Abstract
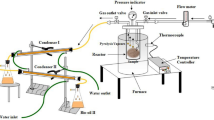
Palm mid fraction produced from Malaysian palm olein was hydrogenated using industrial catalyst (PRICAT 9910) on both pilot and industrial units. Effects of hydrogenation parameters, including temperature, catalyst concentration, reaction time, and circulation rate on final fatty acid composition and trans-fatty acid content were investigated. Mass transfer limitations were indicated particularly in the industrial plant runs. Mass transfer coefficient in the gas–liquid transfer was identified for various circulation rates. The combined mass transfer resistance at the liquid–solid interface and within the catalyst pellet was calculated as a function of different temperatures and circulation rates.

Similar content being viewed by others
References
Chong, C.L.: Chemical and physical properties of palm oil and palm kernel Oil. In: Selected readings on palm oil and its uses. Palm Oil Research Institute of Malaysia (PORIM), Kuala Lumpur, 60–77 (1994)
Chakravarty Y., Bhatia S., Saraf DN.: A simulation study of industrial vegetable oil hydrogenation reactors. J. Am. Oil Chem. Soc. 59, 157 (1982)
Fernandez M.B., Tonetto G.M., Crapiste G.H., Damiani D.E.: Revisiting the hydrogenation of sunflower oil over a Ni catalyst. J. Food Eng. 82, 199–208 (2007)
Cepeda E.A., Calvo B.: Sunflower oil hydrogenation: study using response surface methodology. J. Food Eng. 89, 370–374 (2008)
Anderson K., Hell M., Lowendahl L., Scoon N.: J. Am. Oil. Chem. Soc. 51, 171 (1974)
Choo H.P., Liew K.Y., Liu H.F., Seng C.E: Hydrogenation of palm olein catalyzed by polymer stabilized Pt colloids. J. Mol. Catal. A Chem. 165, 127–134 (2001)
Rangel, E.R.: Contribution to the study of heterogeneous catalytic reactions in SCFs: hydrogenation of sunflower oil in Pd catalysts at single-phase conditions. PhD thesis, University Politècnica de Cataluny, Spain (2005)
Savchenko V.I., Makaryan I.A.: Palladium catalyst for the production of pure Margarine: catalyst and new non-filtration technology improve production and quality. Platinum Metal Rev. 43, 74–82 (1999)
Fillion B., Morsi B., Heier K., Machado R.: The kinetics for soybean oil hydrogenation using a commercial Ni/Al2O3. Ind. Eng. Chem. Res. 4, 697–709 (2002)
Choo H.P., Liew Y.K., Liu H., Chye Eng Seng C.E., Mahmood K., Bettahar M.: Activity and selectivity of noble metal colloids for the hydrogenation of polyunsaturated soybean oil. J. Mol. Catal. A Chem. 191, 113–121 (2003)
Manoj A.T., Vijaykumar V. M.: Studies in catalytic transfer hydrogenation of soybean oil using ammonium formate as donor over 5% Pd/C catalyst. Chem. Eng. J. 123, 31–34 (2006)
Armfield: Instruction manual for hydrogenation pilot plant FT67 (1997)
AOCS: Official Methods of Analysis. Association of Official Analytical Chemists, 18th edn., Virginia, USA (2002)
Singh D., Rezac M.E., Prfomm R.P.: Partial hydrogenation of soybean oil with minimal trans fat production using a Pt-deocorated polymeric membrane reactor. J. Am. Oil Chem. Soc. 86, 93–101 (2009)
Yuan W., Hansen A.C., Zhang Q.: Vapor pressure and normal boiling point prediction for pure methyl esters and biodiesel fuels. Fuel 84, 943–950 (2005)
Sander, R.: Compilation of Henry’s law constant for inorganic and organic species of potential importance in environmental chemistry. Air Chemistry Department, Max-Planck Institute of Chemistry, Germany, Version 3, (1999)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alsobaai, A.M., Shaibani, A.M.A., Moustafa, T. et al. Hydrogenation of Malaysian Palm Mid Fraction on Pricat Nickel Catalyst Using Pilot and Industrial Units. Arab J Sci Eng 38, 2273–2278 (2013). https://doi.org/10.1007/s13369-013-0558-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-013-0558-5