Abstract
Female adnexal tumors of probable Wolffian origin (FATWOs) are rare neoplasms that originate from remnants of the Wolffian duct. These tumors often appear in the broad ligament and para-ovarian area where such remnants exist. FATWOs have specific pathological characteristics similar to those of remnants of the Wolffian duct. Since these tumors neither have any specific tumor markers nor specific features on scanning images, there has been no case report concerning the preoperative diagnosis of FATWOs. Diagnostic imaging technology has become remarkably developed nowadays, enabling physicians to make preoperative diagnosis more precisely. We report here a case of FATWO with detailed discussion of its MR images.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Female adnexal tumors of probable Wolffian origin (FATWOs) were first reported by Kariminejad and Scully [1] in 1973, documenting nine cases of distinctive female adnexal tumors arising in the leaves of the broad ligament or hung on pedicles from it. These tumors frequently arose where Wolffian remnants were, and they had pathological features compatible with their development from such remnants. Although approximately 85 cases of FATWOs, including 11 malignant cases [2, 3], have been reported as of today, most of them discuss pathologic features of these tumors. It is usually hard, before an operation, to confirm the diagnosis of FATWOs because they have no specific tumor markers or features on scanning images. Imaging technology has become remarkably developed nowadays, and one case has been reported regarding MRI features of this type of tumor. Thus, Matsuki et al. [4] reported that FATWOs appear with slightly high intensity on T2-weighted imaging and isointensity on T1-weighted imaging. We report here a case of FATWO focusing on MRI findings specific to this type of tumor.
Case report
A 36-year-old Japanese woman, gravida 2 parity 0, was referred to our hospital with suspicion of ovarian tumor. She did not have any significant past gynecologic history. Pelvic examination revealed a hen-egg-size mass in the right adnexal region. The biochemical and hematological tests showed no abnormality. Serum tumor markers were CA19-9, 19 U/ml; CA-125, 12 U/ml; CEA, 5 ng/ml; SCC, 0.7 ng/ml; and AFP, 5 ng/ml. Pelvic sonography revealed a 60-mm hypoechoic solid mass with small cysts behind the normal uterus. Cytology of the uterine cervix and endometrium were all negative for malignancy.
Imaging findings
A well-defined ovoid mass, measuring approximately 60 mm in diameter, was detected at the right adnexal region on MRI. T2-weighted axial imaging (repetition time (TR)/echo time (TE) = 4000/103 ms) showed the mass was slightly higher in intensity compared to skeletal muscle and had a central high intensity area (Fig. 1). On T1-weighted axial imaging (TR/TE = 540/7.8 ms), the tumor was isointense with skeletal muscle and did not exhibit a high intensity region, suggesting the presence of a hemorrhagic lesion or fat tissue (Fig. 2). The solid portion of the mass was enhanced homogeneously with intravenous gadolinium–DTPA and showed high intensity (b = 800 s/mm2) on diffusion-weighted imaging (DWI) with restricted diffusion on the apparent diffusion coefficient (ADC) map (Fig. 3). On the basis of the MR findings, the tumor was thought to have high cellularity accompanied by a central degeneration or necrosis. On computed tomography (CT), the mass contained curvilinear and punctate calcifications. Adjacent to the tumor, normal right ovary was detected, indicating that the mass had probably originated from the fallopian tube or broad ligament (Fig. 4). However, an ovarian tumor preserving normal ovarian stroma could not be excluded. No abnormality was found in left adnexa or uterus. A small amount of ascites was noted in the Douglas pouch but it was within normal limits, and no apparent pelvic lymphadenopathy could be detected. Preoperative differential diagnoses were ovarian solid tumors such as granulosa cell tumor, thecofibroma, serous carcinoma, lymphoma, and metastatic tumors. The extra-ovarian tumors, including fallopian tube cancer, uterine leiomyoma, and broad ligament, were also considered.
T1- and T2-weighted axial imaging (T1WI and T2WI). A well-defined ovoid mass was demonstrated at the right adnexal region on MRI. T2-weighted axial imaging (TR/TE = 4000/103 ms) showed the mass was slightly higher in intensity compared to skeletal muscle. On T1-weighted axial imaging (TR/TE = 540/7.8 ms), the tumor was isointense with skeletal muscle and did not exhibit a high intensity region. FST1WI fat-suppressed T1-weighted axial image
Operation
An approximately 60-mm tumor mass was found in the right broad ligament. Part of the tumor was connected to the right fallopian tube. No metastatic or disseminated tumor was observed. The uterus and bilateral oviducts and ovaries appeared to be normal. A small amount of brown-colored ascites was found in the pelvic cavity. Right salpingo-oophorectomy including the tumor mass was performed. On an intraoperative frozen section, Wolffian tumor was suspected.
Pathologic findings
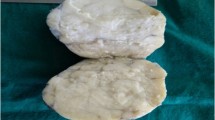
On gross section, the tumor was measured approximately 50 × 60 × 65 mm. Cut surface of the tumor revealed a white- or yellow-colored solid mass with sporadic small cysts (Fig. 5). The tumor was partially connected to the right oviduct. Microscopically, the tumor was well demarcated. Tumor cells were arranged in tubular and solid fashions with a slit or fern-leaf like pattern (Fig. 6). The lumen were surrounded by a single layer of cells. The nuclei of these cells were round with fine chromatins. The cells were round, irregular, or spindle-shaped with indistinct cytoplasm. The coffee-bean-like nuclear grooves seen in a granulosa cell tumor or sex-cord tumor were not observed, although the patient’s age did not exclude these two possibilities. The tumor cells were positive for CAM 5.2, vimentin, calretinin, and CD10. They were negative for EMA, ER, PgR, and D2-40. Scattered small foci of calcification due to tissue degeneration was noted. The overall morphology and the immunohistochemical profile were consistent with FATWO. Mitotic cells were around one to two per 10 high power fields, and no atypical figures were identified. There was no evidence of vascular invasion. The surface of the tumor was not broken down and the disease was consistent with stage 1a.
Discussion
FATWOs are usually discovered incidentally during pelvic examination or at laparotomy. The belief that they are of mesonephric origin is based on (a) the location of the tumor mass detected at the same site as mesonephric remnants, (b) their morphological dissimilarity to other ovarian tumors of either epithelial-stromal or sex cord-stromal types, and (c) some ultrastructural [5, 6] and immunohistochemical [7–9] homology with the mesonephric duct. Their recognition and diagnosis depend largely on anatomical site and varied morphology. Because MRI depicts excellent tissue contrast reflecting pathology and it is valuable for evaluating the female pelvis, this study was undertaken to apply an extensive range of MRI analytical technology to these tumors to find (a) additional support for a mesonephric origin for these lesions and (b) whether imaging features could be established for diagnostic purposes.
The tumor described here first presented in the right adnexal area, and the MRI revealed an unusual solid tumor. MR findings of FATWO have rarely been discussed as of yet. According to Matsuki et al. [4], MRI showed a well-defined ovoid mass in the adnexa. The T2-weighted images showed a slightly hyperintense mass with cystic degeneration which is hardly differentiated from a subserosal leiomyoma or ovarian tumor such as thecoma. On gadolinium–DTPA-enhanced T1-weighted images, the mass is enhanced homogeneously apart from the cystic element. All these findings are consistent with our MR images. The gross appearance as well as the microscopic findings of the surgical specimen was mostly the same as ours. In addition to Matsuki’s report, we made further findings on DWI. The solid portion of the FATWO was enhanced homogenously with intravenous gadolinium–DTPA, and it showed high intensity on DWI with restricted diffusion on ADC map (Fig. 3). DWI is a functional technique that develops image contrast through the inhibitory effect of cell membranes on the mobility of water molecules in tissues. As a result of their dense cellularity, malignant lesions have restricted diffusion, which is reflected in their low mean ADCs, and ADC has been used to characterize tumors and quantify morphologies.
Neither Matuski’s nor our case shows a prominent mitotic activity, suggesting the absence of malignancy, although this could not be predicted on DWI and ADC maps. The above MRI findings might be one of the common features of FATWOs. However, the findings proposed the differential diagnoses of ovarian solid tumors such as granulosa cell tumor, Sertoli-Leydig cell tumor, thecofibroma, and dysgerminoma. The immunohistochemical profile is important for distinguishing these tumors, and FATWO usually demonstrates positive staining for vimentin and negative for ER and EMA [9–11]. The more recent advanced histogram analysis of ADCs can interrogate the biologic heterogeneity of tumors by classifying domains of different diffusivity, which may have prognostic and predictive implications [12, 13]. When cellular biochemical characteristics detected by immunohistochemical profile can be distinguished by the recent advanced MRI technologies, they could be of clinical use for the prediction of this rare tumor.
References
Kariminejad MH, Scully RE (1973) Female adnexal tumor of probable Wolffian origin. A distinctive pathologic entity. Cancer 31:671–677
Steed H, Oza A, Chapman WB et al (2004) Female adnexal tumor of probable Wolffian origin: a clinicopathological case report and a possible new treatment. Int J Gynecol Pathol 14:546–550
Harada O, Ota H, Takagi K et al (2006) Female adnexal tumour of probable Wolffian origin: morphological, immunohistochemical, and ultrastructural study with c-kit gene analysis. Pathol Int 56:95–100
Matsuki M, Kaji Y, Matsuo M (1993) Female adnexal tumor of probable Wolffian origin, MR findings. Br J Radiol 72:911–913
Taxy JB, Battifora H (1976) Female adnexal tumor of probable Wolffian origin: evidence for low grade malignancy. Cancer 37:2349–2354
Demopoulos RI, Sitelman A, Flotte T et al (1980) Ultrastructural study of a female adnexal tumour of probable Wolffian origin. Cancer 46:2273–2280
Tavassoli F, Andrade R, Merino M (1990) Retiform Wolffian adenoma. Prog Surg Pathol 11:121–136
Rahilly MA, Williams AR, Krausz T et al (1995) Female adnexal tumour of probable Wolffian origin: a clinicopathological and immunohistochemical study of three cases. Histopathol 26:69–74
Devouassoux-Shisheboran M, Silver SA, Tavassoli FA (1999) Wolffian adnexal tumor, so-called female adnexal tumor of probable Wolffian origin (FATWO): immunohistochemical evidence in support of a Wolffian origin. Hum Pathol 30:856–863
Deen S, Duncan TJ, Hammond RH (2007) Malignant female adnexal tumors of probable Wolffian origin. Int J Gynecol Pathol 26:383–386
Sheyn I, Mira JL, Bejarano PA et al (2000) Metastatic female adnexal tumor of probable Wolffian origin. A case report and review of the literature. Arch Pathol Lab Med 124:431–434
Barajas RF Jr, Rubenstein JL, Chang JS et al (2010) Diffusion-weighted MR imaging derived apparent diffusion coefficient is predictive of clinical outcome in primary central nervous system lymphoma. AJNR Am J Neuroradiol 31:60–66
Pope WB, Kim HJ, Huo J et al (2009) Recurrent glioblastoma multiforme: ADC histogram analysis predicts response to bevacizumab treatment. Radiology 252:182–189
Conflict of interest
The authors state that there are no known conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Sato, T., Isonishi, S., Sasaki, K. et al. A case of female adnexal tumor of probable Wolffian origin: significance of MRI findings. Int Canc Conf J 1, 108–112 (2012). https://doi.org/10.1007/s13691-012-0021-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13691-012-0021-6