Abstract
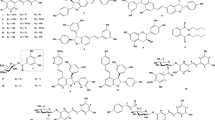
Three new acylated flavonoid C-glycosides, 6‴-(-)-phaseoylspinosin (1), 6‴-(3″″,4″″,5″″-trimethoxyl)-(E)-cinnamoylspinosin (2), and 6‴-(4″″-O-β-D-gluco-pyranosyl)-benzoylspinosin (3), were isolated from the seeds of Ziziphus mauritiana (Rhamnaceae). A further 19 known compounds including eight spinosin analogues (4–11) were also isolated. Their structures were elucidated by means of spectroscopic analysis and chemical method. Among spinosin derivatives 1, 2, 4, 7, 8, and triterpenoid saponin 14, jujuboside A (14) displayed moderate acetylcholinesterase (AchE) inhibitory activity with an inhibition value of 46.2% at a concentration of 1 µM.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ji, C. J.; Zeng, G. Z.; Han, J.; He, W. J.; Zhang, Y. M.; Tan, N. H. Bioorg. Med. Chem. Lett.2012, 22, 6377–6380.
Chen, Y. L.; Zhou, B. K. In Chinese Flora; Science Press: Beijing, 1982; Vol. 48, pp 131–146
Liu, M. J. Acta Hortic. Sin.1999, 26, 302–308.
Cheng, G.; Bai, Y. J.; Zhao, Y, Y.; Tao, J.; Liu, Y.; Tu, G. Z.; Ma, L. B.; Liao, N.; Xu, X. J. Tetrahedron2000, 56, 8915–8920.
Zhang, L.; Xu, Z. L; Wu, C. F.; Yang, J. Y.; Kano, Y.; Yuan, D. J. Asian. Nat. Prod. Res.2012, 14, 121–128.
Xie, Y. Y.; Xu, Z. L.; Wang, H.; Kano, Y.; Yuan, D. J. Asian. Nat. Prod. Res.2011, 13, 1151–1157.
Tan, N. H.; Zhou, J. Chem. Rev.2006, 106, 840–895.
Tschesche, R.; Wilhelm, H.; Fehlhaber, H. W. Tetrahedron Lett.1972, 26, 2609–2612.
Tschesche, R.; Wilhelm, H.; Kauβmann, E. U.; Eckhardt, G. Justus Liebigs Ann. Chem.1974, 1694–1701.
Tschesche, R.; Hillebrand, D.; Wilhelm, H.; Ammermann, E.; Eckhardt, G. Phytochemistry1977, 16, 1025–1028.
Jossang, A.; Zahir, A.; Diakite, D. Phytochemistry1996, 42, 565–567.
Panseeta, P.; Lomchoey, K.; Prabpai, S.; Kongsaeree, P.; Sukasamrarn, A.; Ruchiraeat, S.; Suksamrarn, S. Phytochemistry2011, 72, 909–915.
Srivastava, S. K.; Srivastava, S. D. Phytochemistry1979, 18, 1758–1759.
Agarwal, S. K.; Singh, S. S.; Verma, S.; Kumar, S. Indian J. Chem. B: Org. Chem. Incl. Med. Chem.2000, 39, 872–874.
Krasutsky, P. A. Nat. Prod. Rep.2006, 23, 919–942.
Lee, S. S.; Chen, W. C.; Huang, C. F.; Su, Y. J. Nat. Prod.1998, 61, 1343–1347.
Lv, T.; Xu, M.; Wang, D.; Zhu, H. T.; Yang, C. R.; Zhang, T. T.; Zhang, Y. J. Nat. Prod. Bioprospect.2012, 2, 217–221.
Xu, M.; Wang, D.; Zhang, Y. J; Yang, C. R. J. Nat. Prod.2007, 70, 880–883.
Xu, M.; Zhang, M.; Zhang, Y. J.; Yang, C. R. Helv. Chim. Acta.2009, 92, 321–327.
Tanaka, Y.; Sanada, S. Shoyakugaku Zasshi.1991, 45, 148–152.
Wu, Y.; He, F.; Pan, Q.; Shi, Y.; Min, Z. D.; Liang, J. Y. Chem. Nat. Compd.2011, 47, 369–372.
Xie, C.; Veitch, N. C.; Houghton, P. J.; Simmonds, M. S. J. Chem. Pharm. Bull.2003, 51, 1204–1207.
Kitajima, J.; Kimizuka, K.; Arai, M.; Tanaka, Y. Chem. Pharm. Bull.1998, 46, 1647–1649.
Renault, J. H.; Ghedira, K.; Thepenier, P.; Lavaud, C.; Zeches-Hanrot, M.; Men-Olivier, L. L. Phytochemistry1997, 44, 1321–1327.
Schumacher, B.; Scholle, S.; Holzl, J.; Khudeir, N.; Hess, S.; Muller, C. E. J. Nat. Prod.2002, 65, 1479–1485.
Deyama, T.; Ikawa, T.; Kitagawa, S.; Nishibe, S. Chem. Pharm. Bull.1987, 35, 1785–1789.
Ho, J. C.; Chen, C. M.; Row, L. C. J. Chin. Chem. Soc.2003, 50, 1271–1274.
Wang, C. Z.; Jia, Z. J. Phytochemistry1997, 45, 159–166.
Masataka, S.; Masao, K. Heterocycles1993, 36, 117–121.
Han, B. H.; Park, M. H.; Han, Y. N. Arch. Pharm. Res.1989, 12, 263–268.
Aldrich. Library of 13C and 1H FT NMR Spectra.1992, 3, 143.
Hirai, N.; Kondo, S.; Ohigashi, H. Biosci. Biotechnol. Biochem.2003, 67, 2408–2415.
Kitahara, T.; Touhara, K.; Watanabe, H.; Mori, K. Tetrahedron1989, 45, 6387–6400.
Payo-Hill, A. L.; Domingueza, R. S.; Suareza, M. O.; Batista-Baeza, M.; Castro Velez, H. T.; Rastrelli, L.; Aquinoc, R. Phytochemistry2000, 54, 927–932.
Ellman, G. L.; Courtney, K. D.; Andres, V. J.; Featherstone, R. M. Biochem Pharmacol.1961, 7, 88–95.
Author information
Authors and Affiliations
Corresponding authors
Additional information
This article is published with open access at Springerlink.com
Electronic supplementary material
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Wang, B., Zhu, HT., Wang, D. et al. New spinosin derivatives from the seeds of Ziziphus mauritiana. Nat. Prod. Bioprospect. 3, 93–98 (2013). https://doi.org/10.1007/s13659-013-0028-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13659-013-0028-5