Abstract
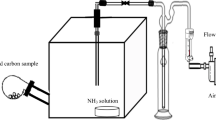
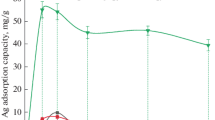
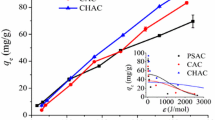
For the first time, an efficient method for the removal of mercury from Arabian gas-condensate samples was reported. Silver nanoparticles (AgNPs) functionalized with activated carbon (AC) prepared from local date-pits were used as an adsorbent. The physical and chemical properties of AgNP-AC were characterized using surface characterization techniques, and the adsorbent was evaluated under different experimental conditions. These factors considered include AgNP concentrations, contact time, the adsorbent dosage of AgNP-AC and initial mercury concentration. Langmuir adsorption isotherm, pseudo-second-order kinetics and Weber intraparticle diffusion models were used to evaluate the adsorption properties of the AgNP-AC. The results obtained revealed that at a low contact time, 25 mM AgNPs functionalized on AC provided the highest adsorption efficiency (98%) in the removal of mercury from Arabian gas-condensate. Also, it was observed that the increase in AgNP-AC dosage and initial mercury concentration plays a significant role in the mercury removal process. With a correlation coefficient of 0.9987, the adsorption process fits the Langmuir isotherm, suggesting that the adsorption is homogenous and monolayer.
Similar content being viewed by others
References
Boening, D.W.: Ecological effects, transport, and the fate of mercury: a general review. Chemosphere 40, 1335–1351 (2000). https://doi.org/10.1016/S0045-6535(99)00283-0
Wilhelm, S.M.; Liang, L.; Kirchgessner, D.: Identification and properties of mercury species in crude oil. Energy Fuels 20, 180–186 (2006). https://doi.org/10.1021/ef0501391
Pacyna, E.G.; Pacyna, J.M.; Steenhuisen, F.; Wilson, S.: Global anthropogenic mercury emission inventory for 2000. Atmos. Environ. 40, 4048–4063 (2006). https://doi.org/10.1016/j.atmosenv.2006.03.041
Wilhelm, S.M.; Bloom, N.: Mercury in petroleum. Fuel Process. Technol. 63, 1–27 (2000). https://doi.org/10.1016/S0378-3820(99)00068-5
Pirrone, N.; Cinnirella, S.; Feng, X.; Finkelman, R.B.; Friedli, H.R.; Leaner, J.; Mason, R.; Mukherjee, A.B.; Stracher, G.B.; Streets, D.G.; Telmer, K.: Global mercury emissions to the atmosphere from anthropogenic and natural sources. Atmos. Chem. Phys. 10, 5951–5964 (2010). https://doi.org/10.5194/acp-10-5951-2010
Rice, K.M.; Walker, E.M.; Wu, M.; Gillette, C.; Blough, E.R.: Environmental mercury and its toxic effects. J. Prev. Med. Public Health. 47, 74–83 (2014). https://doi.org/10.3961/jpmph.2014.47.2.74
US-EPA 1997 Mercury Study Report to Congress (EPA-452/R-97-007) Volume V: Health effects of mercury and mercury compounds. https://www.epa.gov/sites/production/files/2015-09/documents/volume5.pdf
Awad, F.S.; AbouZied, K.M.; Abou El-Maaty, W.M.; El-Wakil, A.M.; Samy El-Shall, M.: Effective removal of mercury(II) from aqueous solutions by chemically modified graphene oxide nanosheets. Arab. J. Chem. (2018). https://doi.org/10.1016/j.arabjc.2018.06.018
Zabihi, M.; Ahmadpour, A.; Haghighi Asl, A.: Removal of mercury from water by carbonaceous sorbents derived from walnut shell. J. Hazard. Mater. 167, 230–236 (2009). https://doi.org/10.1016/j.jhazmat.2008.12.108
Zhang, F.-S.; Nriagu, J.O.; Itoh, H.: Mercury removal from water using activated carbons derived from organic sewage sludge. Water Res. 39, 389–395 (2005). https://doi.org/10.1016/j.watres.2004.09.027
Zhang, F.S.; Nriagu, J.O.; Itoh, H.: Photocatalytic removal and recovery of mercury from water using TiO2-modified sewage sludge carbon. J. Photochem. Photobiol. A Chem. 167, 223–228 (2004)
El-Naas, M.H.; Alhaija, M.A.; Al-Zuhair, S.: Evaluation of an activated carbon packed bed for the adsorption of phenols from petroleum refinery wastewater. Environ. Sci. Pollut. Res. 24, 1–10 (2017). https://doi.org/10.1007/s11356-017-8469-8
Lopes, A.R.; Scheer, A.; de, P.; Silva, G.V.; Yamamoto, C.I.: Pd-Impregnated activated carbon and treatment acid to remove sulfur and nitrogen from diesel. Rev. Mater. 21, 407–415 (2016). https://doi.org/10.1590/S1517-707620160002.0038
Yahya, M.A.; Al-Qodah, Z.; Ngah, C.W.Z.: Agricultural bio-waste materials as potential sustainable precursors used for activated carbon production: a review. Renew. Sust. Energ. Rev. 46, 218–235 (2015). https://doi.org/10.1016/j.rser.2015.02.051
Esmaeili, A.; Saremnia, B.; Kalantari, M.: Removal of mercury(II) from aqueous solutions by biosorption on the biomass of Sargassum glaucescens and Gracilaria corticata. Arab. J. Chem. 8, 506–511 (2015). https://doi.org/10.1016/j.arabjc.2012.01.008
Alhamed, Y.A.; Bamufleh, H.S.: Sulfur removal from model diesel fuel using granular activated carbon from dates’ stones activated by ZnCl 2. Fuel 88, 87–94 (2009). https://doi.org/10.1016/j.fuel.2008.07.019
Daud, W.: Comparison on pore development of activated carbon produced from palm shell and coconut shell. Bioresour. Technol. 93, 63–69 (2004). https://doi.org/10.1016/j.biortech.2003.09.015
Lozano-Castelló, D.; Cazorla-Amorós, D.; Linares-Solano, A.; Quinn, D.: Influence of pore size distribution on methane storage at relatively low pressure: preparation of activated carbon with optimum pore size. Carbon. 40, 989–1002 (2002). https://doi.org/10.1016/S0008-6223(01)00235-4
Al-Fehaid, K.: Saudi Arabia produces 17 percent of world’s dates (2014). http://english.alarabiya.net/en/business/economy/2014/07/28/Saudi-Arabia-produces-17-of-world-dates.html. Accessed 25 July 2018
Haimour, N.M.; Emeish, S.: Utilization of date stones for production of activated carbon using phosphoric acid. Waste Manag. 26, 651–660 (2006). https://doi.org/10.1016/j.wasman.2005.08.004
Agoudjil, B.; Benchabane, A.; Boudenne, A.; Ibos, L.; Fois, M.: Renewable materials to reduce building heat loss: characterization of date palm wood. Energy Build. 43, 491–497 (2011). https://doi.org/10.1016/j.enbuild.2010.014
Hua, M.; Zhang, S.; Pan, B.; Zhang, W.; Lv, L.; Zhang, Q.: Heavy metal removal from water / wastewater by nanosized metal oxides?: A review. J. Hazard. Mater. 212, 317–331 (2012). https://doi.org/10.1016/j.jhazmat.2011.10.016
Ge, F.; Li, M.-M.; Ye, H.; Zhao, B.-X.: Effective removal of heavy metal ions Cd2+, Zn2+, Pb2+, Cu2+ from aqueous solution by polymer-modified magnetic nanoparticles. J. Hazard. Mater. 211–212, 366–372 (2012). https://doi.org/10.1016/j.jhazmat.2011.12.013
Luo, G.; Yao, H.; Xu, M.; Cui, X.; Chen, W.; Gupta, R.; Xu, Z.: Carbon nanotube-silver composite for mercury capture and analysis. Energy Fuels. 24, 419–426 (2010). https://doi.org/10.1021/ef900777v
Ren, W.; Zhu, C.; Wang, E.: Enhanced sensitivity of a direct SERS technique for Hg2+ detection based on the investigation of the interaction between silver nanoparticles and mercury ions. Nanoscale 4, 5902 (2012). https://doi.org/10.1039/c2nr31410j
Ren, W.; Zhu, C.; Wang, E.: Enhanced sensitivity of a direct SERS technique for Hg2+ detection based on the investigation of the interaction between silver nanoparticles and mercury ions. Nanoscale 4, 5902–5909 (2012)
Deng, L.; Ouyang, X.; Jin, J.; Ma, C.; Jiang, Y.; Zheng, J.; Li, J.; Li, Y.; Tan, W.; Yang, R.: Exploiting the higher specificity of silver amalgamation: selective detection of mercury(II) by forming Ag/Hg amalgam. Anal. Chem. 85, 8594–8600 (2013)
Mulfinger, L.; Solomon, S.D.; Bahadory, M.; Jeyarajasingam, A.V.; Rutkowsky, S.A.; Boritz, C.: Synthesis and Study of Silver Nanoparticles. J. Chem. Educ. 84, 322 (2007)
Tang, C.; Sun, W.; Yan, W.: Green and facile fabrication of silver nanoparticles loaded activated carbon fibers with long-lasting antibacterial activity. RSC Adv. 4, 523–530 (2014). https://doi.org/10.1039/C3RA44799E
Tuan, T.Q.; Son, N.Van; Dung, H.T.K.; Luong, N.H.; Thuy, B.T.; Anh, N.T.Van; Hoa, N.D.; Hai, N.H.: Preparation and properties of silver nanoparticles loaded in activated carbon for biological and environmental applications. J. Hazard. Mater. 192, 1321–1329 (2011). https://doi.org/10.1016/j.jhazmat.2011.06.044
Inbaraj, B.; Sulochana, N.: Mercury adsorption on a carbon sorbent derived from fruit shell of Terminalia catappa. J. Hazard. Mater. 133, 283–290 (2006). https://doi.org/10.1016/j.jhazmat.2005.10.025
Li, Q.; Qi, Y.; Gao, C.: Chemical regeneration of spent powdered activated carbon used in decolorization of sodium salicylate for the pharmaceutical industry. J. Clean. Prod. 86, 424–431 (2015). https://doi.org/10.1016/j.jclepro.2014.08.008
Xin-hui, D.; Srinivasakannan, C.; Jin-sheng, L.: Process optimization of thermal regeneration of spent coal-based activated carbon using steam and application to methylene blue dye adsorption. J. Taiwan Inst. Chem. Eng. 45, 1618–1627 (2014). https://doi.org/10.1016/j.jtice.2013.10.019
Kuang, M.; Yang, G.; Chen, W.; Zhang, Z.: Study on mercury desorption from silver-loaded activated carbon fibre and activated carbon fibre. J. Fuel Chem. Technol. 36, 468–473 (2008). https://doi.org/10.1016/S1872-5813(08)60030-4
Acknowledgements
The authors would like to acknowledge the Deanship of Scientific Research and the King Fahd University of Petroleum & Minerals for the research support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Adio, S.O., Rana, A., Chanabsha, B. et al. Silver Nanoparticle-Loaded Activated Carbon as an Adsorbent for the Removal of Mercury from Arabian Gas-Condensate. Arab J Sci Eng 44, 6285–6293 (2019). https://doi.org/10.1007/s13369-018-3682-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-018-3682-4