Abstract
Purpose
SOX18 is a transcription factor known to be involved in hair follicle, blood and lymphatic vessel development, as well as wound healing processes (together with SOX7 and SOX17). In addition, it has been reported that SOX18 may affect the growth of cancer cells in vitro. Until now, the exact role of SOX18 expression in invasive ductal breast carcinoma (IDC) has remained unknown.
Methods
In this study, we have investigated SOX18 expression in cancer cells and endothelial cells in 122 IDC samples using immunohistochemistry (IHC). SOX18 expression was also determined using real-time PCR and Western blotting in a series of breast cancer-derived cell lines (i.e., MCF-7, BT-474, SK-BR-3, MDA-MB-231, BO2).
Results
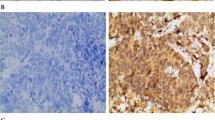
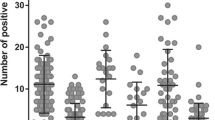
Using IHC, we observed SOX18 nuclear expression in cancer cells, as well as in blood and lymphatic vessels of the IDC samples tested. SOX18 expression in the IDC samples correlated with a higher malignancy grade (Grade 2 and Grade 3 versus Grade 1; p = 0.02 and p = 0.009, respectively) and VEGF-D expression (r = 0.27, p = 0.007). SOX18 expression was also associated with HER2 positivity (p = 0.02). A significantly higher SOX18 expression was found in the HER2-positive cell line BT-474, and a significantly lower expression in the triple negative cell lines MDA-MB-231 and BO2. Laser capture microdissection of IDC samples revealed significantly higher mRNA SOX7, SOX17 and SOX18 expression levels in the vessels as compared to the cancer cells (p = 0.02 and p = 0.0002, p < 0.0001, respectively). SOX18 positive intratumoral and peritumoral microvessel counts (MVC) were associated with higher malignancy grades (p = 0.04 and p = 0.02, respectively). Moreover, peritumoral SOX18 positive MVC were found to act as an independent marker for a poor prognosis (p = 0.04).
Conclusion
SOX18 expression may serve as a marker for a poor prognosis in IDC.







Similar content being viewed by others
References
R. Siegel, D. Naishadham, A. Jemal, Cancer statistics, 2013. CA Cancer J Clin. 63, 11–30 (2013)
M. Wegner, From head to toes: the multiple facets of Sox proteins. Nucleic Acids Res. 27, 1409–1420 (1999)
V.R. Harley, R. Lovell-Badge, P.N. Goodfellow, Definition of a consensus DNA binding site for SRY. Nucleic Acids Res. 22, 1500–1501 (1994)
M. Wegner, All purpose Sox: The many roles of Sox proteins in gene expression. Int J Biochem Cell Biol. 42, 381–390 (2010)
T.L. Dunn, L. Mynett-Johnson, E.M. Wright, B.M. Hosking, P.A. Koopman, G.E. Muscat, Sequence and expression of Sox-18 encoding a new HMG-box transcription factor. Gene. 161, 223–225 (1995)
Y. Kanai, M. Kanai-Azuma, T. Noce, T.C. Saido, T. Shiroishi, Y. Hayashi, K. Yazaki, Identification of two Sox17 messenger RNA isoforms, with and without the high mobility group box region, and their differential expression in mouse spermatogenesis. J Cell Biol. 133, 667–681 (1996)
K. Taniguchi, Y. Hiraoka, M. Ogawa, Y. Sakai, S. Kido, S. Aiso, Isolation and characterization of a mouse SRY-related cDNA, mSox7. Biochim Biophys Acta. 1445, 225–231 (1999)
M. Francois, P. Koopman, M. Beltrame, SoxF genes: Key players in the development of the cardio-vascular system. Int J Biochem Cell Biol. 42, 445–448 (2010)
S. Cermenati, S. Moleri, S. Cimbro, P. Corti, L. Del Giacco, R. Amodeo, E. Dejana, P. Koopman, F. Cotelli, M. Beltrame, Sox18 and Sox7 play redundant roles in vascular development. Blood. 111, 2657–2666 (2008)
B. Hosking, M. Francois, D. Wilhelm, F. Orsenigo, A. Caprini, T. Svingen, D. Tutt, T. Davidson, C. Browne, E. Dejana, P. Koopman, Sox7 and Sox17 are strain-specific modifiers of the lymphangiogenic defects caused by Sox18 dysfunction in mice. Development. 136, 2385–2391 (2009)
T. Matsui, M. Kanai-Azuma, K. Hara, S. Matoba, R. Hiramatsu, H. Kawakami, M. Kurohmaru, P. Koopman, Y. Kanai, Redundant roles of Sox17 and Sox18 in postnatal angiogenesis in mice. J Cell Sci. 119, 3513–3526 (2006)
Y. Sakamoto, K. Hara, M. Kanai-Azuma, T. Matsui, Y. Miura, N. Tsunekawa, M. Kurohmaru, Y. Saijoh, P. Koopman, Y. Kanai, Redundant roles of Sox17 and Sox18 in early cardiovascular development of mouse embryos. Biochem Biophys Res Commun. 360, 539–544 (2007)
M. Downes, M. Francois, C. Ferguson, R.G. Parton, P. Koopman, Vascular defects in a mouse model of hypotrichosis-lymphedema-telangiectasia syndrome indicate a role for SOX18 in blood vessel maturation. Hum Mol Genet. 18, 2839–2850 (2009)
M. Francois, A. Caprini, B. Hosking, F. Orsenigo, D. Wilhelm, C. Browne, K. Paavonen, T. Karnezis, R. Shayan, M. Downes, T. Davidson, D. Tutt, K.S. Cheah, S.A. Stacker, G.E. Muscat, M.G. Achen, E. Dejana, P. Koopman, Sox18 induces development of the lymphatic vasculature in mice. Nature. 456, 643–647 (2008)
A. Irrthum, K. Devriendt, D. Chitayat, G. Matthijs, C. Glade, P.M. Steijlen, J.P. Fryns, M.A. Van Steensel, M. Vikkula, Mutations in the transcription factor gene SOX18 underlie recessive and dominant forms of hypotrichosis-lymphedema-telangiectasia. Am J Hum Genet. 72, 1470–1478 (2003)
R. Herpers, E. van de Kamp, H.J. Duckers, S. Schulte-Merker, Redundant roles for sox7 and sox18 in arteriovenous specification in zebrafish. Circ Res. 102, 12–15 (2008)
R.A. Mohammed, I.O. Ellis, A.M. Mahmmod, E.C. Hawkes, A.R. Green, E.A. Rakha, S.G. Martin, Lymphatic and blood vessels in basal and triple-negative breast cancers: characteristics and prognostic significance. Mod Pathol. 24, 774–785 (2011)
R.A. Mohammed, I.O. Ellis, S. Elsheikh, E.C. Paish, S.G. Martin, Lymphatic and angiogenic characteristics in breast cancer: morphometric analysis and prognostic implications. Breast Cancer Res Treat. 113, 261–273 (2009)
C.S. Williams, R.D. Leek, A.M. Robson, S. Banerji, R. Prevo, A.L. Harris, D.G. Jackson, Absence of lymphangiogenesis and intratumoural lymph vessels in human metastatic breast cancer. J Pathol. 200, 195–206 (2003)
I.A. Darby, T. Bisucci, S. Raghoenath, J. Olsson, G.E. Muscat, P. Koopman, Sox18 is transiently expressed during angiogenesis in granulation tissue of skin wounds with an identical expression pattern to Flk-1 mRNA. Lab Invest. 81, 937–943 (2001)
N. Young, C.N. Hahn, A. Poh, C. Dong, D. Wilhelm, J. Olsson, G.E. Muscat, P. Parsons, J.R. Gamble, P. Koopman, Effect of disrupted SOX18 transcription factor function on tumor growth, vascularization, and endothelial development. J Natl Cancer Inst. 98, 1060–1067 (2006)
T. Saitoh, M. Katoh, Expression of human SOX18 in normal tissues and tumors. Int J Mol Med. 10, 339–344 (2002)
B. W. Eom, M. J. Jo, M. C. Kook, K. W. Ryu, I. J. Choi, B. H. Nam, Y. W. Kim, J. H. Lee: The lymphangiogenic factor SOX 18: A key indicator to stage gastric tumor progression, Int J Cancer. (2011)
M. Garcia-Ramirez, J. Martinez-Gonzalez, J.O. Juan-Babot, C. Rodriguez, L. Badimon, Transcription factor SOX18 is expressed in human coronary atherosclerotic lesions and regulates DNA synthesis and vascular cell growth. Arterioscler Thromb Vasc Biol. 25, 2398–2403 (2005)
M. Lacroix, G. Leclercq, Relevance of breast cancer cell lines as models for breast tumours: an update. Breast Cancer Res Treat. 83, 249–289 (2004)
P. Dziegiel, T. Owczarek, E. Plazuk, A. Gomulkiewicz, M. Majchrzak, M. Podhorska-Okolow, K. Driouch, R. Lidereau, M. Ugorski, Ceramide galactosyltransferase (UGT8) is a molecular marker of breast cancer malignancy and lung metastases. Br J Cancer. 103, 524–531 (2010)
H.J. Bloom, W.W. Richardson, Histological grading and prognosis in breast cancer; a study of 1409 cases of which 359 have been followed for 15 years. Br J Cancer. 11, 359–377 (1957)
C.W. Elston, Grading of invasive carcinoma of the breast, in Diagnostic histopathology of the breast, ed. by D.L. Page (Churchill Livingstone, Edinburgh, 1987), pp. 300–311
B. Pula, A. Jethon, A. Piotrowska, A. Gomulkiewicz, T. Owczarek, J. Calik, A. Wojnar, W. Witkiewicz, J. Rys, M. Ugorski, P. Dziegiel, M. Podhorska-Okolow, Podoplanin expression by cancer-associated fibroblasts predicts poor outcome in invasive ductal breast carcinoma. Histopathology. 59, 1249–1260 (2011)
W. Remmele, H.E. Stegner, Recommendation for uniform definition of an immunoreactive score (IRS) for immunohistochemical estrogen receptor detection (ER-ICA) in breast cancer tissue. Pathologe. 8, 138–140 (1987)
P.B. Vermeulen, G. Gasparini, S.B. Fox, C. Colpaert, L.P. Marson, M. Gion, J.A. Belien, R.M. de Waal, E. Van Marck, E. Magnani, N. Weidner, A.L. Harris, L.Y. Dirix, Second international consensus on the methodology and criteria of evaluation of angiogenesis quantification in solid human tumours. Eur J Cancer. 38, 1564–1579 (2002)
A. Goldhirsch, J.N. Ingle, R.D. Gelber, A.S. Coates, B. Thurlimann, H.J. Senn, m. Panel: Thresholds for therapies: highlights of the St Gallen International Expert Consensus on the primary therapy of early breast cancer 2009. Ann Oncol. 20, 1319–1329 (2009)
R. Doroudi, M. Andersson, P.A. Svensson, M. Ekman, S. Jern, L. Karlsson, Methodological studies of multiple reference genes as endogenous controls in vascular gene expression studies. Endothelium. 12, 215–223 (2005)
T. Azhikina, A. Kozlova, T. Skvortsov, E. Sverdlov, Heterogeneity and degree of TIMP4, GATA4, SOX18, and EGFL7 gene promoter methylation in non-small cell lung cancer and surrounding tissues. Cancer Genet. 204, 492–500 (2011)
R. Dammann, M. Strunnikova, U. Schagdarsurengin, M. Rastetter, M. Papritz, U.E. Hattenhorst, H.S. Hofmann, R.E. Silber, S. Burdach, G. Hansen, CpG island methylation and expression of tumour-associated genes in lung carcinoma. Eur J Cancer. 41, 1223–1236 (2005)
J. Kao, K. Salari, M. Bocanegra, Y.L. Choi, L. Girard, J. Gandhi, K.A. Kwei, T. Hernandez-Boussard, P. Wang, A.F. Gazdar, J.D. Minna, J.R. Pollack, Molecular profiling of breast cancer cell lines defines relevant tumor models and provides a resource for cancer gene discovery. PLoS One. 4, e6146 (2009)
C.A. Hudis, L. Gianni, Triple-negative breast cancer: an unmet medical need. Oncologist. 16(Suppl 1), 1–11 (2011)
W. Takash, J. Canizares, N. Bonneaud, F. Poulat, M.G. Mattei, P. Jay, P. Berta, SOX7 transcription factor: sequence, chromosomal localisation, expression, transactivation and interference with Wnt signalling. Nucleic Acids Res. 29, 4274–4283 (2001)
D.Y. Fu, Z.M. Wang, C. Li, B.L. Wang, Z.Z. Shen, W. Huang, Z.M. Shao, Sox17, the canonical Wnt antagonist, is epigenetically inactivated by promoter methylation in human breast cancer. Breast Cancer Res Treat. 119, 601–612 (2010)
E. Mylona, A. Nomikos, P. Alexandrou, I. Giannopoulou, A. Keramopoulos, L. Nakopoulou, Lymphatic and blood vessel morphometry in invasive breast carcinomas: relation with proliferation and VEGF-C and -D proteins expression. Histol Histopathol. 22, 825–835 (2007)
W.W. Choi, M.M. Lewis, D. Lawson, Q. Yin-Goen, G.G. Birdsong, G.A. Cotsonis, C. Cohen, A.N. Young, Angiogenic and lymphangiogenic microvessel density in breast carcinoma: correlation with clinicopathologic parameters and VEGF-family gene expression. Mod Pathol. 18, 143–152 (2005)
D.W. van der Schaft, P. Pauwels, S. Hulsmans, M. Zimmermann, L.V. van de Poll-Franse, A.W. Griffioen, Absence of lymphangiogenesis in ductal breast cancer at the primary tumor site. Cancer Lett. 254, 128–136 (2007)
R. Prevo, S. Banerji, D.J. Ferguson, S. Clasper, D.G. Jackson, Mouse LYVE-1 is an endocytic receptor for hyaluronan in lymphatic endothelium. J Biol Chem. 276, 19420–19430 (2001)
S. Breiteneder-Geleff, A. Soleiman, H. Kowalski, R. Horvat, G. Amann, E. Kriehuber, K. Diem, W. Weninger, E. Tschachler, K. Alitalo, D. Kerjaschki, Angiosarcomas express mixed endothelial phenotypes of blood and lymphatic capillaries: podoplanin as a specific marker for lymphatic endothelium. Am J Pathol. 154, 385–394 (1999)
S. Hirakawa, L.F. Brown, S. Kodama, K. Paavonen, K. Alitalo, M. Detmar, VEGF-C-induced lymphangiogenesis in sentinel lymph nodes promotes tumor metastasis to distant sites. Blood. 109, 1010–1017 (2007)
S. Hirakawa, S. Kodama, R. Kunstfeld, K. Kajiya, L.F. Brown, M. Detmar, VEGF-A induces tumor and sentinel lymph node lymphangiogenesis and promotes lymphatic metastasis. J Exp Med. 201, 1089–1099 (2005)
N.E. Tobler, M. Detmar, Tumor and lymph node lymphangiogenesis–impact on cancer metastasis. J Leukoc Biol. 80, 691–696 (2006)
Y. Nakamura, H. Yasuoka, M. Tsujimoto, S. Imabun, M. Nakahara, K. Nakao, M. Nakamura, I. Mori, K. Kakudo, Lymph vessel density correlates with nodal status, VEGF-C expression, and prognosis in breast cancer. Breast Cancer Res Treat. 91, 125–132 (2005)
P. Bono, V.M. Wasenius, P. Heikkila, J. Lundin, D.G. Jackson, H. Joensuu, High LYVE-1-positive lymphatic vessel numbers are associated with poor outcome in breast cancer. Clin Cancer Res. 10, 7144–7149 (2004)
T. Duong, S. T. Proulx, P. Luciani, J. C. Leroux, M. Detmar, P. Koopman, M. Francois: Genetic ablation of SOX18 function suppresses tumor lymphangiogenesis and metastasis of melanoma in mice, Cancer Res. (2012)
Acknowledgments
The authors wish to thank Mrs. Teresa Klepuszewszka from the Department of Pathology, University of Environmental and Life Sciences, and Mrs. Magdalena Chmielewska, Mrs. Aleksandra Piotrowska, Ms. Aleksandra Jethon, Ms. Renata Brykner and Mrs. Lucja Cwynar-Zajac from the Department of Histology and Embryology, Wroclaw Medical University for their technical support. Ms. Anna Frydlewicz from the Regional Specialist Hospital, Research and Development Centre, Wroclaw is acknowledged for sample collection.
This publication was financially supported by the “Wrovasc—Integrated Cardiovascular Centre” project, co-financed by the European Regional Development Fund, within the Innovative Economy Operational Program, 2007–2013.
Conflict of interest
The authors have no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pula, B., Olbromski, M., Wojnar, A. et al. Impact of SOX18 expression in cancer cells and vessels on the outcome of invasive ductal breast carcinoma. Cell Oncol. 36, 469–483 (2013). https://doi.org/10.1007/s13402-013-0151-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13402-013-0151-7