Abstract
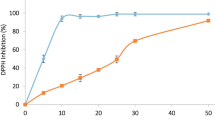
This study was conducted to investigate the antioxidant, antibacterial, anthelmintic, and cytotoxic activities of the crude methanol extract of Bonamia semidigyna (Roxb.) leaves and its derived fractions in different experimental models. Leaves of B. semidigyna were extracted with methanol (MBS) and the resultant successively fractionated into n-hexane (NBS), petroleum ether (PBS), chloroform (CBS), and ethyl acetate (EBS) soluble fractions. Later, the plant extractives were investigated for antioxidant, antibacterial, anthelmintic and cytotoxic properties. Phytochemical study was carried out using standard methods and polyphenols content were determined by spectrophotometric techniques. Antioxidant activity was evaluated by DPPH (1,1-diphenyl-2-picrylhydrazyl) radical scavenging activity, hydrogen peroxide (H2O2) scavenging activity, ferric reducing power, and total antioxidant capacity assays. Disc diffusion technique used to evaluate the antibacterial efficacy and anthelmintic activity was determined using aquarium worm (Tubifex tubifex) whereas cytotoxicity was evaluated using brine shrimp lethality bioassay. The phytochemical study revealed the presence of alkaloids, flavonoids, terpenoids, tannins, saponins, phenolic, steroids, sterols, quinones, oxalate, coumarins, and emodins in the plant extractives. In addition, the quantitative phytochemical analysis showed the significant amount of polyphenol contents. In antioxidant assays, the crude methanol extract and EBS fraction exhibited the most potent antioxidant activity among all the extractives. The crude methanol extract and EBS fraction also possessed significant antibacterial and anthelmintic potentials while moderate cytotoxic activities were found for all the plant extractives. The present study confirmed that the crude methanol extract of B. semidigyna and its EBS fraction, possibly due to its high polyphenol contents possess significant antioxidant, antibacterial, and anthelmintic properties.






Similar content being viewed by others
Abbreviations
- MBS:
-
Methanol extract of B. semidigyna
- NBS:
-
n-Hexane fraction of B. semidigyna
- PBS:
-
Petroleum ether fraction of B. semidigyna
- CBS:
-
Chloroform fraction of B. semidigyna
- EBS:
-
Ethyl acetate fraction of B. semidigyna
- BHT:
-
Butylated hydroxytoluene
- BHA:
-
Butylated hydroxyanisole
- nm:
-
Nanometer
- mm:
-
Millimeter
- DNA:
-
Deoxyribonucleic acid
- ANOVA:
-
Analysis of variance
- rpm:
-
Rotate per minute
References
Ahmad I, Mehmood Z, Mohammad F (1998) Screening of some Indian medicinal plants for their antimicrobial properties. J Ethnopharmacol 62:183–193
Ahmed D, Fatima K, Saeed R (2014) Analysis of phenolic and flavonoid contents, and the anti-oxidative potential and lipid peroxidation inhibitory activity of methanolic extract of Carissa opaca roots and its fractions in different solvents. Antioxidants 3:671–683
Aiyegoro OA, Okoh AI (2010) Preliminary phytochemical screening and in vitro antioxidant activities of the aqueous extract of Helichrysum longifolium DC. BMC Complement Altern Med 10:1–8
Ajaiyeoba EO, Onocha PA, Olarenwaju OT (2001) In vitro anthelmintic properties of Buchholzia coriaceae and Gynandropsis gynandra extracts. Pharm Biol 39:217–220
Akter KN, Karmakar P, Das A, Anonna SN (2014) Evaluation of antibacterial and anthelmintic activities with total phenolic contents of Piper betel leaves. Avicenna J Phytomed 4:320–329
Alam MN, Bristi NJ, Rafiquzzaman M (2013) Review on in vivo and in vitro methods evaluation of antioxidant activity. Saudi Pharm J 21:143–152. https://doi.org/10.1016/j.jsps.2012.05.002
Bangladesh Ethnobotany Online Database. http://www.ebbd.info/bonamia-semidigyna.html. Accessed 27 May 2018
Bate-Smith EC (1962) The phenolic constituents of plants and their taxonomic significance. I. Dicotyledons. Bot J Linn Soc 58:95–173
Bauer AW, Kirby WM, Sherris JC, Turck M (1966) Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol 45:493
Billah MM, Islam R, Khatun H et al (2013) Antibacterial, antidiarrhoeal, and cytotoxic activities of methanol extract and its fractions of Caesalpinia bonducella (L.) Roxb leaves. BMC Complement Altern Med 13:101
Braca A, De Tommasi N, Di Bari L et al (2001) Antioxidant principles from bauhinia tarapotensis. J Nat Prod 64:892–895
Chamayarit K, Staples G, Santisuk T, Larsen K (2010) Flora of Thailand, 1st edn. Royal Forest Department, Bangkok, pp 330–468
Cowan MM (1999) Plant products as antimicrobial agents. Clin Microbiol Rev 12:564–582
Das SS, Dey M, Ghosh AK (2011) Determination of anthelmintic activity of the leaf and bark extract of Tamarindus indica Linn. Indian J Pharm Sci 73:104
de Moraes J, Carvalho AAL, Nakano E et al (2013) Anthelmintic activity of carvacryl acetate against Schistosoma mansoni. Parasitol Res 112:603–610
Eich E (2008) Solanaceae and Convolvulaceae: secondary metabolites: biosynthesis, chemotaxonomy, biological and economic significance (a handbook). Springer, Berlin
Epand RF, Savage PB, Epand RM (2007) Bacterial lipid composition and the antimicrobial efficacy of cationic steroid compounds (Ceragenins). Biochim Biophys Acta Biomembr 1768:2500–2509
Giarman NJ (1955) The quantitative analysis of drugs. Yale J Biol Med 28:489–490
Gülçin I, Küfrevioǧlu Öİ, Oktay M, Büyükokuroǧlu ME (2004) Antioxidant, antimicrobial, antiulcer and analgesic activities of nettle (Urtica dioica L.). J Ethnopharmacol 90:205–215
Harborne AJ (1998) Phytochemical methods a guide to modern techniques of plant analysis, 3rd edn. Springer, Dordrecht
Hena Khanam S (2014) Bioactive Benzofuran derivatives: a review. Eur J Med Chem 30:1e22
Hossain MM, Ahamed SK, Dewan SMR et al (2014) In vivo antipyretic, antiemetic, in vitro membrane stabilization, antimicrobial, and cytotoxic activities of different extracts from Spilanthes paniculata leaves. Biol Res 47:1–9. https://doi.org/10.1186/0717-6287-47-45
Khan RA, Khan MR, Sahreen S, Ahmed M (2012) Assessment of flavonoids contents and in vitro antioxidant activity of Launaea procumbens. Chem Cent J 6:43
Krishnaiah D, Sarbatly R, Nithyanandam R (2011) A review of the antioxidant potential of medicinal plant species. Food Bioprod Process 89:217–233
Lobo V, Patil A, Phatak A, Chandra N (2010) Free radicals, antioxidants and functional foods: impact on human health. Pharmacogn Rev 4:118
Mazid MA, Datta BK, Nahar L, Sarker SD (2008) Assessment of anti-bacterial activity and brine shrimp toxicity of two Polygonum species. Ars Pharm 49:127–134
Mbaebie BO, Edeoga HO, Afolayan AJ (2012) Phytochemical analysis and antioxidants activities of aqueous stem bark extract of Schotia latifolia Jacq. Asian Pac J Trop Biomed 2:118–124. https://doi.org/10.1016/S2221-1691(11)60204-9
Meyer BN, Ferrigni NR, Putnam JE et al (1982) Brine shrimp: a convenient general bioassay for active plant constituents. Planta Med 45:31–34
Niezen JH, Waghorn TS, Charleston WAG, Waghorn GC (1995) Growth and gastrointestinal nematode parasitism in lambs grazing either lucerne (Medicago sativa) or sulla (Hedysarum coronarium) which contains condensed tannins. J Agric Sci 125:281–289
Nigri GR, Kossodo S, Waterman P et al (2004) Free radical attenuation prevents thrombosis and enables photochemical inhibition of vein graft intimal hyperplasia. J Vasc Surg 39:843–849
Okeke MI, Iroegbu CU, Eze EN et al (2001) Evaluation of extracts of the root of Landolphia owerrience for antibacterial activity. J Ethnopharmacol 78:119–127
Olliaro P, Seiler J, Kuesel A et al (2011) Potential drug development candidates for human soil-transmitted helminthiases. PLoS Negl Trop Dis 5:e1138
Oyaizu M (1986) Studies on products of browning reaction. Jpn J Nutr Diet 44:307–315
Oyedemi SO, Bradley G, Afolayan AJ (2010) In-vitro and -vivo antioxidant activities of aqueous extract of Strychnos henningsii Gilg. Afr J Pharm Pharmacol 4:70–78
Özçelik B, Kartal M, Orhan I (2011) Cytotoxicity, antiviral and antimicrobial activities of alkaloids, flavonoids, and phenolic acids. Pharm Biol 49:396–402
Pal DK, Sahoo M, Mishra AK (2007) Anthelminthic activity of stems of Opuntia vulgaris mill. Asian J Chem 19:793
Saeed N, Khan MR, Shabbir M (2012) Antioxidant activity, total phenolic and total flavonoid contents of whole plant extracts Torilis leptophylla L. BMC Complement Altern Med 12:1174. https://doi.org/10.1186/1472-6882-12-221
Sahu MC, Padhy RN (2013) In vitro antibacterial potency of Butea monosperma Lam. against 12 clinically isolated multidrug resistant bacteria. Asian Pac J Trop Dis 3:217–226
Saleh-e-In MM, Sultana N, Hossain MN et al (2016) Pharmacological effects of the phytochemicals of Anethum sowa L. root extracts. BMC Complement Altern Med 16:464. https://doi.org/10.1186/s12906-016-1438-9
Schetter AJ, Heegaard NHH, Harris CC (2009) Inflammation and cancer: interweaving microRNA, free radical, cytokine and p53 pathways. Carcinogenesis 31:37–49
Sharififar F, Dehghn-Nudeh G, Mirtajaldini M (2009) Major flavonoids with antioxidant activity from Teucrium polium L. Food Chem 112:885–888
Solis PN, Wright CW, Anderson MM et al (1993) A microwell cytotoxicity assay using Artemia salina (brine shrimp). Planta Med 59:250–252
Sreejith M, Kannappan N, Santhiagu A, Mathew AP (2013) Phytochemical, anti-oxidant and Anthelmintic activities of various leaf extracts of Flacourtia sepiaria Roxb. Asian Pac J Trop Biomed 3:947–953
Sun J-S, Tsuang Y-H, Chen I-J et al (1998) An ultra-weak chemiluminescence study on oxidative stress in rabbits following acute thermal injury. Burns 24:225–231
Tiwari P, Kumar B, Kaur M et al (2011) Phytochemical screening and extraction: a review. Int Pharm Sci 1:98–106
VanWagenen BC, Larsen R, Cardellina JH et al (1993) Ulosantoin, a potent Insecticide from the sponge Ulosa ruetzleri. J Org Chem 58:335–337. https://doi.org/10.1021/jo00054a013
Von Gadow A, Joubert E, Hansmann CF (1997) Comparison of the antioxidant activity of rooibos tea (Aspalathus linearis) with green, oolong and black tea. Food Chem 60:73–77
Yang C-H, Cheng M-J, Chiang MY et al (2008) Dihydrobenzo [c] phenanthridine alkaloids from stem bark of Zanthoxylum nitidum. J Nat Prod 71:669–673
Acknowledgements
The authors are grateful to the Department of Pharmacy, International Islamic University Chittagong, Bangladesh for providing all the laboratory facilities and support to complete this research work.
Author information
Authors and Affiliations
Contributions
Md. Adnan and Md. Nazim Uddin Chy conceived and designed the experiments, carried out all the experimental works, and collected data. Md. Nazim Uddin Chy has furnished the designing of the work, analyzed and interpreted experimental results, and drafted the final manuscript. Sajib Rudra, Afroza Tahamina, and Raju Das performed in vitro experiments (DPPH, hydrogen peroxide and reducing power assays) and contributed to data collection. Mohammad Akramul Hoque Tanim, Tanvir Iqram Siddique and Asadul Hoque did the phytochemical study and participated in the total phenols and flavonoids content determination. Syeda Mashnia Tasnim and Arkajyoti Paul participated in the antibacterial assay and helped to write the relevant portion. A.T.M. Mostafa Kamal supervised the study, evaluated the data and reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical statement
N\A.
Conflict of interest
This manuscript described has not been published before; not under consideration for publication anywhere else; and has been approved by all co-authors.
Rights and permissions
About this article
Cite this article
Adnan, M., Chy, M.N.U., Rudra, S. et al. Evaluation of Bonamia semidigyna (Roxb.) for antioxidant, antibacterial, anthelmintic and cytotoxic properties with the involvement of polyphenols. Orient Pharm Exp Med 19, 187–199 (2019). https://doi.org/10.1007/s13596-018-0334-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13596-018-0334-x