Abstract
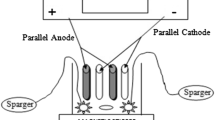
In this study, efficiency and cost of the Fenton, electro-Fenton, and biosorption processes for treatment of ariazol brown GR and indigo carmine dye mixture were compared. The Box-Behnken experimental design and response surface methodology (RSM) were used to optimize the process variables such as pH, [Fe2+], [H2O2], voltage, biosorbent dose, and time. Finally, the costs of 1 m3 wastewater treatment were calculated in optimal conditions. The operating costs for classical Fenton, electro-Fenton, and biosorption processes were 0.92, 0.48, and 0.13 US$ per 1 m3 wastewater, respectively. The efficiency of the Fenton and electro-Fenton processes for dye removal was similar, but the cost of the electro-Fenton method was half of the Fenton. Furthermore, the electro-Fenton was nine times faster than the Fenton. Efficiency of electro-Fenton was better than the biosorption process even if it is repeated four times with fresh biosorbent (total cost is the same as electro-Fenton). As a general conclusion, the electro-Fenton process is an appropriate choice based on speed and cost in wastewater treatment with respect to Fenton and biosorption processes.




Similar content being viewed by others
Data availability
Not applicable.
References
Forgacs E, Cserhati T, Oros G (2004) Removal of synthetic dyes from wastewaters: a review. Environ Int 30:953–971
Elgarahy AM, Elwakeel KZ, Mohammad SH, Elshoubaky GA (2021) A critical review of biosorption of dyes, heavy metals and metalloids from wastewater as an efficient and green process. Cleaner Eng Technol 4:100209
Tabaraki R, Heidarizadi E (2018) Simultaneous biosorption of arsenic (III) and arsenic (V): application of multiple response optimizations. Ecotox Environ Safe 166:35–41
Tabaraki R, Nateghi A (2014) Multimetal biosorption modeling of Zn2+, Cu2+and Ni2+ by Sargassum ilicifolium. Ecol Eng 71:197–205
Tabaraki R, Sadeghinejad N (2018) Comparison of magnetic Fe3O4/chitosan and arginine-modified magnetic Fe3O4/chitosan nanoparticles in simultaneous multidye removal: Experimental design and multicomponent analysis. Int J Biol Macromol 120:2313–2323
Tan KB, Vakili M, Amini Horri B, Poh PE, Abdullah AZ, Salamatinia B (2015) Adsorption of dyes by nanomaterials: recent developments and adsorption mechanisms. Sep Purif Technol 150:229–242
Bhatia D, Sharma NR, Singh J, Kanwar RS (2017) Biological methods for textile dye removal from wastewater: A review. Crit Rev Env Sci Tec 47:1836–1876
Ribeiro JP, Nunes MI (2021) Recent trends and developments in Fenton processes for industrial wastewater treatment – a critical review. Env Res 197:110957
Jain B, Singh AK, Kim H, Lichtfouse E, Sharma VK (2018) Treatment of organic pollutants by homogeneous and heterogeneous Fenton reaction processes. Env chem lett 16:947–967
Kuo WG (1992) Decolorizing dye wastewater with Fenton’s reagent. Water Res 26:881–886
Casado J (2019) Towards industrial implementation of electro-Fenton and derived technologies for wastewater treatment: a review. J Env Chem Eng 7:102823
Brillas E, Martinez-Huitle CA (2015) Decontamination of wastewaters containing synthetic organic dyes by electrochemical methods. An updated review. Appl Catal B Environ 166–167:603–643
Malakootian M, Moridi A (2017) Efficiency of electro-Fenton process in removing acid red 18 dye from aqueous solutions. Process Saf Environ 111:138–147
Fomina M, Gadd GM (2014) Biosorption: current perspectives on concept, definition and application. Bioresource Technol 160:3–14
Yaseen DA, Scholz M (2019) Textile dye wastewater characteristics and constituents of synthetic effluents: a critical review. Int J Environ Sci Te 16:1193–1226
Baird RB, Eaton AD, Rice EW (eds) (2017) Standard methods for the examination of water and wastewater, 23rd edn. American Public Health Association, American Water Works Association, Water Environment Federation Washington, DC
Zhou M, Yu Q, Lei L, Barton G (2007) electro-Fenton method for the removal of methyl red in an efficient electrochemical system. Sep Purif Technol 57:380–387
Nawaz S, Siddique M, Khan R, Riaz N, Waheed U, Shahzadi I, Ali A (2022) Ultrasound-assisted hydrogen peroxide and iron sulfate mediated Fenton process as an efficient advanced oxidation process for the removal of congo red dye. Pol J Environ Stud 31:1–13
Gonçalves AC, Braccini AL, Schwantes D, Campagnolo MA, Da Paz SA, Manfrin J, Conradi E, Zimmermann J (2020) Adsorbents developed from residual biomass of canola grains for the removal of lead from water. Desalin Water Treat 197:261–279
Tabaraki R, Heidarizadi E (2017) Simultaneous multidye biosorption by chemically modified Sargassum glaucescens: Doehlert optimization and kinetic, equilibrium, and thermodynamic study in ternary system. Sep Sci Technol 52:583–595
Rodrigues CS, Madeira LM, Boaventura RA (2014) Synthetic textile dyeing wastewater treatment by integration of advanced oxidation and biological processes–pserformance analysis with costs reduction. J Environ Chem Eng 2:1027–1039
Jaafarzadeh N, Takdastan A, Jorfi S, Ghanbari F, Ahmadi M, Barzegar G (2018) The performance study on ultrasonic/Fe3O4/H2O2 for degradation of azo dye and real textile wastewater treatment. J Mol Liq 256:462–470
Author information
Authors and Affiliations
Contributions
Reza Tabaraki: supervision, project administration, resources, writing – review and editing. Saeed Zadkhast carried out the experiment and wrote the original draft. Amene Najafi carried out the experiment and wrote the original draft. Vahideh Rezaei Moghaddam: carried out the experiment.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tabaraki, R., Zadkhast, S., Najafi, A. et al. Performance and cost analysis of dye wastewater treatment by Fenton, electro-Fenton, and biosorption: Box-Behnken experimental design and response surface methodology. Biomass Conv. Bioref. 13, 13527–13537 (2023). https://doi.org/10.1007/s13399-022-02809-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-022-02809-2