Abstract
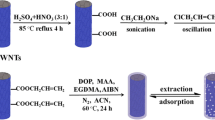
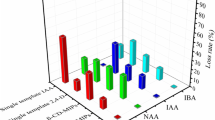
Molecularly imprinted polymers (MIPs) are polymers possessing receptor cavities with selective characteristics. The molecularly imprinted polymer of 1,8-cineole on hydroxyl-functionalized multiwall carbon nanotubes was synthesized for selective extraction of 1,8-cineole in water distillates of Artemisia sieberi (sagebrush) and thyme samples. The MIP was synthesized with cineole as template, methacrylic acid as functional monomer, ethylene glycol dimethacrylate as cross-linker and benzoyl peroxide as initiator. The main parameters affecting the synthesis of MIP and also the extraction efficiency of the analyte were optimized. The limit of detection of 1,8-cineole was as low as 0.04 μg/mL, while the dynamic linear range was found to be in the range of 0.125–100 µg/mL. The relative standard deviations (RSDs%) found for samples spiked at 1 and 70 µg/mL were 1.45–4.3%, respectively. The relative recoveries of the spiked samples at the above concentration levels were 93.8–98.2%.











Similar content being viewed by others
References
D.L.J. Opdyke, Food Chem. Toxicol. 13, 105 (1975)
F. Santos, V. Rao, Phytother. Res. 14, 240 (2000)
U.R. Juergens, U. Dethlefsen, G. Steinkamp, A. Gillissen, R. Repges, H. Vetter, Respir. Med. 97, 250 (2003)
G.F.R. Caldas, A.R. da Silva Oliveira, A.V. Araújo, S.S.L. Lafayette, G.S. Albuquerque, J. da Costa Silva-Neto, J.H. Costa-Silva, F. Ferreira, J.G.M. da Costa, A.G. Wanderley, PLoS ONE 10, e0134558 (2015)
A.R. Jassbi, R. Miri, I.T. Baldwin, Chem. Nat. Compd. 49, 1148 (2014)
S.F. van Vuuren, A.M. Viljoen, Flavour Fragr. J. 22, 540 (2007)
A.R. Jassbi, S. Zamanizadehnajari, I.T. Baldwin, J. Chem. Ecol. 36, 1398 (2010)
N.W. Davies, J. Chromatogr. A 503, 1 (1990)
N. Tabanca, B. Demirci, I. Gurbuez, F. Demirci, J. J. Becnel, D. E. Wedge, K. H. Baser, DTIC Document 2011
C. Boutekedjiret, F. Bentahar, R. Belabbes, J.M. Bessiere, Flavour Fragr. J. 18, 481 (2003)
A. Daneshfar, S. Babaee, Anal. Bioanal. Chem. Res. 3, 131 (2016)
C. Aubert, S. Baumann, H. Arguel, J. Agric. Food Chem. 53, 8881 (2005)
B.B. Prasad, K. Tiwari, M. Singh, P.S. Sharma, A.K. Patel, S. Srivastava, J. Chromatogr. A 1198–1199, 59 (2008)
M.A. Golsefidi, Z. Es’haghi, A. Sarafraz-Yazdi, J. Chromatogr. A 1229, 24 (2012)
X.Y. Gong, X.J. Cao, J. Biotechnol. 153, 8 (2011)
R. Suedee, W. Intakong, F.L. Dickert, Talanta 70, 194 (2006)
G.Z. Fang, J. Tan, X.P. Yan, Anal. Chem. 77, 1734 (2005)
A. Pardo, T. Josse, L. Mespouille, B. Blankert, P. Dubois, P. Duez, Phytochem. Anal. 28, 289 (2017)
D.M. Hawkins, D. Stevenson, S.M. Reddy, Anal. Chim. Acta 542, 61 (2005)
S. Wei, M. Jakusch, B. Mizaikoff, Anal. Chim. Acta 578, 50 (2006)
B. Sellergren, Angew. Chem. Int. Ed. 39, 1031 (2000)
N.M. Maier, W. Lindner, Anal. Bioanal. Chem. 389, 377 (2007)
B.T.S. Bui, K. Haupt, Anal. Bioanal. Chem. 398, 2481 (2010)
C. Sulitzky, B. Rückert, A.J. Hall, F. Lanza, K. Unger, B. Sellergren, Macromolecules 35, 79 (2002)
M. Behzadi, E. Noroozian, M. Mirzaei, Talanta 108, 66 (2013)
Acknowledgements
We are thankful for the financial supports of the research council of Shiraz University of Medical Science and Shahid Bahonar of Kerman University. The authors would also like to thank Mehdi Zare for his professional efforts in identifying the plants under study and Zahra Shojaeifard for her comments on the first draft of the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Ahmadi, R., Noroozian, E. & Jassbi, A.R. Molecularly imprinted polymer solid-phase extraction for the analysis of 1,8-cineole in thyme and sagebrush distillates. J IRAN CHEM SOC 17, 1153–1161 (2020). https://doi.org/10.1007/s13738-019-01840-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-019-01840-x