Abstract
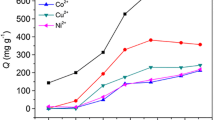
A chitosan/attapulgite/poly(acrylic acid) (CS/ATP/PAA) hydrogel was prepared by glow-discharge electrolysis plasma (GDEP) technique, in which N,N'-methylene-bis-acrylamide was acted as a crossing-linker, and then it was used as an adsorbent for the removal of Pb2+ ions from aqueous solutions. A possible polymerization mechanism is proposed. The structure, morphology and adsorption mechanism of CS/ATP/PAA hydrogel were characterized by FTIR, XRD, SEM and XPS techniques. The influences of pH, contact time and initial concentration on the Pb2+ adsorption were systematically examined. The selective adsorption of CS/ATP/PAA hydrogel for Pb2+ ions with the coexistence of Cd2+, Co2+ and Ni2+ ions was investigated, as well. In addition, regeneration of hydrogel was also discussed in detail. The results indicated that the optimal adsorption pH was 4.8, and the time of adsorption equilibrium was 60 min. The adsorption behaviors fitted well to the pseudo-second-order kinetic model and Langmuir isotherm. The maximum adsorption capacity of CS/ATP/PAA hydrogel for Pb2+ ions based on Langmuir isotherm was 531.9 mg g–1. The CS/ATP/PAA adsorbent exhibited promising selectivity for Pb2+ ions with the coexistence of Cd2+, Co2+ and Ni2+ ions, and excellent reusability using EDTA-4Na solution as desorption solution. The adsorption process of Pb2+ ions on CS/ATP/PAA hydrogel was presented by the coordination between N or O atoms and Pb2+ ion, and the ion-exchange between Na+ and Pb2+ ions.











Similar content being viewed by others
References
Ali I (2012) New generation adsorbents for water treatment. Chem Rev 112:5073–5091
Ali I, Peng CS, Lin DC, Saroj DP, Naz I, Khan ZM, Sultan M, Ali M (2019) Encapsulated green magnetic nanoparticles for the removal of toxic Pb2+ and Cd2+ from water: development, characterization and application. J Environ Manag 234:273–289
Khazaei M, Nasseri S, Ganjali MR, Khoobi M, Nabizadeh R, Gholibegloo E, Nazmara S (2018) Selective removal of lead ions from aqueous solutions using 1,8-dihydroxyanthraquinone (DHAQ) functionalized graphene oxide: isotherm, kinetic and thermodynamic studies. RSC Adv 8:5685–5694
Wang NN, Xu XJ, Li HY, Zhai JL, Yuan LZ, Zhang KX, Yu HW (2016) Preparation and application of a xanthate-modified thiourea chitosan sponge for the removal of Pb(II) from aqueous solutions. Ind Eng Chem Res 55:4960–4968
Liu YM, Ju XJ, Xin Y, Zheng WC, Wang W, Wei J, Xie R, Liu Z, Chu LY (2014) A novel smart microsphere with magnetic core and ion-recognizable shell for Pb2+ adsorption and separation. ACS Appl Mater Interfaces 6:9530–9542
Liu B, Lv X, Meng X, Yu G, Wang D (2013) Removal of Pb(II) from aqueous solution using dithiocarbamate modified chitosan beads with Pb(II) as imprinted ions. Chem Eng J 220:412–419
World Health Organisation (2011) Guidelines for drinking-water quality, 4th ed, Geneva, 383–384.
Yu J, Zheng JD, Lu QF, Yang SX, Zhang XM, Wang X, Yang W (2016) Selective adsorption and reusability behavior for Pb2+ and Cd2+ on chitosan/poly(ethylene glycol)/poly(acrylic acid) adsorbent prepared by glow-discharge electrolysis plasma. Colloid Polym Sci 294:1585–1598
Akkaya R, Ulusoy U (2008) Adsorptive features of chitosan entrapped in polyacrylamide hydrogel for Pb2+, UO2 2+, and Th4+. J Hazard Mater 151:380–388
Li Z, Xiao D, Ge Y, Koehler S (2015) Surface-functionalized porous lignin for fast and efficient lead removal from aqueous solution. ACS Appl Mater Interf 7:15000–15009
Fan QH, Li Z, Zhao HG, Jia ZH, Xu JZ, Wu WS (2009) Adsorption of Pb(II) on palygorskite from aqueous solution: effects of pH, ionic strength and temperature. Appl Clay Sci 45:111–116
Wang S, Ariyanto E (2007) Competitive adsorption of malachite green and Pb ions on natural zeolite. J Colloid Interf Science 314:25–31
Lu QF, Yu J, Gao JZ, Yang W, Li Y (2011) Glow-discharge electrolysis plasma induced synthesis of polyvinylpyrrolidone/acrylic acid hydrogel and its adsorption properties for heavy-metal ions. Plasma Process Polym 8:803–814
Yu J, Zhang HT, Li Y, Lu QF, Wang QZ, Yang W (2016) Synthesis, characterization, and property testing of PGS/P(AMPS-co-AM) superabsorbent hydrogel initiated by glow-discharge electrolysis plasma. Colloid Polym Sci 294:257–270
Yu J, Zheng JD, Lu QF, Yang SX, Wang X, Zhang XM, Yang W (2016) Reusability and selective adsorption of Pb2+ on chitosan/P(2-acryl amido-2-methyl-1-propanesulfonic acid-co-acrylic acid) hydrogel. Iran Polym J 25:1009–1019
Gao JZ, Ma DL, Lu QF, Li Y, Li XF, Yang W (2010) Synthesis and characterization of montmorillonite-graft-acrylic acid superabsorbent by using glow-discharge electrolysis plasma. Plasma Chem Plasma Process 30:873–883
Liu P, Jiang L, Zhu L, Wang A (2014) Novel approach for attapulgite/poly(acrylic acid) (ATP/PAA) nanocomposite microgels as selective adsorbent for Pb(II) ion. React Funct Polym 74:72–80
Deng Y, Gao Z, Liu B, Hu X, Wei Z, Sun C (2013) Selective removal of lead from aqueous solutions by ethylenediamine-modified attapulgite. Chem Eng J 223:91–98
Liu Y, Zheng Y, Wang AQ (2010) Enhanced adsorption of methylene blue from aqueous solution by chitosan-g-poly(acrylic acid)/vermiculite hydrogel composites. J Environ Sci-China 22:486–493
Yu J, Li Y, Lu QF, Zheng JD, Yang SX, Jin F, Wang QZ, Yang W (2016) Synthesis, characterization and adsorption of cationic dyes by CS/P(AMPS-co-AM) hydrogel initiated by glow-discharge electrolysis plasma. Iran Polym J 25:423–435
Vakili M, Deng SB, Li T, Wang W, Wang WJ, Yu G (2018) Novel crosslinked chitosan for enhanced adsorption of hexavalent chromium in acidic solution. Chem Eng J 347:782–790
Aliabadi M, Irani M, Ismaeili J, Piri H, Parnian MJ (2013) Electrospun nanofiber membrane of PEO/chitosan for the adsorption of nickel, cadmium, lead and copper ions from aqueous solution. Chem Eng J 220:237–243
Ge H, Hua T, Chen X (2016) Selective adsorption of lead on grafted and crosslinked chitosan nanoparticles prepared by using Pb2+ as template. J Hazard Mater 308:225–232
Gad YH (2008) Preparation and characterization of poly(2-acrylamido-2- methylpropane-sulfonic acid)/chitosan hydrogel using gamma irradiation and its application in wastewater treatment. Radiat Phys Chem 77:1101–1107
Nguyen NT, Liu JH (2013) Fabrication and characterization of poly(vinyl alcohol)/chitosan hydrogel thin films via UV-irradiation. Eur Polym J 49:4201–4211
Yu J, Yang GG, Pan YP, Lu QF, Yang W, Gao JZ (2014) Poly (acrylamide-co-acrylic acid) hydrogel induced by glow-discharge electrolysis plasma and its adsorption properties for cationic dyes. Plasma Sci Technol 16:767–776
Zou LZ, Shao PH, Zhang K, Yang LM, You D, Shi H, Pavlostathis SG, Lai WQ, Liang DH, Lou XB (2019) Tannic acid-based adsorbent with superior selectivity for lead(II) capture: adsorption site and selective mechanism. Chem Eng J 364:160–166
Yu J, Zhang XM, Lu QF, Sun DX, Wang X, Zhu SW, Zhang ZC, Yang W (2018) Evaluation of analytical performance for the simultaneous detection of trace Cu, Co and Ni by using liquid cathode glow discharge-atomic emission spectrometry. Spectrochim Acta B 145:64–70
Lu QF, Yang SX, Sun DX, Zheng JD, Li Y, Yu J, Su MG (2016) Direct determination of Cu by liquid cathode glow discharge-atomic emission spectrometry. Spectrochim Acta B 125:136–139
Liu Y, Sun B, Wang L, Wang D (2012) Characteristics of light emission and radicals formed by contact glow discharge electrolysis of an aqueous solution. Plasma Chem Plasma Process 32:359–368
Hsieh KC, Wang H, Locke BR (2016) Analysis of electrical discharge plasma in a gas-liquid flow reactor using optical emission spectroscopy and the formation of hydrogen peroxide. Plasma Process Polym 13:908–917
Joshi RP, Thagard SM (2013) Streamer-like electrical discharges in water: part II. Environmental applications. Plasma Chem Plasma Process 33:17–49
Brisset JL, Moussa D, Doubla A, Hnatiuc E, Hnatiuc B, Youbi GK, Herry JM, Naïtali M, Bellon-Fontaine MN (2008) Chemical reactivity of discharges and temporal post-discharges in plasma treatment of aqueous media: examples of gliding discharge treated solutions. Ind Eng Chem Res 47:5761–5781
Huang J, Liu Y, Jin Q, Wang X, Yang J (2007) Adsorption studies of a water soluble dye, Reactive Red MF-3B, using sonication-surfactant-modified attapulgite clay. J Hazard Mater 143:541–548
Fan QH, Shao DD, Hu J, Wu WS, Wang XK (2008) Comparison of Ni2+ sorption to bare and ACT-graft attapulgites: effect of pH, temperature and foreign ions. Surf Sci 602:778–785
Liu J, Wang Q, Wang A (2007) Synthesis and characterization of chitosan-g-poly(acrylic acid)/sodium humate superabsorbent. Carbohyd Polym 70:166–173
Zhao F, Repo E, Sillanpää M, Meng Y, Yin D, Tang WZ (2015) Green synthesis of magnetic EDTA- and/or DTPA-cross-linked chitosan adsorbents for highly efficient removal of metals. Ind Eng Chem Res 54:1271–1281
Ren Y, Abbood HA, He F, Peng H, Huang K (2013) Magnetic EDTA-modified chitosan/SiO2/Fe3O4 adsorbent: preparation, characterization, and application in heavy metal adsorption. Chem Eng J 226:300–311
Benamer S, Mahlous M, Tahtat D, Nacer-Khodja A, Arabi M, Lounici H, Mameri N (2011) Radiation synthesis of chitosan beads grafted with acrylic acid for metal ions sorption. Radiat Phys Chem 80:1391–1397
Vasconcelos HL, Guibal E, Laus R, Vitali L, Fávere VT (2009) Competitive adsorption of Cu(II) and Cd(II) ions on spray-dried chitosan loaded with Reactive Orange 16. Mater Sci Eng C 29:613–618
Manasi Rajesh V, Rajesh N (2015) An indigenous Halomonas BVR1 strain immobilized in crosslinked chitosan for adsorption of lead and cadmium. Int J Biol Macromol 79:300–308
Yan H, Dai J, Yang Z, Yang H, Cheng R (2011) Enhanced and selective adsorption of copper(II) ions on surface carboxymethylated chitosan hydrogel beads. Chem Eng J 174:586–594
Lu Y, He J, Luo G (2013) An improved synthesis of chitosan bead for Pb(II) adsorption. Chem Eng J 226:271–278
Chauhan D, Sankararamakrishnan N (2008) Highly enhanced adsorption for decontamination of lead ions from battery wastewaters using chitosan functionalized with xanthate. Bioresour Technol 99:9021–9024
Laus R, Costa TG, Szpoganicz B, Fávere VT (2010) Adsorption and desorption of Cu(II), Cd(II) and Pb(II) ions using chitosan crosslinked with epichlorohydrin-triphosphate as the adsorbent. J Hazard Mater 183:233–241
Lyu FY, Yu HQ, Hou TL, Yan LG, Zhang XH, Du B (2019) Efficient and fast removal of Pb2+ and Cd2+ from an aqueous solution using a chitosan/Mg-Al-layered double hydroxide nanocomposite. J Colloid Interf Sci 539:184–193
Zhou D, Zhang L, Guo S (2005) Mechanisms of lead biosorption on cellulose/chitin beads. Water Res 39:3755–3762
Chen M, Shafer-Peltier K, Randtke SJ, Peltier E (2018) Competitive association of cations with poly(sodium 4-styrenesulfonate) (PSS) and heavy metal removal from water by PSS-assisted ultrafiltration. Chem Eng J 344:155–164
Wan Ngah WS, Fatinathan S (2010) Pb(II) biosorption using chitosan and chitosan derivatives beads: equilibrium, ion exchange and mechanism studies. J Environ Sci 22:338–346
Cai H, Sun Y, Zhang X, Zhang L, Liu H, Li Q, Bo TZ, Zhou DZ, Wang C, Lian J (2019) Reduction temperature-dependent nanoscale morphological transformation and electrical conductivity of silicate glass microchannel plate. Materials 12:1183
Acknowledgements
This work was supported in part by Natural Science Foundation of Gansu Province (17JR5RA075 and 17JR5RA077), and National Natural Science Foundation of China (Nos. 21567025, 21864022 and 21367023), China.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yu, J., Lu, Q., Zheng, J. et al. Chitosan/attapulgite/poly(acrylic acid) hydrogel prepared by glow-discharge electrolysis plasma as a reusable adsorbent for selective removal of Pb2+ ions. Iran Polym J 28, 881–893 (2019). https://doi.org/10.1007/s13726-019-00751-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13726-019-00751-1