Abstract
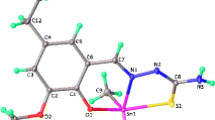
Organotin complexes have been synthesized by refluxing 2-mercapto-5-methyl benzimidazole with R2SnCl2/R3SnCl (R = Me, n-Bu, Ph) in 1:1 molar ratio in the first step. In the second step, synthesized organotin(IV) complexes were treated with CS2 and R2SnCl2/R3SnCl/PdCl2 to yield homo- and heterobimetallic complexes. The composition of the synthesized complexes, the bonding behavior of the donor groups, and structural assignments were studied by elemental analysis and different spectral techniques, including IR and multinuclear NMR (1H, 13C). The IR data shows bidentate nature of the ligand which is also confirmed by semiempirical study, while NMR data confirms the four-coordinated geometry in solution. The free ligand and its respective homo- and heterobimetallic complexes were screened in vitro against a number of microorganisms to assess their biocidal properties. The biological activity data show that complexes exhibits significant antibacterial and antifungal activities as compared to ligand with few exceptions.



Similar content being viewed by others
References
P.A. Cusack, Applied Organometallic Chemistry, 2nd ed. (Blackie Academic and Professional, London 1998)
C.S. Parulker, V.K. Jain, T. Kesvadas, E.R.T. Tiekink, J. Organomet. Chem. 163, 386 (1990)
K.C. Molloy, T.G. Percell, R. Hahn, H. Schuman, J. Zuckerman, Organometallics 5, 85 (1986)
P.J. Smith, A.J. Crow, R. Hill, Laboratory Evaluation of Tributyltin(IV) Compounds as Wood Preservatives, Publication 559 (International Tin Research Institute, London, 1979)
S. Shahzadi, S. Ali, M. Fettouhi, J. Chem. Cryst. 38, 273 (2008)
W.N. Aldridge, J.J. Zuckerman, Organotin Compounds: New Chemistry and Applications, Advanced Chemistry (American Chemical Society, Washington, 1976)
A. Mustafa, A.A. Taqa, Synth. React. Inorg. Met. -Org. Chem. 31, 517 (2001)
L. Maurizio, M. Luciano, M. Clara, A.P. Maria, Inorg. Chim. Acta 357, 367 (2004)
N.H. Buttrus, M.M. Suleman, T.A.K. Al-Allaf, Synth. React. Inorg. Met. -Org. Chem. 31, 837 (2001)
N.H. Buttrus, Synth. React. Inorg. Met. Org. Chem. 28, 1643 (1998)
S. Shahzadi, S. Ali, K. Shahid, M. Yousaf, S.K. Sharma, K. Qanungo, J. Chin. Chem. Soc. 57, 659 (2010)
S. Hussain, S. Ali, S. Shahzadi, M.A. Chaudhary, S.K. Sharma, K. Qanungo, I.H. Bukhari, J. Coord. Chem. 65, 278 (2012)
H.N. Khan, S. Ali, S. Shahzadi, M. Helliwell, Russian J. Inorg. Chem. 57, 665 (2012)
W.L.F. Armarego, C.L.L. Chai, Purification of Laboratory Chemicals (Elsevier, New York, 2003)
MOPAC2007, J.J.P. Stewart. Stewart Computational, Chemistry, Version: 7.334 W
J.J.P. Stewart, J. Mol. Model. 13, 1173 (2007)
CLSI (The Clinical Laboratory Standards Institute), Agar Dilution and Disc Diffusion Susceptibility Testing of Campylobacter spp. Clinical Microbio. 45 (2007) 2758
Q.K. Huynh, J.R. Borgmeyer, C.E. Smith, L.D. Bell, D.M. Shah, J. Bio. Chem. 316, 723 (2001)
S.D. Sarker, L. Naharb, Y. Kumarasamyc, Methods 42, 321 (2007)
F.G.D. Steel, J.H. Torrie, D.A. Dikey, Principles and procedures of a biometrical approach, 3rd edn. (W.C. MIC Graw Hill:New York, 1997)
S. Shahzadi, S. Ali, K. Wurst, M.N. Haq, Heteroatom Chem. 18, 664 (2007)
K. Singh, P. Puri, Dharampal Turk. J. Chem. 34, 499 (2010)
R. Singh, N.K. Kaushik, Mol. &Biomol. Spectro. 72, 691 (2009)
K.S. Siddiqi, R.I. Kureshy, N.H. Khan, S.A.A. Zaidi, Indian J. Chem. A 24A, 578 (1985)
E.J. Kupchi, P.J. Calabretta, Inorg. Chem. 4, 973 (1965)
A.A.Z. Zaidi, K.S. Siddiqi, N. Islam, Indian J. Chem. 12, 1197 (1974)
M. Honda, M. Komura, G. Kawaski, T. Tanaka, K. Okawara, J. Inorg. Nucl. Chem. 30, 3231 (1968)
Y. Win, S.G. Teoh, M.R. Vikneswaran, Y. Sivasothy, S. Tiong, P. Ibrahim, Aust. J. Basic Appl. Sci. 4, 5923 (2010)
P.A. Ajbade, D.C. Onwucliwe, M.J. Moloto, Inorg. Chim. Acta 362, 5031 (2011)
D.A. Brown, W.K. Glass, M.A. Burke, Spectrochim. Acta 32, 137 (1976)
H.O. Kalinowski, S. Berger, S. Braun, 13 C NMR Spektroskopio (Thieme, Stuttgart, 1984)
S. Hussain, S. Ali, S. Shahzadi, S.K. Sharma, K. Qanungo, M. Altaf, H.S. Evans, Phosphorus Sulphur Silicon Relat. Elem. 186, 542 (2011)
S. Jabbar, I. Shahzadi, R. Rehman, H. Iqbal, Qurat-ul-Ain, A. Jamil, R. Kaosar, S. Ali, S. Shahzadi, M.A. Chaudhary, Q.M. Khan, M. Shahid, S.K. Sharma, K. Qanungo, J. Coord. Chem., 65, 572 (2012)
F. Marchetti, M. Pellei, C. Pettinari, R. Pettinari, E. Rivarola, C. Santini, B.W. Skeltom, A.H. White, Appl. Organomet. Chem. 690, 1878 (2005)
J. Holecek, M. Nadvornik, K. Handlir, A. Lycka, J. Organomet. Chem. 241, 177 (1983)
M. Nadvornik, J. Holecek, K. Handlir, A. Lycka, J. Organomet. Chem. 275, 43 (1984)
A. Lycka, J. Holecek, M. Nadvornik, K. Handlir, J. Organomet. Chem. 280, 323 (1985)
K. Tahira, S. Ali, S. Shahzadi, S.K. Sharma, K. Qanungo, J. Coord. Chem. 64, 1871 (2011)
M.M. Amin, S. Ali, S. Shahzadi, S.K. Sharma, K. Qanungo, J. Coord. Chem. 64, 337 (2011)
M. Jabeen, S. Ali, S. Shahzadi, M. Shahid, Q.M. Khan, S.K. Sharma, K. Qanungo, J. Iran. Chem. Soc. 9, 307 (2012)
M.K. Samota, G. Seth, Heteroatom Chem. 21, 44 (2010)
S.S. Konstantinovic, B.C. Radovanovic, S.P. Sovilj, S. Stanojevic, J. Serb. Chem. Soc. 73, 7 (2008)
G. Huang, S. Moore, J. Jiaxin, D. Dehui, Food Sci. Tech. 11, 25 (2001)
S. Shahzadi, K. Shahid, S. Ali, M. Mazhar, K.M. Khan, J. Iran. Chem. Soc. 2, 277 (2005)
L. Dostal, R. Jambor, A. Ruzicka, R. Jirasko, J. Taraba, J. Holecek, J. Organomet. Chem. 692, 3750 (2007)
S. Shahzadi, M.H. Bhatti, K. Shahid, S. Ali, S.R. Tariq, M. Mazhar, K.M. Khan, Monatsh. Chem. 133, 1089 (2002)
J. Koshy, A. Lo, K.M. Ansary, V.G.K. Das, Met.-Based Drugs 8, 107 (2001)
S. Shahzadi, S. Ali, J. Iran. Chem. Soc. 5, 16 (2008)
Acknowledgments
Pakistan Science Foundation is highly acknowledged for support under the project No. PSF/Res/P-GCU/Chem(436). SKS and KQ thanks the Head, App Sci. and Dean FET, MITS for encouragement and support.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Rafiq, M., Ali, S., Shahzadi, S. et al. Synthesis, characterization, and biological activities of homo- and heterobimetallic complexes of Sn(IV) and Pd(II) with 2-mercapto-5-methyl benzimidazole. J IRAN CHEM SOC 11, 169–178 (2014). https://doi.org/10.1007/s13738-013-0287-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-013-0287-4