. 1491, 182–194 (2017)
H.C. Kraemer, D.J. Hand, C.C. Taylor, Multivariate analysis of variance and repeated measures: a practical approach for behavioural scientists. J. Am. Stat. Assoc. 84, 840 (1989). https://doi.org/10.2307/2289683
L. Vera Candioti, M.M. De Zan, M.S. Cámara, H.C. Goicoechea, Experimental design and multiple response optimization. Using the desirability function in analytical methods development. Talanta. 124, 123–138 (2014). https://doi.org/10.1016/j.talanta.2014.01.034
B. Williams, G. Mancia, W. Spiering, E.A. Rosei, M. Azizi, M. Burnier, D. Clement, A. Coca, G. De Simone, A. Dominiczak, T. Kahan, F. Mahfoud, J. Redon, L. Ruilope, A. Zanchetti, M. Kerins, S. Kjeldsen, R. Kreutz, S. Laurent, G.Y.H. Lip, R. McManus, K. Narkiewicz, F. Ruschitzka, R. Schmieder, E. Shlyakhto, K. Tsioufis, V. Aboyans, I. Desormais, Practice guidelines for the management of arterial hypertension of the European society of cardiology and the European society of hypertension ESC/ESH task force for the management of arterial hypertension. J. Hypertens. 36, 2284–2309 (2018). https://doi.org/10.1097/HJH.0000000000001961
C. Armstrong, JNC 8 guidelines for the management of hypertension in adults. Am. Fam. Physician. 90, 503–504 (2014)
P.A. James, S. Oparil, B.L. Carter, W.C. Cushman, C. Dennison-Himmelfarb, J. Handler, D.T. Lackland, M.L. LeFevre, T.D. MacKenzie, O. Ogedegbe, S.C. Smith, L.P. Svetkey, S.J. Taler, R.R. Townsend, J.T. Wright, A.S. Narva, E. Ortiz, Evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA J. Am. Med. Assoc. 311, 507–520 (2014). https://doi.org/10.1001/jama.2013.284427
United States Pharmacopeia and National Formulary (USP 43-NF 38). (United States Pharmacopeia Convention, Rockville, MD, 2019)
British Pharmacopoeia Commission. (Medicines Commission, British Pharmacopoeia, TSO, London, 2020)
D. Wile, Diuretics: a review. Ann. Clin. Biochem. 49, 419–431 (2012). https://doi.org/10.1258/acb.2011.011281
H. Fares, J.J. DiNicolantonio, J.H. O’Keefe, C.J. Lavie, Amlodipine in hypertension: a first-line agent with efficacy for improving blood pressure and patient outcomes. Open Hear (2016). https://doi.org/10.1136/openhrt-2016-000473
L. Wagner, C. Kenreigh, Amlodipine, XPharm. Compr. Pharmacol. Ref. 1–5 (2007)
F. Contreras, M.A. de la Parte, J. Cabrera, N. Ospino, Z.H. Israili, M. Velasco, Role of angiotensin II AT1 receptor blockers in the treatment of arterial hypertension. Am. J. Ther. 10, 401–408 (2003). https://doi.org/10.1097/00045391-200311000-00005
M. Volpe, L. Christian Rump, B. Ammentorp, P. Laeis, Efficacy and safety of triple antihypertensive therapy with the olmesartan/amlodipine/hydrochlorothiazide combination. Clin. Drug Investig. 32, 649–664 (2012). https://doi.org/10.2165/11636320-000000000-00000
E.D. Deeks, Olmesartan medoxomil/ amlodipine/ hydrochlorothiazide: fixed-dose combination in hypertension. Drugs 71, 209–220 (2011). https://doi.org/10.2165/11206770-000000000-00000
P. Lancellotti, Exforge HCT®: Combination of amlodipine besylate, valsartan and hydrochlorothiazide in a single tablet. Rev. Med. Liege. 65, 471–475 (2010)
K.L. Goa, A.J. Wagstaff, Losartan potassium: a review of its pharmacology, clinical efficacy and tolerability in the management of hypertension. Drugs 51, 820–845 (1996). https://doi.org/10.2165/00003495-199651050-00008
T.M. MacDonald, B. Williams, D.J. Webb, S. Morant, M. Caulfield, J.K. Cruickshank, I. Ford, P. Sever, I.S. Mackenzie, S. Padmanabhan, G.P. McCann, J. Salsbury, G. McInnes, M.J. Brown, A. Schumann, J. Helmy, C. Maniero, T.J. Burton, U. Quinn, L. Hobbs, J. Palmer, D. Collier, N. Markandu, M. Saxena, A. Zak, E. Enobakhare, J. Mackay, S.A. McG Thom, C. Coughlan, A.R. McGinnis, E. Findlay, A.G. Stanley, C. White, P. Lacy, C.J. Gardiner-Hill, S. Nazir, G.T. McInnes, S. Muir, L. McCallum, V. Melville, I.M. MacIntyre, R. Brown, R. Simon, H. Soran, S. Kwok, K. Balakrishnan, R. Hobbs, R. Iles, K.S. Myint, J. Cannon, S. Hood, K. Rutkowski, A. Webb, U. Martin, S. Kean, R. Wilson, R. Papworth, Combination therapy is superior to sequential monotherapy for the initial treatment of hypertension: a double-blind randomized controlled trial. J. Am. Heart Assoc. (2017). https://doi.org/10.1161/JAHA.117.006986
N. Dubey, A. Jain, A.K. Raghuwanshi, D.K. Jain, Simultaneous determination and validation of olmesartan medoxomil, amlodipine besilate and hydrochlorothiazide in combined tablet dosage form using RP-HPLC method. Asian J. Chem. 24, 1–3 (2012)
K.K. Kumar, C.K. Rao, G. Madhusudan, K. Mukkanti, Rapid simultaneous determination of olmesartan,—amlodipine and hydrochlorothiazide in combined pharmaceutical dosage form by stability-indicating ultra performance liquid chromatography. Am. J. Anal. Chem. 3, 50–58 (2012). https://doi.org/10.4236/ajac.2012.31008
R. El Sheikh, A.M. Annadi, A.A. Mohamed, A novel RP-HPLC method for the simultaneous assessment of olmesartan, amlodipine and hydrochlorothiazide and its application to in-vitro dissolution. Pharma. Chem. 10, 55–61 (2018)
S. Ashutosh Kumar, M. Debnath, J.V.L.N. Seshagiri Rao, D. Gowri Sankar, A new and rapid analytical method development and validation for simultaneous estimation of hydrochlorothiazide, amlodipine and olmesartan in tablet dosage form by using RP-HPLC. J. Chem. Pharm. Res. 6, 1208–1213 (2014)
B.N. Nalluri, D. Venkateswara Naik, B. Sunandana, K. Sushmitha, Development and validation of RP-HPLC-PDA method for the simultaneous estimation of hydrochlorothiazide, amlodipine besylate and olmesartan medoxomil in bulk and pharmaceutical dosage forms. J. Chem. Pharm. Res. 5, 329–335 (2013)
P. Ravisankar, J. Swathi, K.V.S.S. Kumar, P.S. Babu, Novel RP-HPLC method for simultaneous determination of olmesartan medoxomil, amlodipine besylate and hydrochlorothiazide in tablet dosage form. Int. J. Biol. Pharm. Res. 5, 927–936 (2014)
P.S. Jain, M.K. Patel, A.P. Gorle, A.J. Chaudhari, S.J. Surana, Stability-indicating method for simultaneous estimation of olmesartan medoxomile, amlodipine besylate and hydrochlorothiazide by RP-HPLC in tablet dosage form. J. Chromatogr. Sci. 50, 680–687 (2012)
J.R. Rao, M.P. Rajput, S.S. Yadav, T.S. Mulla, V.V. Bharekar, Simultaneous quantitation of olmesartan medoxomil, Amlodipine besylate and hydrochlorothiazide in pharmaceutical dosage form by using HPLC. Int. J. PharmTech Res. 3, 1435–1440 (2011)
G. Geetha, High-sensitivity simultaneous liquid chromatography/tandem mass spec-trometry (UPLC/MS/MS) assay of olmesartan medoxomil, hydrochlorothi-azide and amlodipine besylate in human plasma, Int. J. Res. Pharm. Sci. 9 (2018)
E.F. Elkady, A.A. Mandour, F.K. Algethami, A.A. Aboelwafa, F. Farouk, Sequential liquid-liquid extraction coupled to LC-MS/MS for simultaneous determination of amlodipine, olmesartan and hydrochlorothiazide in plasma samples: Application to pharmacokinetic studies. Microchem. J. 155, 104757 (2020). https://doi.org/10.1016/j.microc.2020.104757
A.R. Tengli, B.M. Gurupadayya, N. Soni, Simultaneous estimation of hydrochlorothiazide, amlodipine, and losartan in tablet dosage form by RP-HPLC. Int. J. Chem. Anal. Sci. 4, 33–38 (2013). https://doi.org/10.1016/j.ijcas.2013.03.003
S.B. Wankhede, K.C. Raka, S.B. Wadkar, S.S. Chitlange, Spectrophotometric and HPLC methods for simultaneous estimation of amlodipine besilate, losartan potassium and hydrochlorothiazide in tablets. Indian J. Pharm. Sci. 72, 136–140 (2010). https://doi.org/10.4103/0250-474X.62239
P. Haritha, B. Sreenivasa Rao, Y. Sunandamma, Stability indicating RP-HPLC method for simultaneous estimation of hydrochlorothiazide, amlodipine besylate and losartan potassium in bulk and tablet dosage form. Int. J. Chem. Sci. 14, 335–354 (2016)
K.K. Dutta, Z.A. Al-Othman, G. Mandal, I. Ali, New antihypertensive tablets formulation and HPLC analyses using new generation core shell column. Am. J. Adv. Drug Deliv. 2, 534–556 (2014)
R. Sharma, S. Khanna, G.P. Mishra, Separation of Marketed Formulation containing Hydrochlorothiazide Amlodipine and Losartan through RP-HPLC Method. Int. J. Pharm. Life Sci. 7, 4937–4941 (2016)
ICH, International conference on harmonisation of technical requirements for registration of pharmaceuticals for human use. ICH harmonised tripartite guideline, Validation Of Analytical Procedures: Text And Methodology Q2(R1), 2005
Waters, Empower PDA Software: Getting Started Guide. 145. (2002)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fouad, M.A., Elsabour, S.A., Elkady, E.F. et al. Design of experiment (DOE), multiple response optimization and utilizing the desirability function in the simultaneous HPLC separation of five antihypertensive drugs. J IRAN CHEM SOC 19, 269–282 (2022). https://doi.org/10.1007/s13738-021-02316-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-021-02316-7