Abstract
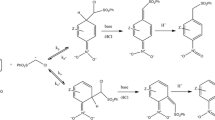
The reactions of a nucleophilic dianion with a series of activated aryl bromides were studied in the gas phase. Nucleophilic aromatic substitution (SNAr) as well as proton transfer reactions were observed. Rate constants and branching ratios were determined for all the reactions and the experimental data are supported by ab initio calculations. Reactions with bis-trifluoromethylbromobenzenes give only SNAr reactions and the rate constants follow the expected pattern, with substituents at the ortho and para positions having the greatest impact. Reactions of polyfluorobromobenzenes give a mix of proton transfer (when possible) and SNAr, with both bromide and fluoride acting as leaving groups. The latter is much less thermodynamically favorable but is the dominant pathway in each case. The selectivity of the reactions indicate that the products are determined early on the potential energy surface, before there is significant cleavage of the bond to the leaving group—the reaction is potentially directed by the initial formation of a hydrogen bond with the arene. The computational data also suggest that hydrogen bonding in the product ion–ion complexes can stabilize the system until there is sufficient charge separation to use the internal Coulomb repulsion to drive the reactions to products. Overall, the results highlight (1) the ability of multiply-charged systems to efficiently funnel their Coulomb repulsion into reaction processes that are intrinsically unfavorable, and (2) the high degree of selectivity that can be attained even in systems with multiple, low-barrier pathways.

ᅟ








Similar content being viewed by others
References
Bunnett, J.F., Zahler, R.E.: Aromatic nucleophilic substitution reactions. Chem. Rev. 49, 273–412 (1951)
Miller, J.: Aromatic Nucleophilic Substitution. Elsevier, Amsterdam. 1–26 (1968)
Bernasconi, C.F.: Kinetic-behavior of short-lived anionic sigma-complexes. Acc. Chem. Res. 11, 147–152 (1978)
Buncel, E., Dust, J.M., Terrier, F.: Rationalizing the regioselectivity in polynitroarene anionic sigma-adduct formation—relevance to nucleophilic aromatic-substitution. Chem. Rev. 95, 2261–2280 (1995)
Briscese, S.M., Riveros, J.M.: Gas-phase nucleophilic reactions of aromatic systems. J. Am. Chem. Soc. 97, 230–231 (1975)
Giroldo, T., Xavier, L.A., Riveros, J.M.: An unusually fast nucleophilic aromatic displacement reaction: the gas-phase reaction of fluoride ions with nitrobenzene. Angew. Chem. Int. Ed. 43, 3588–3590 (2004)
Ingemann, S., Nibbering, N.M.M.: Gas-phase reactions of anions with 2-fluoroanisole, 3-fluoroanisole, and 4-fluoroanisole. J. Org. Chem. 48, 183–191 (1983)
Ingemann, S., Nibbering, N.M.M.: Gas-phase reactions between anions and alkyl pentafluorophenyl ethers. Nouv. J. Chim. 8, 299–304 (1984)
Ingemann, S., Nibbering, N.M.M., Sullivan, S.A., Depuy, C.H.: Nucleophilic aromatic-substitution in the gas-phase—the importance of F-ion molecule complexes formed in gas-phase reactions between nucleophiles and some alkyl pentafluorophenyl ethers. J. Am. Chem. Soc. 104, 6520–6527 (1982)
Laerdahl, J.K., Uggerud, E.: Gas phase nucleophilic substitution. Int. J. Mass Spectrom. 214, 277–314 (2002)
Linnert, H.V., Riveros, J.M.: Benzyne-related mechanisms in the gas-phase ion–molecule reactions of haloarenes. Int. J. Mass Spectrom. Ion Process. 140, 163–176 (1994)
Gronert, S., Fagin, A.E., Wong, L.: Direct measurements of deuterium kinetic isotope effects in anionic, gas-phase substitution and elimination reactions. J. Am. Chem. Soc. 129, 5330–5331 (2007)
Gronert, S., Fagin, A.E., Okamoto, K., Mogali, S., Pratt, L.M.: Leaving group effects in gas-phase substitutions and eliminations. J. Am. Chem. Soc. 126, 12977–12983 (2004)
Gronert, S.: Gas phase studies of the competition between substitution and elimination reactions. Acc. Chem. Res. 36, 848–857 (2003)
Nettey, S., Swift, C.A., Joviliano, R., Noin, D.O., Gronert, S.: The impact of substituents on the transition states of S(N)2 and E2 reactions in aliphatic and vinylic systems: remarkably facile vinylic eliminations. J. Am. Chem. Soc. 134, 9303–9310 (2012)
Gronert, S.: Coulomb repulsion in multiply charged ions: a computational study of the effective dielectric constants of organic spacer groups. Int. J. Mass Spectrom. 185, 351–357 (1999)
Gronert, S.: Quadrupole ion trap studies of fundamental organic reactions. Mass Spectrom. Rev. 24, 100–120 (2005)
Gronert, S.: Estimation of effective ion temperatures in a quadrupole ion trap. J. Am. Soc. Mass Spectrom. 9, 845–848 (1998)
Frisch, M.J., Trucks, G.W., Schlegel, H.B., Scuseria, G.E., Robb, M.A., Cheeseman, J.R., Montgomery Jr., J.A., Vreven, T., Kudin, K.N., Burant, J.C., Millam, J.M., Iyengar, S.S., Tomasi, J., Barone, V., Mennucci, B., Cossi, M., Scalmani, G., Rega, N., Petersson, G.A., Nakatsuji, H., Hada, M., Ehara, M., Toyota, K., Fukuda, R., Hasegawa, J., Ishida, M., Nakajima, T., Honda, Y., Kitao, O., Nakai, H., Klene, M., Li, X., Knox, J.E., Hratchian, H.P., Cross, J.B., Adamo, C., Jaramillo, J., Gomperts, R., Stratmann, R.E., Yazyev, O., Austin, A.J., Cammi, R., Pomelli, C., Ochterski, J.W., Ayala, P.Y., Morokuma, K., Voth, G.A., Salvador, P., Dannenberg, J.J., Zakrzewski, V.G., Dapprich, S., Daniels, A.D., Strain, M.C., Farkas, O., Malick, D.K., Rabuck, A.D., Raghavachari, K., Foresman, J.B., Ortiz, J.V., Cui, Q., Baboul, A.G., Clifford, S., Cioslowski, J., Stefanov, B.B., Liu, G., Liashenko, A., Piskorz, P., Komaromi, I., Martin, R.L., Fox, D.J., Keith, T., Al-Laham, M.A., Peng, C.Y., Nanayakkara, A., Challacombe, M., Gill, P.M.W., Johnson, B., Chen, W., Wong, M.W., Gonzalez, C., Pople, J.A.: Gaussian 03 Revision B04. Gaussian Inc, Pittsburgh (2003)
Pople, J.A., Scott, A.P., Wong, M.W., Radom, L.: Scaling factors for obtaining fundamental vibrational frequencies and zero-point energies from HF/6-31G* and MP2/6-31G* harmonic frequencies. Isr. J. Chem. 33, 345–350 (1993)
Bartmess, J.E.: In: Nist Standard Reference Database Number 69; Mallard, W.G., Linstrom, P.J., Eds. National Institute of Standards and Technology (available at: http://webbook.nist.gov): Gaithersburg, MD (2013). Accessed 1 June 2013
Su, T., Bowers, M.T.: In: Bowers, M.T. (ed.) Gas Phase Ion Chemistry, Vol. 1, pp. 83–118. Academic Press, New York (1979)
Gronert, S., Fong, L.M.: Gas phase reactions of dianions. 2. The effect of a second charge on S(N)2 potential energy surfaces: an ab initio study. Int. J. Mass Spectrom. 192, 185–190 (1999)
Bunnett, J.F., Garbisch, E.W., Pruitt, K.M.: The “Element Effect” as a criterion of mechanism in activated aromatic nucleophilic substitution reactions. J. Am. Chem. Soc. 78, 385–391 (1957)
Senger, N.A., Bo, B., Cheng, Q., Keeffe, J.R., Gronert, S., Wu, W.M.: The element effect revisited: factors determining leaving group ability in activated nucleophilic aromatic substitution reactions. J. Org. Chem. 77, 9535–9540 (2012)
Kato, S., DePuy, C.H., Gronert, S., Bierbaum, V.M.: Gas phase hydrogen/deuterium exchange reactions of fluorophenyl anions. J. Am. Soc. Mass Spectrom. 10, 840–847 (1999)
Manikandan, P., Zhang, J.X., Hase, W.L.: Chemical dynamics simulations of X– + CH3Y => XCH3 + Y– gas-phase S(N)2 nucleophilic substitution reactions. Nonstatistical dynamics and nontraditional reaction mechanisms. J. Phys. Chem. A 116, 3061–3080 (2012)
Craig, S.L., Zhong, M.L., Brauman, J.I.: Translational energy dependence and potential energy surfaces of gas phase S(N)2 and addition-elimination reactions. J. Am. Chem. Soc. 121, 11790–11797 (1999)
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 291 kb)
Rights and permissions
About this article
Cite this article
Eanes, A.D., Noin, D.O., Kebede, M.K. et al. Nucleophilic Aromatic Substitution with Dianions: Reactions Driven by the Release of Coulomb Repulsion. J. Am. Soc. Mass Spectrom. 25, 10–17 (2014). https://doi.org/10.1007/s13361-013-0758-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13361-013-0758-y