Abstracts
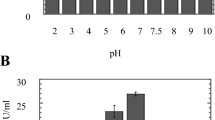
The strain Aspergillus fumigatus TKF1 (Genbank-JQ268555), isolated from soil of poultry waste site, produced extracellular keratinase in basal medium [0.05 % K2HPO4, 0.025 % MgSO4, 0.02 % CaCO3, 0.5 % NaCl and 0.015 % FeSO4 (w/v)] at 30 °C in pH 6 with 0.5 % supplementation of raw chicken feather after 84 h. Further statistical modeling (response surface methodology) was applied to optimized condition for keratinase production was followed by 84.33 h fermentation time at 30.2 °C and 0.74 % (w/v) feather supplement with initial pH 6.4. The response surface data and parametric optimization of the fermentation medium (one variable at a time) both condition satisfactorily gave 2.5-fold higher production than its unoptimized state. This enzyme preparation could disintegrate whole chicken feathers and reduced of disulphide bridges were also detected, increasing with incubation time. The purified keratinase was 24.3-kDa molecular weight, monomer in nature, and exhibited a significant stability and compatibility with temperature and pH. Feather degradation lead to an increase in oligopeptides and free amino acids such as cystein, therionine, phenylalanine, leucine, valine, and isoleucine. Moreover, methionine and phenylalanine were also produced as microbial metabolites. The importance of these finding is appropriate for future applications in agricultural and pharmaceutical industries.








Similar content being viewed by others
References
Moran ET, Summers JD, Slinger SJ (1966) Keratin as a source of protein for the growing chick. Amino acid imbalance as the cause for inferior performance of feather meal and the implication of disulfide bonding in raw feathers as the reason for poor digestibility. Poult Sci 45:1257–1266
Marcondes NR, Taira CL, Vandresen DC, Svidzinski TIE, Kadowaki MK, Peralta RM (2008) New feather-degrading filamentous fungi. Microbiol Ecol 56:13–13
Ignatova Z, Gousterova A, Spassov G, Nedkov P (1999) Isolation and partial characterization of extracellular keratinase from a wool degrading thermophilic actinomycete strain Thermoactinomyces candidus. Can J Microbiol 45:217–222
Bressolier P, Letourneau F, Urdaci M, Verneuil B (1999) Purification and characterization of a keratinolytic serine proteinase from Streptomyces albidoflavus. Appl Environ Microbiol 65:2570–2576
Santos RMD, Firmino AAP, Sá CM, Felix CR (1996) Keratinolytic activity of Aspergillus fumigatus Fresenius. Curr Microbiol 33:364–370
Friedrich AB, Antranikian G (1996) Keratin degradation by Fervidobacterium pennavorans, a novel thermophilic anaerobic species of the order Thermatogales. Appl Environ Microbiol 61:3705–3710
Gushterova A, Vasileva-Tonkova E, Dimova E, Nedkov P, Haertlé T (2005) Keratinase production by newly isolated Antarctic actinomycete strains. World J Microbiol Biotechnol 21:831–834
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL_X Windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882
Paul T, Halder SK, Das A, Bera S, Maity C, Mandal A, Das PS, Mohapatra PKD, Pati BR, Mondal KC (2012) Exploitation of chicken feather waste as a plant growth promoting agent using keratinase producing novel isolate Paenibacillus woosongensis TKB2. Biocat Argricul Biotechnol 2:50–57
Paul T, Das A, Mandal A, Jana A, Maity C, Adak A, Halder SK, DasMohapatra PK, Pati BR, Mondal KC (2013) Effective dehairing properties of keratinase from Paenibacillus woosongensis TKB2 obtained under solid state fermentation. Waste Biomass Valor. doi:10.1007/s12649-013-9217-z
Bockle B, Galunski B, Müller R (1995) Characterization of a keratinolytic serine protease from Streptomyces pactum DSM40530. Appl Environ Microbiol 61:3705–3710
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Rai SK, Konwarh R, Mukherjee AK (2009) Purification, characterization and biotechnological application of an alkaline β-keratinase produced by Bacillus subtilis RM-01 in solid-state fermentation using chicken feather as substrate. Biochem Eng J 45:218–225
Gerard A, Favre C, Garcon F, Nemorin JG, Duplay P, Pastor S, Collette Y, Olive D, Nunes JA (2004) Functional interaction of RasGAP-binding proteins Dok-1 and Dok-2 with the Tec protein tyrosine kinase. Oncogene 23:1594–1598
Sullivan MX, Hess WC, Howard HW (1942) The quantitative estimation of both cystine and cysteine in mixture. J Biol Chem 145:621–624
Zhang B, Sun Z, Jiang D, Niu T (2009) Isolation and purification of alkaline keratinase from Bacillus sp. 50–3. Afr J Biotechnol 8:2598–2603
Laemmli UK (1970) Cleavage of structure protein during the assembly of the head of bacteriophage T4. Nature 227:680–685
Heussen C, Dowdle EB (1980) Electrophoresis analysis of plasminogen activators in polyacrylamine gels containing sodium dodecyl sulphate and copolymerized substrates. Anal Biochem 102:196–202
Pouvreau A (1991) Morphology and histology of tarsal glands in bumble bees of the genera Bombus, Pyrobombus, and Megabombus. Can J Zool 69:866–872
Kathiresan K, Manivannan S (2007) Production of alkaline protease by Streptomyces sp. isolated from coastal mangrove sediment. Res J Environ Sci 1:173–178
Nilegaonkar SS, Zambare VP, Kanekar PP, Dhakephalkar PK, Sarnaik SS (2007) Production and partial characterization of dehairing protease from Bacillus cereus MCM B-326. Bioresour Technol 98:1238–1245. doi:10.1016/j.biortech.2006.05.003#_blank
Cheng SW, Hu HM, Shen SW, Takagi H, Asano M, Tsai YC (1995) Production and characterization of keratinase of a feather-degrading Bacillus licheniformis PWD-1. Biosci Biotechnol Biochem 59:2239–2243
Nam GW, Lee DW, Lee HS, Lee NJ, Kim BJ, Choe EA (2002) Native feather degradation by Fervidobacterium islandicum AW-1, a newly isolating keratinase-producing thermophilic anaerobe. Arch Microbiol 178:538–547
Ramnani P, Gupta R (2004) Optimization of medium composition for keratinase production on feather by Bacillus licheniformis RG1 using statistical methods involving response surface methodology. Biotech Appl Biochem 40:491–496
Mona AE (2007) Isolation and partial characterization of extracellular keratinase from a novel mesophilic Streptomyces albus AZA. Res J Agric Biol Sci 3(6):808–817
Böckle B, Müller R (1997) Reduction of disulfide bonds by Streptomyces pactum during growth on chicken feathers. Appl Environ Microbiol 63:790–792
Page WJ, Stock JJ (1974) Phosphate-mediated alteration of the Microsporum gypseum germination protease specificity for substrate: enhanced keratinase activity. J Bacteriol 117:422–431
Yu RJ, Harmon SR, Grappel SF, Blank F (1971) Two cell-bound keratinases of Trichophyton mentagrophytes. J Inv Dermatol 56:27–32
Kang SW, Hong SI, Kim SW (2005) Identification of Aspergillus strain with antifungal activity against Phytophthora species. J Microbiol Biotechnol 15:227–233
Gradisar H, Friedrich J, Krizaj I, Jerala R (2005) Similarities and specificities of fungal keratinolytic proteases: comparison of keratinases of Paecilomyces marquandii and Doratomyces microsporus to some known proteases. Appl Environ Microbiol 71:3420–3426
Sivakumar T, Ramasubramanian V, Arasu VT, Shankar T, Prabhu D (2012) Screening of keratinolytic bacteria from the feather dumping site of Sivakasi. Insights Bacteriol 1(1):1–6
Kaul S, Sumbali G (2000) Production of extracellular keratinase by keratinophilic fungal species inhabiting feathers of living poultry birds (Gallus domesticus): a comparison. Mycopathologia 146:19–24
Kumar EV, Srijana M, Chaitanya K, Reddy YHK, Reddy G (2012) Biodegradation of poultry feathers by a novel bacterial isolate Bacillus altitudinis GVC11. Ind J Biotechnol 10:502–507
Letourneau F, Soussotte V, Bressollier P, Branland P, Nerneuil B (1998) Keratinolytic activity of Streptomyces sp. SK1-02: a new isolated strain. Lett Appl Microbiol 26:77–80
Acknowledgment
The authors are thankful to the University Grant Commission [sanction no. F.11-114/2008 (BRS)], Government of India, India, for the financial assistance. The assistance with HPLC and MALDI-TOF MS analysis by Dr. Santi Mohan Mandal, IIT-Khargpur, India, is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 420 kb)
Rights and permissions
About this article
Cite this article
Paul, T., Das, A., Mandal, A. et al. Production and purification of keratinase using chicken feather bioconversion by a newly isolated Aspergillus fumigatus TKF1: detection of valuable metabolites. Biomass Conv. Bioref. 4, 137–148 (2014). https://doi.org/10.1007/s13399-013-0090-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-013-0090-6