Abstract
Background
The signal transducer and activator of transcription 6 (Stat6), a member of the family of DNA-binding proteins, has been identified as a critical cell differentiation modulator in breast cancer cells. As of yet, the mechanisms underlying this function have remained largely unknown. To further elucidate the role of Stat6 in breast cancer development, we investigated the consequences of exogenous Stat6 expression.
Methods
Proliferation assays and flow cytometry assays were conducted to evaluate the putative role of Stat6 on cell proliferation. To this end, we produced synchronized cells after a double thymidine block, as confirmed by FACS analysis. mRNA levels of Stat6 were measured by RNase protection analysis. To confirm the interaction among proteins, we employed GST pull-down assays and immunoprecipitation assays. Luciferase assays and ChIP assays were used to assess the transcriptional activity.
Results
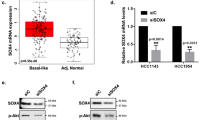
Compared to control breast cancer cells, we found that exogenous Stat6 expression plays a critical role in controlling cell proliferation. Also in different breast tumor cell lines, endogenous Stat6 expression was found to be positively related to a lower proliferation rate. Interestingly, in human breast cancer cells Stat6 functions in G1/S cell cycle progression, and the growth-inhibitory effect of Stat6 was shown to be mediated by induction of the G1 cyclin-dependent kinase inhibitors p21Cip1/WAF1 (p21) and p27Kip1 (p27). Simultaneously, G1-related cyclin/cyclin-dependent kinase activities and pRB phosphorylation were markedly reduced, and cell cycle progression was blocked in the G1 phase. Stat6 knockdown resulted in enhanced cell proliferation and a decrease in p21 and p27 mRNA levels in the steroid-responsive and non-responsive T-47D and MDA-MB-231 cell lines, respectively. In addition, the stimulatory effect of Stat6 on p21 and p27 gene transcription was found to be associated with interaction of Stat6 with the transcription factor Sp1 at the proximal Sp1-binding sites in their respective promoters.
Conclusions
Together, these results identify Stat6 as an important cell differentiation regulatory protein functioning, at least in part, by interacting with Sp1 to activate the p21 and p27 gene promoters in breast cancer cells.







Similar content being viewed by others
References
J. Hasty, D. McMillen, J.J. Collins, Engineered gene circuits. Nature 420, 224–230 (2002)
Q. Wang, Y. Zhou, X. Wang, B.M. Evers, p27Kip1 nuclear localization and cyclin-dependent kinase inhibitory activity are regulated by glycogen synthase kinase-3 in human colon cancer cells. Cell Death Differ 15, 908–919 (2008)
I. Soerjomataram, M.W. Louwman, J.G. Ribot, J.A. Roukema, J.W. Coebergh, An overview of prognostic factors for long-term survivors of breast cancer. Breast Cancer Res Treat 107, 309–330 (2008)
C.E. Caldon, C.S. Lee, R.L. Sutherland, E.A. Musgrove, Wilms' tumor protein 1: an early target of progestin regulation in T-47D breast cancer cells that modulates proliferation and differentiation. Oncogene 27, 126–138 (2008)
S. Sheahan, C.O. Bellamy, D.R. Dunbar, D.J. Harrison, S. Prost, Deficiency of G1 regulators P53, P21Cip1 and/or pRb decreases hepatocyte sensitivity to TGFbeta cell cycle arrest. BMC Cancer 7, 215 (2007)
L.L. Seville, N. Shah, A.D. Westwell, W.C. Chan, Modulation of pRB/E2F functions in the regulation of cell cycle and in cancer. Curr Cancer Drug Targets 5, 159–170 (2005)
S. Paternot, T. Arsenijevic, K. Coulonval, L. Bockstaele, J.E. Dumont, P.P. Roger, Distinct specificities of pRb phosphorylation by CDK4 activated by cyclin D1 or cyclin D3: differential involvement in the distinct mitogenic modes of thyroid epithelial cells. Cell Cycle 5, 61–70 (2006)
S. Mani, C. Wang, K. Wu, R. Francis, R. Pestell, Cyclin-dependent kinase inhibitors: novel anticancer agents. Expert Opin Investig Drugs 9, 1849–1870 (2000)
L.E. Horton, D.J. Templeton, The cyclin box and C-terminus of cyclins A and E specify CDK activation and substrate specificity. Oncogene 14, 491–498 (1997)
A. Contreras, T.K. Hale, D.L. Stenoien, J.M. Rosen, M.A. Mancini, R.E. Herrera, The dynamic mobility of histone H1 is regulated by cyclin/CDK phosphorylation. Mol. Cell. Biol. 23, 8626–8636 (2003)
R.E. Herrera, F. Chen, R.A. Weinberg, Increased histone H1 phosphorylation and relaxed chromatin structure in Rb-deficient fibroblasts. Proc. Natl. Acad. Sci. U. S. A. 93, 11510–11515 (1996)
D. Donjerkovic, D.W. Scott, Regulation of the G1 phase of the mammalian cell cycle. Cell Res 10, 1–16 (2000)
G.W. Woodfield, A.D. Horan, Y. Chen, R.J. Weigel, TFAP2C controls hormone response in breast cancer cells through multiple pathways of estrogen signaling. Cancer Res. 67, 8439–8443 (2007)
A. Fritah, C. Saucier, J. Mester, G. Redeuilh, M. Sabbah, p21WAF1/CIP1 selectively controls the transcriptional activity of estrogen receptor alpha. Mol. Cell. Biol. 25, 2419–2430 (2005)
Y. Ye, H. Yang, H.B. Grossman, C. Dinney, X. Wu, J. Gu, Genetic variants in cell cycle control pathway confer susceptibility to bladder cancer. Cancer 112, 2467–2474 (2008)
P.B. Burton, C.J. Anderson, C.M. Corbishly, Caspase 3 and p27 as predictors of invasive bladder cancer. N. Engl. J. Med. 343, 1418–1420 (2000)
S. Goenka, J. Youn, L.M. Dzurek, U. Schindler, L.-Y. Yu-Lee, M. Boothby, Paired Stat6 C-terminal transcription activation domains required both for inhibition of an IFN-responsive promoter and trans-activation. J. Immunol. 163, 4663–4672 (1999)
J.L. Gooch, B. Christy, D. Yee, STAT6 mediates interleukin-4 growth inhibition in human breast cancer cells. Neoplasia 4, 324–331 (2002)
B. Borud, T. Hoang, M. Bakke, A.L. Jacob, J. Lund, G. Mellgren, The nuclear receptor coactivators p300/CBP/cointegrator-associated protein (p/CIP) and transcription intermediary factor 2 (TIF2) differentially regulate PKA-stimulated transcriptional activity of steroidogenic factor 1. Mol. Endocrinol. 16, 757–773 (2002)
N.A. Hanley, S.G. Ball, M. Clement-Jones, D.M. Hagan, T. Strachan, S. Lindsay, S. Robson, H. Ostrer, K.L. Parker, D.I. Wilson, Expression of steroidogenic factor 1 and Wilms' tumour 1 during early human gonadal development and sex determination. Mech. Dev. 87, 175–180 (1999)
S. Minami, N. Ohtani-Fujita, E. Igata, T. Tamaki, T. Sakai, Molecular cloning and characterization of the human p27Kip1 gene promoter. FEBS Lett. 411, 1–6 (1997)
M. Wei, B. Liu, L. Su, J. Li, J. Zhang, Y. Yu, M. Yan, Z. Yang, X. Chen, J. Liu, X. Lv, H. Nie, Q. Zhang, Z. Zheng, B. Yu, J. Ji, Z. Zhu, Q. Gu, A novel plant homeodomain finger 10-mediated antiapoptotic mechanism involving repression of caspase-3 in gastric cancer cells. Mol. Cancer Ther. 9, 1764–1774 (2010)
Y. Qu, J.F. Li, Q. Cai, Y.W. Wang, Q.L. Gu, Z.G. Zhu, B.Y. Liu, Over-expression of FRZB in gastric cancer cell suppresses proliferation and induces differentiation. J. Cancer Res. Clin. Oncol. 134, 353–364 (2008)
D. Wei, M. Kanai, Z. Jia, X. Le, K. Xie, Kruppel-like factor 4 induces p27Kip1 expression in and suppresses the growth and metastasis of human pancreatic cancer cells. Cancer Res. 68, 4631–4639 (2008)
E. di Martino, C.G. L'Hote, W. Kennedy, D.C. Tomlinson, M.A. Knowles, Mutant fibroblast growth factor receptor 3 induces intracellular signaling and cellular transformation in a cell type- and mutation-specific manner. Oncogene 28, 4306–4316 (2009)
A. Freund, C. Chauveau, J.P. Brouillet, A. Lucas, M. Lacroix, A. Licznar, F. Vignon, G. Lazennec, IL-8 expression and its possible relationship with estrogen-receptor-negative status of breast cancer cells. Oncogene 22, 256–265 (2003)
M. Wei, Z. Wang, H. Yao, Z. Yang, Q. Zhang, B. Liu, Y. Yu, L. Su, Z. Zhu, Q. Gu, P27(Kip1), regulated by glycogen synthase kinase-3beta, results in HMBA-induced differentiation of human gastric cancer cells. BMC Cancer 11, 109–121 (2011)
M. Belyavskyi, S.C. Braunagel, M.D. Summers, The structural protein ODV-EC27 of Autographa californica nucleopolyhedrovirus is a multifunctional viral cyclin. Proc. Natl. Acad. Sci. U. S. A. 95, 11205–11210 (1998)
P.C. Hsieh, J.C. Chang, W.T. Sun, S.C. Hsieh, M.C. Wang, F.F. Wang, p53 downstream target DDA3 is a novel microtubule-associated protein that interacts with end-binding protein EB3 and activates beta-catenin pathway. Oncogene 26, 4928–4940 (2007)
M.L. Whitfield, G. Sherlock, A.J. Saldanha, J.I. Murray, C.A. Ball, K.E. Alexander, J.C. Matese, C.M. Perou, M.M. Hurt, P.O. Brown, D. Botstein, Identification of genes periodically expressed in the human cell cycle and their expression in tumors. Mol. Biol. Cell. 13, 1977–2000 (2002)
E.A. Musgrove, B. Sarcevic, R.L. Sutherland, Inducible expression of cyclin D1 in T-47D human breast cancer cells is sufficient for Cdk2 activation and pRB hyperphosphorylation. J. Cell. Biochem. 60, 363–378 (1996)
M. Kitagawa, H. Higashi, I.S. Takahashi, T. Okabe, H. Ogino, Y. Taya, S. Hishimura, A. Okuyama, A cyclin-dependent kinase inhibitor, butyrolactone I, inhibits phosphorylation of RB protein and cell cycle progression. Oncogene 9, 2549–2557 (1994)
E.S. Child, D.J. Mann, Novel properties of the cyclin encoded by Human Herpesvirus 8 that facilitate exit from quiescence. Oncogene 20, 3311–3322 (2001)
L. Arpa, A.F. Valledor, J. Lloberas, A. Celada, IL-4 blocks M-CSF-dependent macrophage proliferation by inducing p21Waf1 in a STAT6-dependent way. Eur. J. Immunol. 39, 514–526 (2009)
B. Cen, A. Deguchi, I.B. Weinstein, Activation of protein kinase G Increases the expression of p21CIP1, p27KIP1, and histidine triad protein 1 through Sp1. Cancer Res. 68, 5355–5362 (2008)
S. Lu, G. Jenster, D.E. Epner, Androgen induction of cyclin-dependent kinase inhibitor p21 gene: role of androgen receptor and transcription factor Sp1 complex. Mol. Endocrinol. 14, 753–760 (2000)
T.H. Lee, H.C. Chang, L.Y. Chuang, W.C. Hung, Involvement of PKA and Sp1 in the induction of p27(Kip1) by tamoxifen. Biochem. Pharmacol. 66, 371–377 (2003)
S. Ryhanen, T. Jaaskelainen, A. Mahonen, P.H. Maenpaa, Inhibition of MG-63 cell cycle progression by synthetic vitamin D3 analogs mediated by p27, Cdk2, cyclin E, and the retinoblastoma protein. Biochem. Pharmacol. 66, 495–504 (2003)
K. Okamoto, Y. Okamoto, T. Kawakubo, J. Iwata, Y. Yasuda, T. Tsukuba, K. Yamamoto, Role of the transcription factor Sp1 in regulating the expression of the murine cathepsin E gene. J. Biochem. 151, 263–272 (2012)
A.M. Stutz, L.A. Pickart, A. Trifilieff, T. Baumruker, E. Prieschl-Strassmayr, M. Woisetschlager, The Th2 cell cytokines IL-4 and IL-13 regulate found in inflammatory zone 1/resistin-like molecule alpha gene expression by a STAT6 and CCAAT/enhancer-binding protein-dependent mechanism. J. Immunol. 170, 1789–1796 (2003)
V.T. Nguyen, E.N. Benveniste, Involvement of STAT-1 and ets family members in interferon-gamma induction of CD40 transcription in microglia/macrophages. J. Biol. Chem. 275, 23674–23684 (2000)
J. Fukuzawa, G.W. Booz, R.A. Hunt, N. Shimizu, V. Karoor, K.M. Baker, D.E. Dostal, Cardiotrophin-1 increases angiotensinogen mRNA in rat cardiac myocytes through STAT3: an autocrine loop for hypertrophy. Hypertension 35, 1191–1196 (2000)
K. Barbulescu, C. Becker, J.F. Schlaak, E. Schmitt, K.H. Meyer zum Buschenfelde, M.F. Neurath, IL-12 and IL-18 differentially regulate the transcriptional activity of the human IFN-gamma promoter in primary CD4+ T lymphocytes. J. Immunol. 160, 3642–3647 (1998)
H.J. Park, Y.S. Choi, C.E. Lee, Identification and activation mechanism of the interleukin-4-induced nuclear factor binding to the CD23(b) promoter in human B lymphocytes. Mol Cells 7, 755–761 (1997)
S. Asonuma, A. Imatani, N. Asano, T. Oikawa, H. Konishi, K. Iijima, T. Koike, S. Ohara, T. Shimosegawa, Helicobacter pylori induces gastric mucosal intestinal metaplasia through the inhibition of interleukin-4-mediated HMG box protein Sox2 expression. Am. J. Physiol. Gastrointest. Liver Physiol. 297, G312–322 (2009)
M. Zhang, Y. Zhou, C. Xie, F. Zhou, Y. Chen, G. Han, W.J. Zhang, STAT6 specific shRNA inhibits proliferation and induces apoptosis in colon cancer HT-29 cells. Cancer Lett. 243, 38–46 (2006)
X.W. Zhang, Y.P. Sheng, Q. Li, W. Qin, Y.W. Lu, Y.F. Cheng, B.Y. Liu, F.C. Zhang, J. Li, G.P. Dimri, W.J. Guo, BMI1 and Mel-18 oppositely regulate carcinogenesis and progression of gastric cancer. Mol. Cancer 9, 40–52 (2010)
J.P. Wiebe, M.J. Lewis, Activity and expression of progesterone metabolizing 5alpha-reductase, 20alpha-hydroxysteroid oxidoreductase and 3alpha(beta)-hydroxysteroid oxidoreductases in tumorigenic (MCF-7, MDA-MB-231, T-47D) and nontumorigenic (MCF-10A) human breast cancer cells. BMC Cancer 3, 9 (2003)
E.A. Musgrove, L.J. Hunter, C.S. Lee, A. Swarbrick, R. Hui, R.L. Sutherland, Cyclin D1 overexpression induces progestin resistance in T-47D breast cancer cells despite p27(Kip1) association with cyclin E-Cdk2. J. Biol. Chem. 276, 47675–47683 (2001)
R. Hui, G.L. Finney, J.S. Carroll, C.S. Lee, E.A. Musgrove, R.L. Sutherland, Constitutive overexpression of cyclin D1 but not cyclin E confers acute resistance to antiestrogens in T-47D breast cancer cells. Cancer Res. 62, 6916–6923 (2002)
C.R. Yu, R.M. Mahdi, S. Ebong, B.P. Vistica, J. Chen, Y. Guo, I. Gery, C.E. Egwuagu, Cell proliferation and STAT6 pathways are negatively regulated in T cells by STAT1 and suppressors of cytokine signaling. J. Immunol. 173, 737–746 (2004)
W. Reed, V.A. Flørenes, R. Holm, E. Hannisdal, J.M. Nesland, Elevated levels of p27, p21 and cyclin D1 correlate with positive oestrogen and progesterone receptor status in node-negative breast carcinoma patients. 116–124 (1999)
S. Vanzulli, A. Efeyan, F. Benavides, L.A. Helguero, G. Peters, J. Shen, C.J. Conti, C. Lanari, A. Molinolo, p21, p27 and p53 in estrogen and antiprogestin-induced tumor regression of experimental mouse mammary ductal carcinomas. Carcinogenesis 23, 749–758 (2002)
S.D. Groshong, G.I. Owen, B. Grimison, I.E. Schauer, M.C. Todd, T.A. Langan, R.A. Sclafani, C.A. Lange, K.B. Horwitz, Biphasic regulation of breast cancer cell growth by progesterone: role of the cyclin-dependent kinase inhibitors, p21 and p27Kip1. Mol. Endocrinol. 11, 1593–1607 (1997)
M.H. Kaplan, C. Daniel, U. Schindler, M.J. Grusby, Stat proteins control lymphocyte proliferation by regulating p27Kip1 expression. Mol. Cell. Biol. 18, 1996–2003 (1998)
J. Liu, M.L. Estes, J.A. Drazba, H. Liu, R. Prayson, S. Kondo, B.S. Jacobs, G.H. Barnett, B.P. Barna, Anti-sense oligonucleotide of p21(waf1/cip1) prevents interleukin 4-mediated elevation of p27(kip1) in low grade astrocytoma cells. Oncogene 19, 661–669 (2000)
B.K. Patel, C.L. Keck, R.S. O'Leary, N.C. Popescu, W.J. LaRochelle, Localization of the human stat6 gene to chromosome 12q13.3-q14.1, a region implicated in multiple solid tumors. Genomics 52, 192–200 (1998)
A. Parcellier, M. Brunet, E. Schmitt, E. Col, C. Didelot, A. Hammann, K. Nakayama, K.I. Nakayama, S. Khochbin, E. Solary, C. Garrido, HSP27 favors ubiquitination and proteasomal degradation of p27Kip1 and helps S-phase re-entry in stressed cells. FASEB J. 20, 1179–1181 (2006)
Q. Zhang, L. Tian, A. Mansouri, A.L. Korapati, T.J. Johnson, F.X. Claret, Inducible expression of a degradation-resistant form of p27Kip1 causes growth arrest and apoptosis in breast cancer cells. FEBS Lett. 579, 3932–3940 (2005)
X. Wang, X. Liu, J. Fang, Y. Lu, J. He, X. Yao, Z. Yao, J. Yang, Coactivator P100 protein enhances STAT6-dependent transcriptional activation but has no effect on STAT1-mediated gene transcription. Anat Rec (Hoboken) 293, 1010–1016 (2010)
Y.L. Chai, J. Cui, N. Shao, E. Shyam, P. Reddy, V.N. Rao, The second BRCT domain of BRCA1 proteins interacts with p53 and stimulates transcription from the p21WAF1/CIP1 promoter. Oncogene 18, 263–268 (1999)
J. Yang, S. Aittomaki, M. Pesu, K. Carter, J. Saarinen, N. Kalkkinen, E. Kieff, O. Silvennoinen, Identification of p100 as a coactivator for STAT6 that bridges STAT6 with RNA polymerase II. EMBO J. 21, 4950–4958 (2002)
Z. Ai, W. Lu, S. Ton, H. Liu, T. Sou, Z. Shen, X. Qin, Arsenic trioxide-mediated growth inhibition in gallbladder carcinoma cells via down-regulation of Cyclin D1 transcription mediated by Sp1 transcription factor. Biochem. Biophys. Res. Commun. 360, 684–689 (2007)
Y. Chai, H.J. Lee, A.A. Shaik, K. Nkhata, C. Xing, J. Zhang, S.J. Jeong, S.H. Kim, J. Lu, Penta-O-galloyl-beta-D-glucose induces G1 arrest and DNA replicative S-phase arrest independently of cyclin-dependent kinase inhibitor 1A, cyclin-dependent kinase inhibitor 1B and P53 in human breast cancer cells and is orally active against triple negative xenograft growth. Breast Cancer Res. 12, R67 (2012)
H.B. Nair, N.B. Kirma, M. Ganapathy, R.K. Vadlamudi, R.R. Tekmal, Estrogen receptor-beta activation in combination with letrozole blocks the growth of breast cancer tumors resistant to letrozole therapy. Steroids 76, 792–796 (2011)
A. Shatnawi, T. Tran, M. Ratnam, R5020 and RU486 act as progesterone receptor agonists to enhance Sp1/Sp4-dependent gene transcription by an indirect mechanism. Mol. Endocrinol. 21, 635–650 (2007)
M. Wei, L. Zhu, Y. Li, W. Chen, B. Han, Z. Wang, J. He, H. Yao, Z. Yang, Q. Zhang, B. Liu, Q. Gu, Z. Zhu, K. Shen, Knocking down cyclin D1b inhibits breast cancer cell growth and suppresses tumor development in a breast cancer model. Cancer Sci. 102, 1537–1544 (2011)
N. Heldring, G.D. Isaacs, A.G. Diehl, M. Sun, E. Cheung, J.A. Ranish, W.L. Kraus, Multiple sequence-specific DNA-binding proteins mediate estrogen receptor signaling through a tethering pathway. Mol. Endocrinol. 25, 564–574 (2011)
W.J. Zhang, B.H. Li, X.Z. Yang, P.D. Li, Q. Yuan, X.H. Liu, S.B. Xu, Y. Zhang, J. Yuan, G.S. Gerhard, K.K. Masker, C. Dong, W.A. Koltun, M.J. Chorney, IL-4-induced Stat6 activities affect apoptosis and gene expression in breast cancer cells. Cytokine 42, 39–47 (2008)
C. McDonald, N.C. Reich, Cooperation of the transcriptional coactivators CBP and p300 with Stat6. J. Interferon Cytokine Res. 19, 711–722 (1999)
Acknowledgements
This study was supported in part by grant funding of NSFC (Natural Science Foundation of China, 81101847), Doctoral Fund of Ministry of Education of China (20110073120089), project supported by the Shanghai Committee of Science and Technology, China (124119a4801) and NUST Research Funding (NO.2010ZYTS068).
Competing interests
All authors of this manuscript declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
SupplementaryFigure 1
The qualifications of the intensity of pRB in Fig. 3b (JPEG 2 kb)
SupplementaryFigure 2
The qualifications of P16 of Fig. 4a (JPEG 20 kb)
SupplementaryFigure 3
The qualifying of the molecular involved in ChIP assays in Fig. 5d (JPEG 10 kb)
SupplementaryFigure 4
Similar results of ChIP assays have been derived from human breast cancer cell line, MCF-7. (JPEG 8 kb)
SupplementaryFigure 5
Studies on the putative Stat6 binding site (TTC(N)3GAA) by using luciferase plasmids carrying mutants for this site showed that none obvious reduction of luciferase activity were detected in transfectants of Stat6 mutants compared with control. Stat6-w, wild type of Stat6. Stat6-M, Mutant of Stat6. (JPEG 117 kb)
SupplementaryFigure 6
A. Full-length Sp1 and Sp1 fragments fused to GST. Schematic diagram of Sp1 and Sp1 deletion mutants used in the GST pull-down assay is shown in the top panel. The letters A to D indicate different domains of the Sp1 protein. Three zinc finger DNA binding motifs of Sp1 presented in the C-terminal region are shown. The numbers before the names of each fragment indicate the amino acids of Sp1 linked to the GST protein. The pulldown assay in the center panel and the Coomassie-stained gel of the GST fusion protein inputs in the bottom panel. B. The structural domains of Stat6 and the Stat6 mutants used in this work are diagrammed in the top panel. Domains indicated include the transcriptional activation domain (TAD), DNA binding domain (DBD), Src homology 2 (SH2). The numbers before the names of each fragment indicate the amino acids of Stat6 linked to the GST protein. The pulldown assay in the center panel and the Coomassie-stained gel of the GST fusion protein inputs in the bottom panel. (JPEG 984 kb)
ESM 1
(DOC 36 kb)
Rights and permissions
About this article
Cite this article
Wei, M., Liu, B., Gu, Q. et al. Stat6 cooperates with Sp1 in controlling breast cancer cell proliferation by modulating the expression of p21Cip1/WAF1 and p27Kip1 . Cell Oncol. 36, 79–93 (2013). https://doi.org/10.1007/s13402-012-0115-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13402-012-0115-3