Abstract
Purpose
Gastrointestinal stromal tumors (GISTs) are the most common mesenchymal tumors in the gastrointestinal tract. Previously, PDCD2 (programmed cell death protein 2) has been identified as a putative tumor suppressor in gastric cancer. As yet, however, no reports on PDCD2 expression and its physical interactor NCoR1 (nuclear receptor co-repressor), and their effects in GIST have been reported.
Methods
The expression of PDCD2 and NCoR1 was assessed in 43 primary gastric GIST and normal gastric tissue samples using Western blotting and quantitative real-time PCR. Next, associations between PDCD2 and NCoR1 expression and various clinicopathological features, including survival, were determined. To assess the effects of PDCD2 and NCoR1 expression in vitro, two GIST-derived cell lines (GIST-T1 and GIST882) were (co-)transfected with the expression vectors pEGFP-N1-PDCD2 and pcDNA3.1-NCoR1, after which the cells were subjected to CCK-8, PI staining and Annexin V-FITC/PI double staining assays, respectively. Finally, the mechanisms of action of PDCD2 and NCoR1 in GIST-derived cells were determined using immunoprecipitation and Western blotting assays.
Results
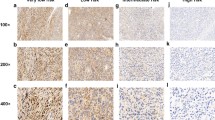
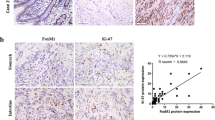
We found that the PDCD2 and NCoR1 protein levels were lower in gastric GIST tissues than in normal gastric tissues. The PDCD2 and NCoR1 expression levels were found to be significantly associated with the survival of the patients. Through exogenous expression analyses, we found that PDCD2 and NCoR1 can decrease proliferation, and increase apoptosis and G1 cell cycle arrest, in GIST-derived cells. Furthermore, we found that PDCD2 and NCoR1 can activate Smad2 and Smad3.
Conclusions
Our data indicate that both PDCD2 and NCoR1 may act as tumor suppressors in GIST cells through the Smad signaling pathway.




Similar content being viewed by others
References
M. Miettinen, M. Majidi, J. Lasota, Pathology and diagnostic criteria of gastrointestinal stromal tumors (GISTs): a review. Eur. J. Cancer 38, S39–S51 (2002)
H. S. Kim, S. S. Kim, S. G. Park, Bowel perforation associated sunitinib therapy for recurred gastric gastrointestinal stromal tumor. Ann. Surg. Treat. Res. 86, 220–225 (2014)
L. G. Kindblom, H. E. Remotti, F. Aldenborg, J. M. Meis-Kindblom, Gastrointestinal pacemaker cell tumor (GIPACT): gastrointestinal stromal tumors show phenotypic characteristics of the interstitial cells of Cajal. Am. J. Pathol. 152, 1259–1269 (1998)
S. Minakhina, N. Changela, R. Steward, Zfrp8/PDCD2 is required in ovarian stem cells and interacts with the piRNA pathway machinery. Development 141, 259–268 (2014)
M. Merup, T. C. Moreno, M. Heyman, K. Rönnberg, D. Grandér, R. Detlofsson, O. Rasool, Y. Liu, S. Söderhäll, G. Juliusson, G. Gahrton, S. Einhorn, 6q deletions in acute lymphoblastic leukemia and non-Hodgkin’s lymphomas. Blood 91, 3397–3400 (1998)
J. Zhang, W. Wei, H. C. Jin, R. C. Ying, A. K. Zhu, F. J. Zhang, The roles of APOBEC3B in gastric cancer. Int. J. Clin. Exp. Pathol. 8, 5089–5096 (2015)
J. Zhang, W. Wei, H. C. Jin, R. C. Ying, A. K. Zhu, F. J. Zhang, Programmed cell death 2 protein induces gastric cancer cell growth arrest at the early S phase of the cell cycle and apoptosis in a p53-dependent manner. Oncol. Rep. 33, 103–110 (2015)
D. Szklarczyk, A. Franceschini, M. Kuhn, M. Simonovic, A. Roth, P. Minguez, T. Doerks, M. Stark, J. Muller, P. Bork, L. J. Jensen, C. von Mering, The STRING database in 2011: functional interaction networks of proteins, globally integrated and scored. Nucleic Acids Res. 39, D561–D568 (2011)
R. B. Scarr, P. A. Sharp, PDCD2 is a negative regulator of HCF-1 (C1). Oncogene 21, 5245–5254 (2002)
X. G. Zhu, D. W. Kim, M. L. Goodson, M. L. Privalsky, S. Y. Cheng, NCoR1 regulates thyroid hormone receptor isoform-dependent adipogenesis. J. Mol. Endocrinol. 46, 233–244 (2011)
T. Taguchi, H. Sonobe, S. Toyonaga, I. Yamasaki, T. Shuin, A. Takano, K. Araki, K. Akimaru, K. Yuri, Conventional and molecular cytogenetic characterization of a new human cell line, GIST-T1, established from gastrointestinal stromal tumor. Lab. Investig. 82, 663–665 (2002)
B. W. Baron, N. Zeleznik-Le, M. J. Baron, C. Theisler, D. Huo, M. D. Krasowski, M. J. Thirman, R. M. Baron, J. M. Baron, Repression of the PDCD2 gene by BCL6 and the implications for the pathogenesis of human B and T cell lymphomas. Proc. Natl. Acad. Sci. U. S. A. 104, 7449–7454 (2007)
J. Kramer, C. J. Granier, S. Davis, K. Piso, J. Hand, A. B. Rabson, H. E. Sabaawy, PDCD2 controls hematopoietic stem cell differentiation during development. Stem Cells Dev. 22, 58–72 (2013)
R. M. Lavinsky, K. Jepsen, T. Heinzel, J. Torchia, T. M. Mullen, R. Schiff, A. L. Del-Rio, M. Ricote, S. Ngo, J. Gemsch, S. G. Hilsenbeck, C. K. Osborne, C. K. Glass, M. G. Rosenfeld, D. W. Rose, Diverse signaling pathways modulate nuclear receptor recruitment of N-CoR and SMRT complexes. Proc. Natl. Acad. Sci. U. S. A. 95, 2920–2925 (1998)
J. Kurebayashi, T. Otsuki, H. Kunisue, K. Tanaka, S. Yamamoto, H. Sonoo, Expression levels of estrogen receptor-alpha, estrogen receptor-beta, coactivators, and corepressors in breast cancer. Clin. Cancer Res. 6, 512–518 (2000)
Z. H. Zhang, H. Yamashita, T. Toyama, Y. Yamamoto, T. Kawasoe, M. Ibusuki, S. Tomita, H. Sugiura, S. Kobayashi, Y. Fujii, H. Iwase, Nuclear corepressor 1 expression predicts response to first-line endocrine therapy for breast cancer patients on relapse. Chin. Med. J. 122, 1764–1768 (2009)
B. W. Baron, E. Hyjek, B. Gladstone, M. J. Thirman, J. M. Baron, PDCD2, a protein whose expression is repressed by BCL6, induces apoptosis in human cells by activation of the caspase cascade. Blood Cells Mol. Dis. 45, 169–175 (2010)
H. Kashima, A. Horiuchi, J. Uchikawa, T. Miyamoto, A. Suzuki, T. Ashida, I. Konishi, T. Shiozawa, Up-regulation of nuclear receptor corepressor (NCoR) in progestin-induced growth suppression of endometrial hyperplasia and carcinoma. Anticancer Res. 29, 1023–1029 (2009)
M. O. Lee, H. J. Kang, Role of coactivators and corepressors in the induction of the RAR beta gene in human colon cancer cells. Biol. Pharm. Bull. 25, 1298–1302 (2002)
B. Schmierer, C. S. Hill, TGFbeta-SMAD signal transduction: molecular specificity and functional flexibility. Nat. Rev. Mol. Cell. Biol. 8, 970–982 (2007)
M. K. Wendt, J. A. Smith, W. P. Schiemann, p130Cas is required for mammary tumor growth and transforming growth factor-beta-mediated metastasis through regulation of Smad2/3 activity. J. Biol. Chem. 284, 34145–34156 (2009)
M. R. Tang, Y. X. Wang, S. Guo, S. Y. Han, D. Wang, CSMD1 exhibits antitumor activity in A375 melanoma cells through activation of the Smad pathway. Apoptosis 17, 927–937 (2012)
D. Zurlo, C. Leone, G. Assante, S. Salzano, G. Renzone, A. Scaloni, C. Foresta, V. Colantuoni, A. Lupo, Cladosporol a stimulates G1-phase arrest of the cell cycle by up-regulation of p21(waf1/cip1) expression in human colon carcinoma HT-29 cells. Mol. Carcinog. 52, 1–17 (2013)
X. R. Han, Y. Sun, X. Z. Bai, The anti-tumor role and mechanism of integrated and truncated PDCD5 proteins in osteosarcoma cells. Cell. Signal. 24, 1713–1721 (2012)
M. E. Han, S. J. Baek, S. Y. Kim, C. D. Kang, S. O. Oh, ATOH1 can regulate the tumorigenicity of gastric cancer cells by inducing the differentiation of cancer stem cells. PLoS One 10, e0126085 (2015)
Acknowledgments
This study was supported by the Natural Scientific Foundation of Liaoning Province (2014021036), General Project Scientific Research from the Department of Education of Liaoning Province (L2012288) and Natural Scientific Foundation of China (81502107).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that no competing interests exist.
Rights and permissions
About this article
Cite this article
Wang, W., Song, XW., Bu, XM. et al. PDCD2 and NCoR1 as putative tumor suppressors in gastric gastrointestinal stromal tumors. Cell Oncol. 39, 129–137 (2016). https://doi.org/10.1007/s13402-015-0258-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13402-015-0258-0