Abstract
Purpose
The overall survival rate of patients with advanced gastric cancer is poor. Therefore, there is an urgent need for new treatment options for these patients. The identification of drug target genes located on DNA regions exhibiting high-level copy number gains (CNG) may be an effective approach, as has e.g. previously been shown for HER2. The aim of the present study was to identify putative drug targets in patients with gastric cancer by applying this strategy.
Methods
Genome-wide array comparative genomic hybridization (array CGH) data available from 183 primary gastric cancer samples were analyzed through Ingenuity Pathway Analysis (IPA) to assess whether any established or potential anticancer drug target genes showed high-level CNG, including focal amplifications.
Results
A total of 147 high-level gained regions were identified in the gastric cancer samples, harboring 167 genes that had previously been annotated as drug target genes. Thirty (18 %) of these genes showed high-level gains in at least 2 % of the tumors. The identified drug target genes included those for drugs known to be active in advanced (gastric) cancer, targets for targeted therapies in clinical development, as well as targets for drugs currently used for other indications but of potential interest for anticancer treatment. In addition, 12 potential drug target genes were identified, including genes involved in growth factor signaling and cell cycle regulation.
Conclusion
The majority of gastric cancers carried one or more high-level CNGs or focal amplifications encompassing putative drug target genes. A number of the associated drugs are currently not being considered for treatment of gastric cancer. Based on these results we hypothesize that DNA copy number profiling may be a useful tool to identify new drug targets and to guide individualized treatment strategies in patients with gastric cancer.



Similar content being viewed by others
References
D. Cunningham, N. Starling, S. Rao, T. Iveson, M. Nicolson, F. Coxon, G. Middleton, F. Daniel, J. Oates, A.R. Norman, Capecitabine and oxaliplatin for advanced esophagogastric cancer. N. Engl. J. Med. 358, 36–46 (2008)
E. Van Cutsem, V.M. Moiseyenko, S. Tjulandin, A. Majlis, M. Constenla, C. Boni, A. Rodrigues, M. Fodor, Y. Chao, E. Voznyi, M.L. Risse, J.A. Ajani, Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: a report of the V325 Study Group. J. Clin. Oncol. 24, 4991–4997 (2006)
A.D. Wagner, S. Unverzagt, W. Grothe, G. Kleber, A. Grothey, J. Haerting, W.E. Fleig, Chemotherapy for advanced gastric cancer. Cochrane Database Syst. Rev., CD004064 (2010)
A.D. Wagner, W. Grothe, J. Haerting, G. Kleber, A. Grothey, W.E. Fleig, Chemotherapy in advanced gastric cancer: a systematic review and meta-analysis based on aggregate data. J. Clin. Oncol. 24, 2903–2909 (2006)
V. Catalano, F. Graziano, D. Santini, S. D'Emidio, A.M. Baldelli, D. Rossi, B. Vincenzi, P. Giordani, P. Alessandroni, E. Testa, G. Tonini, G. Catalano, Second-line chemotherapy for patients with advanced gastric cancer: who may benefit? Br. J. Cancer 99, 1402–1407 (2008)
P.C. Thuss-Patience, A. Kretzschmar, D. Bichev, T. Deist, A. Hinke, K. Breithaupt, Y. Dogan, B. Gebauer, G. Schumacher, P. Reichardt, Survival advantage for irinotecan versus best supportive care as second-line chemotherapy in gastric cancer–a randomised phase III study of the Arbeitsgemeinschaft Internistische Onkologie (AIO). Eur. J. Cancer 47, 2306–2314 (2011)
S.J. Gong, C.J. Jin, S.Y. Rha, H.C. Chung, Growth inhibitory effects of trastuzumab and chemotherapeutic drugs in gastric cancer cell lines. Cancer Lett. 214, 215–224 (2004)
K. Fujimoto-Ouchi, F. Sekiguchi, H. Yasuno, Y. Moriya, K. Mori, Y. Tanaka, Antitumor activity of trastuzumab in combination with chemotherapy in human gastric cancer xenograft models. Cancer Chemother. Pharmacol. 59, 795–805 (2007)
H. Grabsch, S. Sivakumar, S. Gray, H.E. Gabbert, W. Muller, HER2 expression in gastric cancer: rare, heterogeneous and of no prognostic value–conclusions from 924 cases of two independent series. Cell. Oncol. 32, 57–65 (2010)
C. Gravalos, A. Jimeno, HER2 in gastric cancer: a new prognostic factor and a novel therapeutic target. Ann. Oncol. 19, 1523–1529 (2008)
M. Tanner, M. Hollmen, T.T. Junttila, A.I. Kapanen, S. Tommola, Y. Soini, H. Helin, J. Salo, H. Joensuu, E. Sihvo, K. Elenius, J. Isola, Amplification of HER-2 in gastric carcinoma: association with Topoisomerase IIalpha gene amplification, intestinal type, poor prognosis and sensitivity to trastuzumab. Ann. Oncol. 16, 273–278 (2005)
Y.J. Bang, C.E. Van, A. Feyereislova, H.C. Chung, L. Shen, A. Sawaki, F. Lordick, A. Ohtsu, Y. Omuro, T. Satoh, G. Aprile, E. Kulikov, J. Hill, M. Lehle, J. Ruschoff, Y.K. Kang, Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet 376, 687–697 (2010)
H.H. Hartgrink, C.J. van de Velde, H. Putter, J.J. Bonenkamp, K.E. Klein, I. Songun, K. Welvaart, J.H. van Krieken, S. Meijer, J.T. Plukker, P.J. van Elk, H. Obertop, D.J. Gouma, J.J. van Lanschot, C.W. Taat, P.W. de Graaf, M.F. von Meyenfeldt, H. Tilanus, M. Sasako, Extended lymph node dissection for gastric cancer: who may benefit? Final results of the randomized Dutch gastric cancer group trial. J. Clin. Oncol. 22, 2069–2077 (2004)
T.E. Buffart, B. Carvalho, N.C. van Grieken, W.N. van Wieringen, M. Tijssen, E.M. Kranenbarg, H.M. Verheul, H.I. Grabsch, B. Ylstra, C.J. van de Velde, G.A. Meijer, Losses of chromosome 5q and 14q are associated with favorable clinical outcome of patients with gastric cancer. Oncologist 17, 653–662 (2012)
T.E. Buffart, N.C. van Grieken, M. Tijssen, J. Coffa, B. Ylstra, H.I. Grabsch, C.J. van de Velde, B. Carvalho, G.A. Meijer, High resolution analysis of DNA copy-number aberrations of chromosomes 8, 13, and 20 in gastric cancers. Virchows Arch. 455, 213–223 (2009)
R.P. Brosens, J.C. Haan, B. Carvalho, F. Rustenburg, H. Grabsch, P. Quirke, A.F. Engel, M.A. Cuesta, N. Maughan, M. Flens, G.A. Meijer, B. Ylstra, Candidate driver genes in focal chromosomal aberrations of stage II colon cancer. J. Pathol. 221, 411–424 (2010)
A.S. Hinrichs, D. Karolchik, R. Baertsch, G.P. Barber, G. Bejerano, H. Clawson, M. Diekhans, T.S. Furey, R.A. Harte, F. Hsu, J. Hillman-Jackson, R.M. Kuhn, J.S. Pedersen, A. Pohl, B.J. Raney, K.R. Rosenbloom, A. Siepel, K.E. Smith, C.W. Sugnet, A. Sultan-Qurraie, D.J. Thomas, H. Trumbower, R.J. Weber, M. Weirauch, A.S. Zweig, D. Haussler, W.J. Kent, The UCSC genome browser database: update 2006. Nucleic Acids Res. 34, D590–D598 (2006)
M.A. Sanz, Treatment of acute promyelocytic leukemia. Hematology. Am. Soc. Hematol. Educ. Program., 147–155 (2006)
J. Voortman, J.H. Lee, J.K. Killian, M. Suuriniemi, Y. Wang, M. Lucchi, W.I. Smith Jr., P. Meltzer, Y. Wang, G. Giaccone, Array comparative genomic hybridization-based characterization of genetic alterations in pulmonary neuroendocrine tumors. Proc. Natl. Acad. Sci. U. S. A. 107, 13040–13045 (2010)
N. Deng, L.K. Goh, H. Wang, K. Das, J. Tao, I.B. Tan, S. Zhang, M. Lee, J. Wu, K.H. Lim, Z. Lei, G. Goh, Q.Y. Lim, A.L. Tan, D.Y. Sin Poh, S. Riahi, S. Bell, M.M. Shi, R. Linnartz, F. Zhu, K.G. Yeoh, H.C. Toh, W.P. Yong, H.C. Cheong, S.Y. Rha, A. Boussioutas, H. Grabsch, S. Rozen, P. Tan, A comprehensive survey of genomic alterations in gastric cancer reveals systematic patterns of molecular exclusivity and co-occurrence among distinct therapeutic targets. Gut 61, 673–684 (2012)
A.M. Dulak, S.E. Schumacher, J. van Lieshout, Y. Imamura, C. Fox, B. Shim, A.H. Ramos, G. Saksena, S.C. Baca, J. Baselga, J. Tabernero, J. Barretina, P.C. Enzinger, G. Corso, F. Roviello, L. Lin, S. Bandla, J.D. Luketich, A. Pennathur, M. Meyerson, S. Ogino, R.A. Shivdasani, D.G. Beer, T.E. Godfrey, R. Beroukhim, A.J. Bass, Gastrointestinal adenocarcinomas of the esophagus, stomach, and colon exhibit distinct patterns of genome instability and oncogenesis. Cancer Res. 72, 4383–4393 (2012)
P. Seve, C. Dumontet, Is class III beta-tubulin a predictive factor in patients receiving tubulin-binding agents? Lancet Oncol. 9, 168–175 (2008)
P. Seve, J. Mackey, S. Isaac, O. Tredan, P.J. Souquet, M. Perol, R. Lai, A. Voloch, C. Dumontet, Class III beta-tubulin expression in tumor cells predicts response and outcome in patients with non-small cell lung cancer receiving paclitaxel. Mol. Cancer Ther. 4, 2001–2007 (2005)
G. Ferrandina, G.F. Zannoni, E. Martinelli, A. Paglia, V. Gallotta, S. Mozzetti, G. Scambia, C. Ferlini, Class III beta-tubulin overexpression is a marker of poor clinical outcome in advanced ovarian cancer patients. Clin. Cancer Res. 12, 2774–2779 (2006)
A. Paradiso, A. Mangia, A. Chiriatti, S. Tommasi, A. Zito, A. Latorre, F. Schittulli, V. Lorusso, Biomarkers predictive for clinical efficacy of taxol-based chemotherapy in advanced breast cancer. Ann. Oncol. 16 Suppl 4, iv14–iv19 (2005)
N. Urano, Y. Fujiwara, Y. Doki, S.J. Kim, Y. Miyoshi, S. Noguchi, H. Miyata, S. Takiguchi, T. Yasuda, M. Yano, M. Monden, Clinical significance of class III beta-tubulin expression and its predictive value for resistance to docetaxel-based chemotherapy in gastric cancer. Int. J. Oncol. 28, 375–381 (2006)
S.Y. Kanta, T. Yamane, Y. Dobashi, F. Mitsui, K. Kono, A. Ooi, Topoisomerase IIalpha gene amplification in gastric carcinomas: correlation with the HER2 gene. An immunohistochemical, immunoblotting, and multicolor fluorescence in situ hybridization study. Hum. Pathol. 37, 1333–1343 (2006)
Z. Liang, X. Zeng, J. Gao, S. Wu, P. Wang, X. Shi, J. Zhang, T. Liu, Analysis of EGFR, HER2, and TOP2A gene status and chromosomal polysomy in gastric adenocarcinoma from Chinese patients. BMC Cancer 8, 363 (2008)
A. Webb, D. Cunningham, J.H. Scarffe, P. Harper, A. Norman, J.K. Joffe, M. Hughes, J. Mansi, M. Findlay, A. Hill, J. Oates, M. Nicolson, T. Hickish, M. O'Brien, T. Iveson, M. Watson, C. Underhill, A. Wardley, M. Meehan, Randomized trial comparing epirubicin, cisplatin, and fluorouracil versus fluorouracil, doxorubicin, and methotrexate in advanced esophagogastric cancer. J. Clin. Oncol. 15, 261–267 (1997)
S. Corporaal, W.M. Smit, M.G. Russel, J. van der Palen, H. Boot, M.C. Legdeur, Capecitabine, epirubicin and cisplatin in the treatment of oesophagogastric adenocarcinoma. Neth. J. Med. 64, 141–146 (2006)
A.H. Marx, L. Tharun, J. Muth, A.M. Dancau, R. Simon, E. Yekebas, J.T. Kaifi, M. Mirlacher, T.H. Brummendorf, C. Bokemeyer, J.R. Izbicki, G. Sauter, HER-2 amplification is highly homogenous in gastric cancer. Hum. Pathol. 40, 769–777 (2009)
C. Bozzetti, F.V. Negri, C.A. Lagrasta, P. Crafa, C. Bassano, I. Tamagnini, G. Gardini, R. Nizzoli, F. Leonardi, D. Gasparro, R. Camisa, S. Cavalli, E.M. Silini, A. Ardizzoni, Comparison of HER2 status in primary and paired metastatic sites of gastric carcinoma. Br. J. Cancer 104, 1372–1376 (2011)
J.T. Jorgensen, Targeted HER2 treatment in advanced gastric cancer. Oncology 78, 26–33 (2010)
A. Varis, A. Zaika, P. Puolakkainen, B. Nagy, I. Madrigal, A. Kokkola, A. Vayrynen, P. Karkkainen, C. Moskaluk, W. El-Rifai, S. Knuutila, Coamplified and overexpressed genes at ERBB2 locus in gastric cancer. Int. J. Cancer 109, 548–553 (2004)
M.F. Press, G. Sauter, M. Buyse, L. Bernstein, R. Guzman, A. Santiago, I.E. Villalobos, W. Eiermann, T. Pienkowski, M. Martin, N. Robert, J. Crown, V. Bee, H. Taupin, K.J. Flom, I. Tabah-Fisch, G. Pauletti, M.A. Lindsay, A. Riva, D.J. Slamon, Alteration of topoisomerase II-alpha gene in human breast cancer: association with responsiveness to anthracycline-based chemotherapy. J. Clin. Oncol. 29, 859–867 (2011)
L.A. Di, C. Desmedt, J.M. Bartlett, F. Piette, B. Ejlertsen, K.I. Pritchard, D. Larsimont, C. Poole, J. Isola, H. Earl, H. Mouridsen, F.P. O'Malley, F. Cardoso, M. Tanner, A. Munro, C.J. Twelves, C. Sotiriou, L. Shepherd, D. Cameron, M.J. Piccart, M. Buyse, HER2 and TOP2A as predictive markers for anthracycline-containing chemotherapy regimens as adjuvant treatment of breast cancer: a meta-analysis of individual patient data. Lancet Oncol. 12, 1134–1142 (2011)
A.F. Okines, L.C. Thompson, D. Cunningham, A. Wotherspoon, J.S. Reis-Filho, R.E. Langley, T.S. Waddell, D. Noor, Z. Eltahir, R. Wong, S. Stenning, Effect of HER2 on prognosis and benefit from peri-operative chemotherapy in early oesophago-gastric adenocarcinoma in the MAGIC trial. Ann. Oncol. 24, 1253–1261 (2013)
T. Deng, J.C. Liu, K.I. Pritchard, A. Eisen, E. Zacksenhaus, Preferential killing of breast tumor initiating cells by N, N-diethyl-2-[4-(phenylmethyl)phenoxy]ethanamine/tesmilifene. Clin. Cancer Res. 15, 119–130 (2009)
L. Reyno, L. Seymour, D. Tu, S. Dent, K. Gelmon, B. Walley, A. Pluzanska, V. Gorbunova, A. Garin, J. Jassem, T. Pienkowski, J. Dancey, L. Pearce, M. MacNeil, S. Marlin, D. Lebwohl, M. Voi, K. Pritchard, Phase III study of N, N-diethyl-2-[4-(phenylmethyl) phenoxy]ethanamine (BMS-217380-01) combined with doxorubicin versus doxorubicin alone in metastatic/recurrent breast cancer: National Cancer Institute of Canada Clinical Trials Group Study MA.19. J. Clin. Oncol. 22, 269–276 (2004)
I.J. Waterman, V.A. Zammit, Differential effects of fenofibrate or simvastatin treatment of rats on hepatic microsomal overt and latent diacylglycerol acyltransferase activities. Diabetes 51, 1708–1713 (2002)
I.R. Konings, A. van der Gaast, L.J. van der Wijk, F.E. de Jongh, F.A. Eskens, S. Sleijfer, The addition of pravastatin to chemotherapy in advanced gastric carcinoma: a randomised phase II trial. Eur. J. Cancer 46, 3200–3204 (2010)
L. Gong, R.B. Altman, T.E. Klein, Bisphosphonates pathway. Pharmacogenet. Genomics 21, 50–53 (2011)
G. Chen, M. Rong, D. Luo, TNFRSF6B neutralization antibody inhibits proliferation and induces apoptosis in hepatocellular carcinoma cell. Pathol. Res. Pract. 206, 631–641 (2010)
V. Brinkmann, A. Billich, T. Baumruker, P. Heining, R. Schmouder, G. Francis, S. Aradhye, P. Burtin, Fingolimod (FTY720): discovery and development of an oral drug to treat multiple sclerosis. Nat. Rev. Drug Discov. 9, 883–897 (2010)
B.J. Tan, B.H. Tan, G.N. Chiu, Effect of triptolide on focal adhesion kinase and survival in MCF-7 breast cancer cells. Oncol. Rep. 26, 1315–1321 (2011)
S. Lapenna, A. Giordano, Cell cycle kinases as therapeutic targets for cancer. Nat. Rev. Drug Discov. 8, 547–566 (2009)
A. Ohtsu, M.A. Shah, C.E. Van, S.Y. Rha, A. Sawaki, S.R. Park, H.Y. Lim, Y. Yamada, J. Wu, B. Langer, M. Starnawski, Y.K. Kang, Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a randomized, double-blind, placebo-controlled phase III study. J. Clin. Oncol. 29, 3968–3976 (2011)
E. Van Cutsem, S. de Haas, Y.K. Kang, A. Ohtsu, N.C. Tebbutt, X.J. Ming, Y.W. Peng, B. Langer, P. Delmar, S.J. Scherer, M.A. Shah, Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a biomarker evaluation from the AVAGAST randomized phase III trial. J. Clin. Oncol. 30, 2119–2127 (2012)
S.A. Lang, D. Klein, C. Moser, A. Gaumann, G. Glockzin, M.H. Dahlke, W. Dietmaier, U. Bolder, H.J. Schlitt, E.K. Geissler, O. Stoeltzing, Inhibition of heat shock protein 90 impairs epidermal growth factor-mediated signaling in gastric cancer cells and reduces tumor growth and vascularization in vivo. Mol. Cancer Ther. 6, 1123–1132 (2007)
L. Xie, X. Su, L. Zhang, X. Yin, L. Tang, X. Zhang, Y. Xu, Z. Gao, K. Liu, M. Zhou, B. Gao, D. Shen, L. Zhang, J. Ji, P.R. Gavine, J. Zhang, E. Kilgour, X. Zhang, Q. Ji, FGFR2 gene amplification in gastric cancer predicts sensitivity to the selective FGFR inhibitor AZD4547. Clin. Cancer Res. 19, 2572–2583 (2013)
J. Lee, J.W. Seo, H.J. Jun, C.S. Ki, S.H. Park, Y.S. Park, H.Y. Lim, M.G. Choi, J.M. Bae, T.S. Sohn, J.H. Noh, S. Kim, H.L. Jang, J.Y. Kim, K.M. Kim, W.K. Kang, J.O. Park, Impact of MET amplification on gastric cancer: possible roles as a novel prognostic marker and a potential therapeutic target. Oncol. Rep. 25, 1517–1524 (2011)
M. Nakajima, H. Sawada, Y. Yamada, A. Watanabe, M. Tatsumi, J. Yamashita, M. Matsuda, T. Sakaguchi, T. Hirao, H. Nakano, The prognostic significance of amplification and overexpression of c-met and c-erb B-2 in human gastric carcinomas. Cancer 85, 1894–1902 (1999)
Y.Y. Janjigian, L.H. Tang, D.G. Coit, D.P. Kelsen, T.D. Francone, M.R. Weiser, S.C. Jhanwar, M.A. Shah, MET expression and amplification in patients with localized gastric cancer. Cancer Epidemiol. Biomarkers Prev. 20, 1021–1027 (2011)
Acknowledgments
We thank the Mapping Core and Map Finishing groups of the Wellcome Trust Sanger Institute for initial clone supply and verification. This work was financially supported by the AEGON International Scholarship in Oncology.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Tineke E. Buffart and Josien C. Haan contributed equally
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary table 1
Overview of all high-level copy number gains observed in 183 gastric cancers including the chromosomal position and size (Mb). (DOCX 38 kb)
Supplementary table 2
Chromosomal start and end positions of the high-level copy number gains containing the putative drug target genes. Positions are represented according to the NCBI Build 35/hg17 and, after conversion, the GRCh37/hg19 assembly of the UCSC Genome Browser. Chr, chromosome; Start resp End, genome position in basepairs. (DOCX 24.4 kb)
Supplementary table 3
Target genes of drugs without apparent anticancer effect showing high-level copy number gain in at least 2 % of the samples. IPA, Ingenuity Pathway Analysis; DIP, Database of Interacting Proteins; NA, not applicable. (DOCX 23.4 kb)
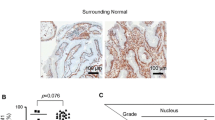
Supplementary figure 1
Two examples of HER2 protein expression in tissue microarray cores of gastric tumors with HER2 amplification showing over-expression (40×). (DOCX 14.2 MB)
Rights and permissions
About this article
Cite this article
Labots, M., Buffart, T.E., Haan, J.C. et al. High-level copy number gains of established and potential drug target genes in gastric cancer as a lead for treatment development and selection. Cell Oncol. 37, 41–52 (2014). https://doi.org/10.1007/s13402-013-0162-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13402-013-0162-4