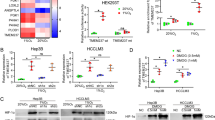
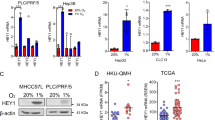
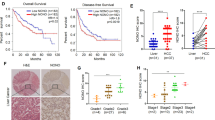
Abstract
Purpose
Hepatocellular carcinoma (HCC) has emerged as a leading cause of cancer-related deaths globally, in which hypoxia and activated hypoxia-inducible factors (HIFs) play important roles. The sibling rivalry between HIF-1α and HIF-2α in hypoxic tumor growth and progression still remains to be resolved, including in HCC. In this study, we aimed to analyze the mechanism by which HIF-1α and HIF-2α balance the proliferative response of HCC cells to hypoxia.
Methods
The expression of HIF-1α, HIF-2α, c-MYC, Rictor and Raptor in corresponding tumor and non-tumor tissues from twenty-six patients with HCC was analyzed. The relationships between HIF-1α and HIF-2α and their respective effects were evaluated further in vitro in hypoxic HCC cells using co-immunoprecipitation, chromatin immunoprecipitation, in situ proximity ligation, annexin V-FITC/PI staining apoptosis and MTT assay. In addition, short hairpin RNA (shRNA) transfections targeting HIF-1α/2α and Rictor and Western blotting were applied in HCC cells to study the underlying mechanism.
Results
We found that HIF-2α expression showed a positive correlation with c-MYC expression in tumor tissues, whereas HIF-1α did not. In vitro, increased HCC cell proliferation and an increased interaction between HIF-2α and c-MYC were observed under mild chronic hypoxic conditions. Although mild hypoxia led to HIF-1α, HIF-2α and c-MYC up-regulation, we found that mTORC2-regulated HIF-2α competed with HIF-1α to bind to c-MYC. Moreover, we found that HIF-2α knockdown decreased the expression of downstream c-MYC, suppressed hypoxic cell proliferation, and induced HCC cell apoptosis, whereas HIF-1α knockdown did not. Additionally, we found that the PI3K inhibitor apitolisib counteracted the effect of HIF-2α, thereby inducing HCC cell apoptosis.
Conclusions
Our data highlight a role of HIF-2α in activating and binding c-MYC, thereby inducing HCC cell proliferation during mild chronic hypoxia. The PI3K/mTORC2/HIF-2α/c-MYC axis may play a key role in this process. The PI3K inhibitor apitolisib may serve as a potential treatment option for patients suffering from HCC, especially in cases with rapidly growing tumors under mild chronic hypoxic conditions.







Similar content being viewed by others
Data Availability
Data and material are available upon reasonable request.
Code Availability
Not applicable.
Abbreviations
- HCC:
-
hepatocellular carcinoma
- HIFs:
-
hypoxia-inducible factors
- TAE/TACE:
-
transcatheter arterial (chemo) embolization
- HK2:
-
glycolytic enzyme hexokinase 2
- PDK1:
-
pyruvate dehydrogenase kinase 1
- VEGFA:
-
vascular endothelial growth factor A
- mTOR:
-
mammalian target of rapamycin
- DAPI:
-
4′,6-diamidino-2-phenylindole
- CoCl2:
-
Cobalt chloride
- IC50:
-
50 % cell growth inhibitory concentrations
- MTT:
-
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- PLA:
-
in situ Proximity Ligation Assay
- Co-IP:
-
Co-Immunoprecipitation
- shRNA:
-
Short hairpin RNA
- ChIP:
-
Chromatin Immunoprecipitation
- HREs:
-
Hypoxia-Response Element
- UICC:
-
Union for International Cancer Control
- PHD:
-
prolyl-4-hydroxylase
- RCC:
-
renal cell carcinoma
References
A. Forner, M. Reig, J. Bruix, Hepatocellular carcinoma. Lancet 391, 1301–1314 (2018)
A. Villanueva, Hepatocellular carcinoma. N. Engl. J. Med. 380, 1450–1462 (2019)
J.M. Llovet, R. Montal, D. Sia, R.S. Finn, Molecular therapies and precision medicine for hepatocellular carcinoma. Nat. Rev. Clin. Oncol. 15, 599–616 (2018)
R.L. Siegel, K.D. Miller, A. Jemal, Cancer statistics, 2020. CA Cancer J. Clin. 70, 7–30 (2020)
S.R. McKeown, Defining normoxia, physoxia and hypoxia in tumours-implications for treatment response. Br. J. Radiol. 87, 20130676 (2014)
G.K. Wilson, D.A. Tennant, J.A. McKeating, Hypoxia inducible factors in liver disease and hepatocellular carcinoma: current understanding and future directions. J. Hepatol. 61, 1397–1406 (2014)
C. Chen, T. Lou, Hypoxia inducible factors in hepatocellular carcinoma. Oncotarget 8, 46691–46703 (2017)
C. Mendez-Blanco, F. Fondevila, A. Garcia-Palomo, J. Gonzalez-Gallego, J.L. Mauriz, Sorafenib resistance in hepatocarcinoma: role of hypoxia-inducible factors. Exp. Mol. Med. 50, 1–9 (2018)
L. Tang, J. Zeng, P. Geng, C. Fang, Y. Wang, M. Sun, C. Wang, J. Wang, P. Yin, C. Hu, L. Guo, J. Yu, P. Gao, E. Li, Z. Zhuang, G. Xu, Y. Liu, Global metabolic profiling identifies a pivotal role of proline and hydroxyproline metabolism in supporting hypoxic response in hepatocellular carcinoma. Clin. Cancer Res. 24, 474–485 (2018)
G.L. Semenza, Hypoxia-inducible factors in physiology and medicine. Cell 148, 399–408 (2012)
K. Helczynska, A.M. Larsson, L. Holmquist Mengelbier, E. Bridges, E. Fredlund, S. Borgquist, G. Landberg, S. Pahlman, K. Jirstrom, Hypoxia-inducible factor-2alpha correlates to distant recurrence and poor outcome in invasive breast cancer. Cancer Res. 68, 9212–9220 (2008)
R. Noguera, E. Fredlund, M. Piqueras, A. Pietras, S. Beckman, S. Navarro, S. Pahlman, HIF-1alpha and HIF-2alpha are differentially regulated in vivo in neuroblastoma: high HIF-1alpha correlates negatively to advanced clinical stage and tumor vascularization. Clin. Cancer Res. 15, 7130–7136 (2009)
W.Y. Kim, S. Perera, B. Zhou, J. Carretero, J.J. Yeh, S.A. Heathcote, A.L. Jackson, P. Nikolinakos, B. Ospina, G. Naumov, K.A. Brandstetter, V.J. Weigman, S. Zaghlul, D.N. Hayes, R.F. Padera, J.V. Heymach, A.L. Kung, N.E. Sharpless, W.G. Kaelin Jr., K.K. Wong, HIF2alpha cooperates with RAS to promote lung tumorigenesis in mice. J. Clin. Invest. 119, 2160–2170 (2009)
N. Qin, A.A. de Cubas, R. Garcia-Martin, S. Richter, M. Peitzsch, M. Menschikowski, J.W. Lenders, H.J. Timmers, M. Mannelli, G. Opocher, M. Economopoulou, G. Siegert, T. Chavakis, K. Pacak, M. Robledo, G. Eisenhofer, Opposing effects of HIF1alpha and HIF2alpha on chromaffin cell phenotypic features and tumor cell proliferation: Insights from MYC-associated factor X. Int. J. Cancer 135, 2054–2064 (2014)
B. Keith, R.S. Johnson, M.C. Simon, HIF1alpha and HIF2alpha: sibling rivalry in hypoxic tumour growth and progression. Nat. Rev. Cancer 12, 9–22 (2011)
L.E. Huang, Carrot and stick: HIF-alpha engages c-Myc in hypoxic adaptation. Cell Death Differ. 15, 672–677 (2008)
C.V. Dang, c-Myc target genes involved in cell growth, apoptosis, and metabolism. Mol. Cell. Biol. 19, 1–11 (1999)
J.W. Kim, P. Gao, Y.C. Liu, G.L. Semenza, C.V. Dang, Hypoxia-inducible factor 1 and dysregulated c-Myc cooperatively induce vascular endothelial growth factor and metabolic switches hexokinase 2 and pyruvate dehydrogenase kinase 1. Mol. Cell. Biol. 27, 7381–7393 (2007)
J. Zhang, M. Sattler, G. Tonon, C. Grabher, S. Lababidi, A. Zimmerhackl, M.S. Raab, S. Vallet, Y. Zhou, M.A. Cartron, T. Hideshima, Y.T. Tai, D. Chauhan, K.C. Anderson, K. Podar, Targeting angiogenesis via a c-Myc/hypoxia-inducible factor-1alpha-dependent pathway in multiple myeloma. Cancer Res. 69, 5082–5090 (2009)
M.R. Doe, J.M. Ascano, M. Kaur, M.D. Cole, Myc posttranscriptionally induces HIF1 protein and target gene expression in normal and cancer cells. Cancer Res. 72, 949–957 (2012)
C. Chen, S. Cai, G. Wang, X. Cao, X. Yang, X. Luo, Y. Feng, J. Hu, c-Myc enhances colon cancer cell-mediated angiogenesis through the regulation of HIF-1α. Biochem. Biophys. Res. Commun. 430, 505–511 (2013)
L. Ma, G. Li, H. Zhu, X. Dong, D. Zhao, X. Jiang, J. Li, H. Qiao, S. Ni, X. Sun, Sun, 2-Methoxyestradiol synergizes with sorafenib to suppress hepatocellular carcinoma by simultaneously dysregulating hypoxia-inducible factor-1 and – 2. Cancer Lett. 355, 96–105 (2014)
M.S. Wiesener, J.S. Jurgensen, C. Rosenberger, C.K. Scholze, J.H. Horstrup, C. Warnecke, S. Mandriota, I. Bechmann, U.A. Frei, C.W. Pugh, P.J. Ratcliffe, S. Bachmann, P.H. Maxwell, K.U. Eckardt, Widespread hypoxia-inducible expression of HIF-2alpha in distinct cell populations of different organs. FASEB J. 17, 271–273 (2003)
D.M. Sabatini, Twenty-five years of mTOR: Uncovering the link from nutrients to growth. Proc. Natl. Acad. Sci. U. S. A. 114, 11818–11825 (2017)
M. Laplante, D.M. Sabatini, mTOR signaling at a glance. J. Cell Sci. 122, 3589–3594 (2009)
P. Liu, H. Cheng, T.M. Roberts, J.J. Zhao, Targeting the phosphoinositide 3-kinase pathway in cancer. Nat. Rev. Drug Discov. 8, 627–644 (2009)
K.M. Dodd, J. Yang, M.H. Shen, J.R. Sampson, A.R. Tee, mTORC1 drives HIF-1alpha and VEGF-A signalling via multiple mechanisms involving 4E-BP1, S6K1 and STAT3. Oncogene 34, 2239–2250 (2015)
F. Melendez-Rodriguez, O. Roche, R. Sanchez-Prieto, J. Aragones, Hypoxia-inducible factor 2-dependent pathways driving Von Hippel-Lindau-deficient renal cancer. Front. Oncol. 8, 214 (2018)
H. Cam, J.B. Easton, A. High, P.J. Houghton, mTORC1 signaling under hypoxic conditions is controlled by ATM-dependent phosphorylation of HIF-1alpha. Mol. Cell 40, 509–520 (2010)
Z. Xu, M. Xu, P. Liu, S. Zhang, R. Shang, Y. Qiao, L. Che, S. Ribback, A. Cigliano, K. Evert, R.M. Pascale, F. Dombrowski, M. Evert, X. Chen, D.F. Calvisi, X. Chen, The mTORC2-Akt1 cascade Is crucial for c-Myc to promote hepatocarcinogenesis in mice and humans. Hepatology 70, 1600–1613 (2019)
P. Liu, M. Ge, J. Hu, X. Li, L. Che, K. Sun, L. Cheng, Y. Huang, M.G. Pilo, A. Cigliano, G.M. Pes, R.M. Pascale, S. Brozzetti, G. Vidili, A. Porcu, A. Cossu, G. Palmieri, M.C. Sini, S. Ribback, F. Dombrowski, J. Tao, D.F. Calvisi, L. Chen, X. Chen, A functional mammalian target of rapamycin complex 1 signaling is indispensable for c-Myc-driven hepatocarcinogenesis. Hepatology 66, 167–181 (2017)
X. Yang, Y. Xu, D. Gao, L. Yang, S.Y. Qian, Dihomo-gamma-linolenic acid inhibits growth of xenograft tumors in mice bearing human pancreatic cancer cells (BxPC-3) transfected with delta-5-desaturase shRNA. Redox Biol. 20, 236–246 (2019)
J.D. Gordan, J.A. Bertout, C.J. Hu, J.A. Diehl, M.C. Simon, HIF-2alpha promotes hypoxic cell proliferation by enhancing c-myc transcriptional activity. Cancer Cell 11, 335–347 (2007)
S. Mohlin, A. Hamidian, K. von Stedingk, E. Bridges, C. Wigerup, D. Bexell, S. Pahlman, PI3K-mTORC2 but not PI3K-mTORC1 regulates transcription of HIF2A/EPAS1 and vascularization in neuroblastoma. Cancer Res. 75, 4617–4628 (2015)
T. Powles, M.R. Lackner, S. Oudard, B. Escudier, C. Ralph, J.E. Brown, R.E. Hawkins, D. Castellano, B.I. Rini, M.D. Staehler, A. Ravaud, W. Lin, B. O’Keeffe, Y. Wang, S. Lu, J.M. Spoerke, L.Y. Huw, M. Byrtek, R. Zhu, J.A. Ware, R.J. Motzer, Randomized open-label Phase II trial of apitolisib (GDC-0980), a novel Inhibitor of the PI3K/mammalian target of rapamycin pathway, versus everolimus in patients with metastatic renal cell carcinoma. J. Clin. Oncol. 34, 1660–1668 (2016)
S.O. Dolly, A.J. Wagner, J.C. Bendell, H.L. Kindler, L.M. Krug, T.Y. Seiwert, M.G. Zauderer, M.P. Lolkema, D. Apt, R.F. Yeh, J.O. Fredrickson, J.M. Spoerke, H. Koeppen, J.A. Ware, J.O. Lauchle, H.A. Burris III., J.S. de Bono, Phase I study of apitolisib (GDC-0980), dual phosphatidylinositol-3-kinase and mammalian target of rapamycin kinase inhibitor, in patients with advanced solid tumors. Clin. Cancer Res. 22, 2874–2884 (2016)
V. Makker, F.O. Recio, L. Ma, U.A. Matulonis, J.O. Lauchle, H. Parmar, H.N. Gilbert, J.A. Ware, R. Zhu, S. Lu, L.Y. Huw, Y. Wang, H. Koeppen, J.M. Spoerke, M.R. Lackner, C.A. Aghajanian, A multicenter, single-arm, open-label, phase 2 study of apitolisib (GDC-0980) for the treatment of recurrent or persistent endometrial carcinoma (MAGGIE study). Cancer 122, 3519–3528 (2016)
M.R. Morris, D.J. Hughes, Y.M. Tian, C.J. Ricketts, K.W. Lau, D. Gentle, S. Shuib, P. Serrano-Fernandez, J. Lubinski, M.S. Wiesener, C.W. Pugh, F. Latif, P.J. Ratcliffe, E.R. Maher, Mutation analysis of hypoxia-inducible factors HIF1A and HIF2A in renal cell carcinoma. Anticancer Res. 29, 4337–4343 (2009)
G. Bangoura, Z.S. Liu, Q. Qian, C.Q. Jiang, G.F. Yang, S. Jing, Prognostic significance of HIF-2alpha/EPAS1 expression in hepatocellular carcinoma. World J. Gastroenterol. 13, 3176–3182 (2007)
K. Wei, S.M. Piecewicz, L.M. McGinnis, C.M. Taniguchi, S.J. Wiegand, K. Anderson, C.W. Chan, K.X. Mulligan, D. Kuo, J. Yuan, M. Vallon, L. Morton, E. Lefai, M.C. Simon, J.J. Maher, G. Mithieux, F. Rajas, J. Annes, O.P. McGuinness, G. Thurston, A.J. Giaccia, C.J. Kuo, A liver Hif-2alpha-Irs2 pathway sensitizes hepatic insulin signaling and is modulated by Vegf inhibition. Nat. Med. 19, 1331–1337 (2013)
S. Anavi, M. Hahn-Obercyger, Z. Madar, O. Tirosh, Mechanism for HIF-1 activation by cholesterol under normoxia: a redox signaling pathway for liver damage. Free Radic. Biol. Med. 71, 61–69 (2014)
Y. Asai, T. Yamada, S. Tsukita, K. Takahashi, M. Maekawa, M. Honma, M. Ikeda, K. Murakami, Y. Munakata, Y. Shirai, S. Kodama, T. Sugisawa, Y. Chiba, Y. Kondo, K. Kaneko, K. Uno, S. Sawada, J. Imai, Y. Nakamura, H. Yamaguchi, K. Tanaka, H. Sasano, N. Mano, Y. Ueno, T. Shimosegawa, H. Katagiri, Activation of the hypoxia inducible factor 1α subunit pathway in steatotic liver contributes to formation of cholesterol gallstones. Gastroenterology 152(6), 1521–1535.e8 (2017)
W. Hu, S. Zheng, H. Guo, B. Dai, J. Ni, Y. Shi, H. Bian, L. Li, Y. Shen, M. Wu, Z. Tian, G. Liu, M.A. Hossain, H. Yang, D. Wang, Q. Zhang, J. Yu, L. Birnbaumer, J. Feng, D. Yu, Y. Yang, PLAGL2-EGFR-HIF-1/2α signaling loop promotes HCC progression and Erlotinib insensitivity. Hepatology 73, 674-691 (2021)
A. Qu, M. Taylor, X. Xue, T. Matsubara, D. Metzger, P. Chambon, F.J. Gonzalez, Y.M. Shah, Hypoxia-inducible transcription factor 2alpha promotes steatohepatitis through augmenting lipid accumulation, inflammation, and fibrosis. Hepatology 54, 472–483 (2011)
X. Wang, J. Dong, L. Jia, T. Zhao, M. Lang, Z. Li, C. Lan, X. Li, J. Hao, H. Wang, T. Qin, C. Huang, S. Yang, M. Yu, H. Ren, HIF-2-dependent expression of stem cell factor promotes metastasis in hepatocellular carcinoma. Cancer Lett. 393, 113–124 (2017)
C. He, X.P. Sun, H. Qiao, X. Jiang, D. Wang, X. Jin, X. Dong, J. Wang, H. Jiang, X. Sun, Downregulating hypoxia-inducible factor-2alpha improves the efficacy of doxorubicin in the treatment of hepatocellular carcinoma. Cancer Sci. 103, 528–534 (2012)
H.Z. Imtiyaz, E.P. Williams, M.M. Hickey, S.A. Patel, A.C. Durham, L.J. Yuan, R. Hammond, P.A. Gimotty, B. Keith, M.C. Simon, Hypoxia-inducible factor 2alpha regulates macrophage function in mouse models of acute and tumor inflammation. J. Clin. Invest. 120, 2699–2714 (2010)
H. Menrad, C. Werno, T. Schmid, E. Copanaki, T. Deller, N. Dehne, B. Brune, Roles of hypoxia-inducible factor-1alpha (HIF-1alpha) versus HIF-2alpha in the survival of hepatocellular tumor spheroids. Hepatology 51, 2183–2192 (2010)
M. Koshiji, K.K. To, S. Hammer, K. Kumamoto, A.L. Harris, P. Modrich, L.E. Huang, HIF-1alpha induces genetic instability by transcriptionally downregulating MutSalpha expression. Mol. Cell 17, 793–803 (2005)
M. Koshiji, Y. Kageyama, E.A. Pete, I. Horikawa, J.C. Barrett, L.E. Huang, HIF-1alpha induces cell cycle arrest by functionally counteracting Myc. EMBO J. 23, 1949–1956 (2004)
T. Lofstedt, E. Fredlund, L. Holmquist-Mengelbier, A. Pietras, M. Ovenberger, L. Poellinger, S. Pahlman, Hypoxia inducible factor-2alpha in cancer. Cell Cycle 6, 919–926 (2007)
L. Holmquist-Mengelbier, E. Fredlund, T. Lofstedt, R. Noguera, S. Navarro, H. Nilsson, A. Pietras, J. Vallon-Christersson, A. Borg, K. Gradin, L. Poellinger, S. Pahlman, Recruitment of HIF-1alpha and HIF-2alpha to common target genes is differentially regulated in neuroblastoma: HIF-2alpha promotes an aggressive phenotype. Cancer Cell 10, 413–423 (2006)
P. Vaupel, M. Hockel, A. Mayer, Detection and characterization of tumor hypoxia using pO2 histography. Antioxid. Redox Signal. 9, 1221–1235 (2007)
M.W. Dewhirst, Y. Cao, B. Moeller, Cycling hypoxia and free radicals regulate angiogenesis and radiotherapy response. Nat. Rev. Cancer 8, 425–437 (2008)
Q. Lin, X. Cong, Z. Yun, Differential hypoxic regulation of hypoxia-inducible factors 1alpha and 2alpha. Mol. Cancer Res. 9, 757–765 (2011)
A.T. Henze, J. Riedel, T. Diem, J. Wenner, I. Flamme, J. Pouyseggur, K.H. Plate, T. Acker, Prolyl hydroxylases 2 and 3 act in gliomas as protective negative feedback regulators of hypoxia-inducible factors. Cancer Res. 70, 357–366 (2010)
M.Y. Koh, R. Lemos Jr., X. Liu, G. Powis, The hypoxia-associated factor switches cells from HIF-1alpha- to HIF-2alpha-dependent signaling promoting stem cell characteristics, aggressive tumor growth and invasion. Cancer Res. 71, 4015–4027 (2011)
A. Toschi, E. Lee, N. Gadir, M. Ohh, D.A. Foster, Differential dependence of hypoxia-inducible factors 1 alpha and 2 alpha on mTORC1 and mTORC2. J. Biol. Chem. 283, 34495–34499 (2008)
P.M. LoRusso, Inhibition of the PI3K/AKT/mTOR Pathway in Solid Tumors. J. Clin. Oncol. 34, 3803–3815 (2016)
Acknowledgements
We appreciated the technical support from Mr. Andreas Schmitt and Ms. Weiwei Ma.
Funding
This work was supported by Tianjin Medical University Cancer Institute and Hospital, Tianjin, China (NO. TJ20170110).
Author information
Authors and Affiliations
Contributions
HM and CFL conceived the project. HM and CFL performed all experiments and drafted the manuscript. MMW and TZ supported the experiments. HKL, GY and YLC collected clinical samples and information. CFL and TQS supervised all studies. All authors participated in preparing the manuscript and approved the submitted and published version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interests.
Ethics approval and consent to participate
All our experiments involving human participants were approved by The Ethics Committee of Tianjin Medical University and performed in accordance with the Declaration of Helsinki. We obtained human HCC tissue and adjacent normal tissue from HCC patients at the Tianjin Medical University Cancer Institute and Hospital with informed consent from all patients.
Consent for publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Fig. S1
The expressions and correlations of Rictor and Raptor in HCC patients. A-B. The number of HCC cases with low or high expressions of Rictor and Raptor in different UICC stage (PNG 119 kb)
Fig. S2
HIF-1α or HIF-2α knockdown in mild chronic hypoxic hepatocellular carcinoma cell. (A) The expressions of HIF-1α and HIF-2α in shHIF-1α or shHIF-2α transfected HCC cells treated by Cocl2low for 72 h. (B) The relative fold changes of HIF-1α, HIF-2α and c-MYC in the QRT-PCR analysis and the relative fold changes of c-MYC in the HIF-2α binding ChiP assay from the HCC samples treated with/without Cocl2low for 72 h (PNG 208 kb)
Fig. S3
c-MYC knockdown suppressed cell growth in mild chronic hypoxic hepatocellular carcinoma cell. (A) The expressions of c-MYC in shRNA c-MYC transfected HCC cells treated by Cocl2low for 72 h. (B) Cell proliferation curve of c-MYC knockdown Huh7 and HepG2 in mild hypoxia for 6 days (PNG 219 kb)
Fig. S4
Rictor knockdown in mild chronic hypoxic hepatocellular carcinoma cell. A. The expressions of Rictor in shRictor transfected HCC cells treated by Cocl2low for 72 h (PNG 83 kb)
Rights and permissions
About this article
Cite this article
Mu, H., Yu, G., Li, H. et al. Mild chronic hypoxia-induced HIF-2α interacts with c-MYC through competition with HIF-1α to induce hepatocellular carcinoma cell proliferation. Cell Oncol. 44, 1151–1166 (2021). https://doi.org/10.1007/s13402-021-00625-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13402-021-00625-w