Abstract
In the present study, we reinvestigate the diversity of Trichoderma in Poland utilizing a combination of morphological and molecular/phylogenetic methods. A total of 170 isolates were collected from six different substrata at 49 sites in Poland. These were divided among 14 taxa as follows: 110 of 170 Trichoderma isolates were identified to the species level by the analysis of their ITS1, ITS2 rDNA sequences as: T. harzianum (43 isolates), T. aggressivum (35), T. citrinoviride (11), T. hamatum (9), T. virens (6), T. longibrachiatum (4), T. polysporum (1), and T. tomentosum (1); 60 isolates belonging to the Viride clade were identified based on a fragment of the translation-elongation factor 1-alpha (tef1) gene as: T. atroviride (20 isolates), T. gamsii (2), T. koningii (17), T. viridescens (13), T. viride (7), and T. koningiopsis (1). Identifications were made using the BLAST interface in TrichOKEY and TrichoBLAST (http://www.isth.info). The most diverse substrata were soil (nine species per 22 isolates) and decaying wood (nine species per 75 isolates). The most abundant species (25%) isolated from all substrata was T. harzianum.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The fungal genus Trichoderma (Ascomycetes, Hypocreales) includes cosmopolitan soil-borne species that are frequently found also on decaying wood, compost, or other organic matter (Harman et al. 2004; Samuels 2006). Several Trichoderma species are significant biocontrol agents against fungal plant pathogens either through direct parasitism, competition with pathogens for nutrients, stimulators of plant health, or inducers of plant systemic resistance to pathogens (Hjeljord and Tronsmo 1998; Harman et al. 2004; Bailey et al. 2006). The ability for mycoparasitism in some species also has a negative economic impact in the commercial production of Agaricus bisporus (J.E. Lange) Imbach and Pleurotus ostreatus (Paulet) Rolland mushrooms, both of which are reported for Poland (Samuels et al. 2002; Krupke et al. 2003; Hatvani et al. 2007; Szczech et al. 2008). While Trichoderma is not pathogenic towards healthy mammals, there is a growing number of immunocompromised individuals who suffer opportunistic infections by some species (Kuhls et al. 1999; Kredics et al. 2003; Piens et al. 2004; Druzhinina et al. 2008), and volatile compounds produced by some Trichoderma species can cause allergic reactions (Tang et al. 2003; Caballero et al. 2007). Trichoderma species produce a wide diversity of metabolites, most notably commercially important cellulase and hemicellulases, antibiotics, peptaibiotics, as well as the toxins (such as trichodermamides) and trichothecenes that display in vitro cytotoxicity (Kubicek and Penttilä 1998; Sivasithamparam and Ghisalberti 1998; Garo et al. 2003; Liu et al. 2005; Nielsen et al. 2005; Degenkolb et al. 2006, 2008).
Because of the intimate relationship between species of Trichoderma and human activity, there is a great need for the accurate identification of Trichoderma species. However, accurate species identification based on morphology is difficult at best because of the paucity and similarity of useful morphological characters (Druzhinina et al. 2005; De Respinis et al. 2010), and increasing numbers of morphologically cryptic species that can be distinguished only through their DNA characters are being described (Atanasova et al. 2010; Samuels et al. 2010). This has already resulted in incorrect identification and the propagation of errors for strains associated with the production of secondary metabolites (Humphris et al. 2002), with human diseases (Gautheret et al. 1995), and biological control (Kullnig et al. 2001). However, with the advent of molecular methods and identification tools, which are based on sequence analysis of multiple genes (rDNA and genes encoding actin, calmodulin, endochitinase, RNA polymerase II, and translation-elongation factor 1-alpha [tef1]), it is now possible to identify every Trichoderma isolate and/or recognize it as a putative new species (Druzhinina et al. 2005; Samuels 2006; Kubicek et al. 2008).
At present, the International Subcommission on Trichoderma and Hypocrea Taxonomy lists 104 species, all of which have been characterized at the molecular level (http://www.isth.info). Seventy-five species of Hypocrea have been identified in temperate Europe, in particular, in Austria (Jaklitsch 2009). Nevertheless, the information about the diversity of Trichoderma/Hypocrea in Poland is scarce. A preliminary checklist of micromycetes in Poland reported 20 Trichoderma species (Mułenko et al. 2008). However, all of these species were identified between 1903 and 2002 based on morphological characters.
The objective of the present study was to document the occurrence and species diversity of Trichoderma collected from different substrata and locations in Poland.
Materials and methods
Substrata, storage, and isolation of pure cultures
Fungal isolates investigated in this study were collected from pieces of decaying wood, cultivated mushroom compost, samples of soil (garden, forest), and cereal grain (triticale, maize) at 49 sites in Poland (Table 1). Samples of decaying wood with white or brown rot were collected in parks and forests of the Wielkopolska region of Poland, placed in paper bags, dried at room temperature if wet, and stored until isolation. The pieces of decaying wood were plated on saltwater nutrient agar (SNA, Nirenberg 1976) and incubated at 20°C for 6 days. Putative Trichoderma colonies were purified by two rounds of subculturing on potato dextrose agar (PDA, Oxoid). Pure culture were transferred to the tube containing SNA and stored at −4°C for further study. Trichoderma spp. originated from other substrata were isolated according to the method described by Mańka (1974). Thirty-seven isolates originating from mushroom compost at mushroom farms in Poznań and in Skierniewice, as well as from forest soil of the Wielkopolski National Park were kindly supplied by Profs. H. Kwaśna and M. Mańka, Department of Forest Pathology, Poznań University of Life Sciences, and by Dr M. Szczech, Department of Plant Protection, Research Institute of Vegetable Crops, Skierniewice.
Morphological analysis
Fungal colonies were grown on PDA and SNA at 25°C for 7 days. Trichoderma species were identified according to Gams and Bissett (1998) and Samuels et al. (2002, 2009; http://nt.ars-grin.gov/taxadescriptions/keys/TrichodermaIndex.cfm).
Isolation of DNA
Mycelium for DNA extraction was obtained by inoculating Czapek-Dox broth (Sigma) with Yeast Extract (Oxoid) and streptomycin sulfate (50 mg/L−1, AppliChem), and after incubation at 25°C for 21 days on a rotary shaker (120 rpm). Mycelium was collected on filter paper in a Büchner funnel, washed with sterile water, frozen at −20°C, and freeze-dried.
Total DNA was extracted using the CTAB method (Doohan et al. 1998).
PCR amplification and sequencing
Primary identification was based on the sequencing of internal transcribed spacer regions 1 and 2 (ITS1 and ITS2) of the rRNA gene cluster. In cases where ITS1 and ITS2 did not provide unambiguous identification, a fragment of the translation-elongation factor 1-alpha (tef1) gene was sequenced. The ITS region of the rDNA of 170 isolates was amplified using primers ITS4, ITS5 (White et al. 1990). A fragment of tef1 gene containing the 4th and 5th introns was amplified using the primers Ef728M (Carbone and Kohn 1999) and tef1R (Kullnig-Gradinger et al. 2002). The PCR reaction was carried out in a 25-μl reaction mixture containing the following: 1 μl 50 ng/μl of DNA, 2.5 μl 10 × PCR buffer (50 mM KCl, 1.5 mM MgCl2, 10 mM Tris-HCl, pH 8.8, 0.1% Triton X-100), 1.5 μl 10 mM dNTP (GH Healthcare), 0.2 μl 100 mM of each primer, 19.35 μl MQ H2O, 0.25 μl (2 U/μl) DyNAzymeTM II DNA Polymerase (Finnzymes). Amplifications were performed in either a PTC-200 or PTC-100 thermocycler (MJ Research, USA) under the following conditions: initial denaturation 5 min at 94°C, 35 cycles of 45 s at 94°C, 45 s at 58°C (for the ITS region), or 63°C (for the tef1 fragment), 1 min at 72°C, with the final extension of 10 min at 72°C. Amplification products were separated on 1.5% agarose gel (Invitrogen) in 1 × TBE buffer (0.178 M Tris-borate, 0.178 M boric acid, 0.004 M EDTA) and stained with ethidium bromide. The 10-μl PCR products were combined with 2 μl of loading buffer (0.25% bromophenol blue, 30% glycerol). A 100-bp DNA Ladder Plus (Fermentas) was used as a size standard. PCR products were electrophoresed at 3 Vcm−1 for about 2 h, visualized under UV light, and photographed (Syngene UV visualizer). The 3-μl PCR products were purified with exonuclease I and shrimp alkaline phosphatase according to Chełkowski et al. (2003). Sequencing reactions were prepared using the ABI Prism BigDye Terminator Cycle Sequencing Ready Reaction Kit in 5 μl volume (Applied Biosystems, Switzerland). DNA sequencing was performed on an ABI PRISM 310 Genetic Analyzer (USA). Sequences were edited and assembled using Chromas v.1.43 (Applied Biosystems). CLUSTAL W (Thompson et al. 1994) and MUSCLE (Edgar 2004) were used to align the sequences; the resulting alignments were inspected and refined manually.
Molecular identification and phylogenetic analysis
For species identification, ITS1 and ITS 2 sequences were submitted to the BLAST interface in TrichOKEY (http://www.isth.info; Druzhinina et al. 2005; Druzhinina and Kubicek 2005). In ambiguous cases, the result was re-checked using the TrichoBLAST program based on tef1 gene sequences (Druzhinina and Kopchinskiy 2004a, b). All positions containing gaps and missing data were eliminated from the dataset. Phylogenetic analyses were performed in MEGA4 (Tamura et al. 2004). Both ITS1, ITS2 and tef1 gene sequences were analyzed using the maximum parsimony (Eck and Dayhoff 1966) approach of close-neighbor-interchange algorithm with search level 3 (Nei and Kumar 2000), in which the initial trees were obtained with the random addition of sequences (10,000 replicates). In total, there were 48 parsimony informative positions retained from an initial alignment of 368 for the ITS1, ITS2 sequences and 491 positions in the final dataset, of which 118 were parsimony informative for tef1 gene sequences. In both cases, to infer the consensus, phylogenetic trees bootstrapping with 10,000 data replicates was conducted (Felsenstein 1985).
Results
Species identification
A total of 170 isolates were obtained from the six different substrata at 49 localities in Poland. Of theses 170 Trichoderma isolates, 110 were identified at the species level by morphological characteristics and analysis of their ITS1, ITS2 nucleotide sequences as: T. harzianum Rifai (43 isolates), T. aggressivum Samuels & W. Gams (35), T. citrinoviride Bisset (11), T. hamatum (Bonord.) Bainier (9), T. virens (J.H. Mill., Giddens & A.A. Foster) Arx (6), T. longibrachiatum Rifai (4), T. polysporum (Link) Rifai (1), and T. tomentosum Bissett (1). In case of the remaining 60 Trichoderma isolates, where ITS1 and ITS2 did not provide unambiguous identification, the fragment of the tef1 gene was sequenced. Thereby, the following species were identified: T. atroviride P. Karst. (20 isolates), T. gamsii Samuels & Druzhin. (2), T. koningii Oudem. (17), T. viridescens (A.S. Horne & H.S. Will.) Jaklitsch & Samuels (13), T. viride Pers. (7), and T. koningiopsis Oudem. (1). The identification, origin, and NCBI GeneBank accession numbers of all isolates are given in Table 1.
Phylogenetic analysis
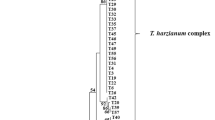
The result of the phylogenetic analysis based on the ITS sequences of 170 Trichoderma isolates is shown in Fig. 1. In the ITS tree, the Harzianum clade, with T. harzianum, T. aggressivum, and T. tomentosum, the Longibrachiatum Clade, with T. longibrachiatum and T. citrinoviride, and the species T. virens, T. hamatum, and T. polysporum were distinguished in a single moderately supported branch with bootstrap support of 79%. Forty-three strains were identified as T. harzianum, but this species is known to include several ITS alleles (Hermosa et al. 2004; Migheli et al. 2009) and is considered to be a species complex (Chaverri et al. 2003). In the present research, seven haplotypes of T. harzianum were found (HR1, HR2, HR3, HR4, HR5, HR6, and HR7, according to Table 1 and Fig. 1). With bootstrap support of only 53%, these seven haplotypes of T. harzianum formed a moderately well-supported (75%) clade with T. aggressivum and an unresolved polytomy with T. tomentosum. Two groups were distinguished within the Longibrachiatum clade with moderate to good bootstrap support. One group, with a bootstrap value of 70%, contains four strains of T. longibrachiatum. The second group, with a bootstrap value of 93% includes 11 strains of T. citrinoviride. Sixty isolates of Trichoderma, belonging to the Viride clade, formed a polytomy. A phylogenetic analysis based on tef1 sequences was performed for them (Fig. 2). As a result of this, the six species (T. koningii, T. atroviride, T. viride, T. viridescens, T. gamsii) were resolved with high bootstrap support.
Phylogenetic tree of the 170 Trichoderma isolates inferred by parsimony analysis of ITS1, ITS 2 sequences. Sequences obtained during this study are listed by their GenBank numbers in Table 1. The numbers given over branches indicate bootstrap coefficient >50%. The symbols given on the right (HR, AG, L, C, HM, cV) indicate the allelic groups of isolates, forming on the basis of ITS sequences identity. The isolates belonging to individual allelic groups are listed in Table 1
Phylogenetic tree of the 60 Trichoderma isolates inferred by parsimony analysis of tef1 sequences. Sequences obtained during this study are listed by their GenBank numbers in Table 1. The numbers given over branches indicate bootstrap coefficient >50%. The symbols given on the right (KO, VI, AT, VD) indicate the allelic groups of isolates, forming on the basis of tef1 sequences identity. The isolates belonging to individual allelic groups are listed in Table 1
Species diversity
Fourteen species of Trichoderma were identified among 170 isolates collected from six different substrata and 49 localities in Poland, using both morphological and molecular analysis. The highest diversity of Trichoderma species was detected in the set of 22 isolates originating from soil, which included nine species (T. atroviride, T. citrinoviride, T. gamsii, T. hamatum, T. harzianum, T. polysporum, T. tomentosum, T. viride, T. viridescens). Most of the isolates were collected from decaying wood (75), but among them, only nine species were found (T. atroviride, T. citrinoviride, T. gamsii, T. hamatum, T. harzianum, T. koningii, T. koningiopsis, T. viride, T. viridescens). The single strains of T. polysporum and T. tomentosum were isolated from soil, whereas all 17 strains of T. koningii were isolated from forest wood at several sites. The 58 isolates from mushroom compost and mushroom farms comprised seven species: T. aggressivum, T. atroviride, T. citrinoviride, T. harzianum, T. longibrachiatum, T. virens, and T. viride. In the limited samples from grains of Zea mays and Triticosecale Wittm. ex A. Camus as well as from garden compost, only three species were identified: T. atroviride, T. harzianum, and T. virens. T. harzianum was the most abundant species (25%) and was isolated from all substrata. It was the most common species isolated from pieces of decaying wood (40%, 30 isolates). After T, harzianum, T. atroviride, T. koningii, T. viridescens, and T. citrinoviride were the most abundant (respectively, 12%, 15%, 12%, and 7% of 112 isolates) Trichoderma species collected from soil, compost, forest wood, and cereal grains, respectively. The most common species isolated from mushroom compost was T. aggressivum (60% of isolates originated from mushroom compost and 20% of all isolates from the collection). T. hamatum, T. virens, T. viride, T. longibrachiatum, T. gamsii, T. koningiopsis, T. polysporum, and T. tomentosum were the most scarcely identified species of the genus (≤5% of all isolates from the collection).
Discussion
The present study is a preliminary domestic assessment of Trichoderma diversity in Poland. A collection of 170 isolates obtained from six different substrata and 49 localities in Poland were identified by phenetic observations and by analysis of the ITS 1, ITS 2 region of rRNA gene cluster and/or a fragment of the tef1 gene. A wide diversity of Trichoderma isolates was found (14 species were identified among 170 isolates) in comparison with the studies on the biodiversity of Trichoderma in South-East Asia (Kubicek et al. 2003), in Austria (Wuczkowski et al. 2003), in South America (Druzhinina et al. 2005), in China (Zhang et al. 2005), and on Sardinia (Migheli et al. 2009). The highest diversity of Trichoderma was found in Colombia, Mexico, Guatemala, Panama, Peru, Ecuador, and Brazil (Hoyos-Carvajal et al. 2009). Hoyos-Carvajal et al. (2009) recorded almost twice as many species from a comparably sized sample of 183 isolates collected in these neotropical regions.
Here and in a previous study, T. harzianum was the predominant taxon (Kubicek et al. 2003; Wuczkowski et al. 2003; Druzhinina et al. 2005, 2010; Zhang et al. 2005; Migheli et al. 2009). T. harzianum is the most commonly reported species in the genus, occurring in diverse ecosystems and ecological niches. However, it must be borne in mind that the name ‘T. harzianum’ applies to a species complex within which several morphologically cryptic phylogenetic species—haplotypes—are found (results presented here) and these ‘haplotype species’ may be seen to comprise a multiplicity of species when subjected to multilocus phylogenetic analysis (Chaverri et al. 2003; Gherbawy et al. 2004; Zhang et al. 2005; Druzhinina et al. 2010). In the present research, seven haplotypes (HR1–HR7) were evident in the analysis of ITS sequences for T. harzianum isolates. Haplotypes HR1, HR3, HR4, HR5, HR6, and HR7 correspond with ITS haplotypes, which are very common in Europe (Jaklitsch 2009, Chaverri et al. [unpublished]; Woo et al. [unpublished]). Haplotype HR2 (isolate AN 223) corresponds to the ex neo type strain of T. harzianum CBS 226.95, and, thus, represents T. harzianum sensu stricto. T. harzianum sensu stricto is also a species with a broad north temperate distribution, including at least North America, Europe, and Asia (Zhang et al. 2005; Chaverri and Samuels [unpublished]).
The second abundant species identified in the present study and the most prevalent species from mushroom compost was T. aggressivum (35 isolates). This result corresponds with the previous study of Szczech et al. (2008), who showed that, between 2004 and 2006, T. aggressivum was the most frequently isolated species of the genus identified in Polish mushroom farms. T. aggressivum has been isolated from mushroom compost used for A. bisporus cultivation in Europe and North America (Samuels et al. 2002). This species has only been isolated once from soil in Kenya (Samuels and Szakacs [unpublished]). It is not yet known whether this species also occurs in natural environments.
Other species identified in the present study were: T. atroviride (20 isolates), T. koningii (17), T. viridescens (13), T. citrinoviride (11), T. hamatum (9), T. viride (7), T. virens (6), T. longibrachiatum (4), T. gamsii (2), T. koningiopsis (1), T. polysporum (1), and T. tomentosum (1). These species are representative of a temperate Trichoderma biota (Kubicek et al. 2008). T. viride, T. viridescens, T. koningii, T. citrinoviride, T. aggressivum, T. tomentosum, and T. polysporum are rather restricted to temperate regions. However, T. longibrachiatum, T. virens, T. koningiopsis, T. hamatum, and T. atroviride were also found in the neotropical study (Hoyos-Carvajal et al. 2009).
The current results suggested that the most diverse habitats were soil (nine species per 22 isolates) and decaying wood (nine species per 75 isolates) gathered in parks and forests of the Wielkopolska region of Poland. The decaying wood was also the substrata from which the most isolates of Trichoderma (75) were collected. In this connection, we will continue to analyze the genetic and metabolic biodiversity of Trichoderma isolates originating from Polish mountains and isolated from forest wood with decay symptoms.
References
Atanasova L, Jaklitsch WM, Komoń-Zelazowska M, Kubicek CP, Druzhinina IS (2010) Clonal species Trichoderma parareesei sp. nov. likely resembles the ancestor of the cellulase producer Hypocrea jecorina/T. reesei. Appl Environ Microbiol 76:7259–7267
Bailey BA, Bae H, Strem MD, Roberts DP, Thomas SE, Crozier J, Samuels GJ, Choi I-Y, Holmes KA (2006) Fungal and plant gene expression during the colonization of cacao seedlings by endophytic isolates of four Trichoderma species. Planta 224:1449–1464
Caballero ML, Gómez M, González-Muñoz M, Reinoso L, Rodríguez-Pérez R, Alday E, Moneo I (2007) Occupational sensitization to fungal enzymes used in animal feed industry. Int Arch Allergy Immunol 144:231–239
Carbone I, Kohn LM (1999) A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 91:553–556
Chaverri P, Castlebury LA, Samuels GJ, Geiser DM (2003) Multilocus phylogenetic structure within the Trichoderma harzianum/Hypocrea lixii complex. Mol Phylogenet Evol 27:302–313
Chełkowski J, Golka L, Stępień Ł, 2003. Application of STS markers for leaf rust resistance genes in near-isogenic lines of spring wheat cv. Thatcher. J App Genet 44:323–338
De Respinis S, Vogel G, Benagli C, Tonolla M, Petrini O, Samuels GJ (2010) MALDI-TOF MS of Trichoderma: a model system for the identification of microfungi. Mycol Prog 9(1):79–100
Degenkolb T, Gräfenhan T, Berg A, Nirenberg HI, Gams W, Brückner H (2006) Peptaibiomics: screening for polypeptide antibiotics (peptaibiotics) from plant-protective Trichoderma species. Chem Biodivers 3:593–610
Degenkolb T, Dieckmann R, Nielsen KF, Gräfenhan T, Theis C, Zafari D, Chaverri P, Ismaiel A, Brückner H, von Döhren, Thrane U, Petrini O, Samuels GJ (2008) The Trichoderma brevicompactum clade: a separate lineage with new species, new peptaibiotics, and mycotoxins. Mycol Prog 7:177–219
Doohan FM, Parry DW, Jenkinson P, Nicholson P (1998) The use of species-specific PCR-based assays to analyse Fusarium ear blight of wheat. Plant Pathol 47:197–205
Druzhinina I, Kopchinskiy A (2004a) TrichoBLAST version 1.0, Multiloci database of phylogenetic markers and similarity search. Published online by the International Subcommission on Trichoderma and Hypocrea Taxonomy (ISTH). Home page at: http://www.isth.info/tools/blast/index.php
Druzhinina I, Kopchinskiy A (2004b) TrichoBLAST version 1.0, Trichoderma oligonucleotide key. Published online by the International Subcommission on Trichoderma and Hypocrea Taxonomy (ISTH). Home page at: http://www.isth.info/tools/molkey/index.php
Druzhinina I, Kubicek CP (2005) Species concepts and biodiversity in Trichoderma and Hypocrea: from aggregate species to species clusters? J Zhejiang Univ Sci B 6:100–112
Druzhinina IS, Kopchinskiy AG, Komoń M, Bissett J, Szakacs G, Kubicek CP (2005) An oligonucleotide barcode for species identification in Trichoderma and Hypocrea. Fungal Genet Biol 42:813–28
Druzhinina IS, Komoń-Zelazowska M, Kredics L, Hatvani L, Antal Z, Belayneh T, Kubicek CP (2008) Alternative reproductive strategies of Hypocrea orientalis and genetically close but clonal Trichoderma longibrachiatum, both capable of causing invasive mycoses of humans. Microbiology 154:3447–3459
Druzhinina IS, Kubicek CP, Komoń-Zelazowska M, Mulaw TB, Bissett J (2010) The Trichoderma harzianum demon: complex speciation history resulting in coexistence of hypothetical biological species, recent agamospecies and numerous relict lineages. BMC Evol Biol 10:94
Eck RV, Dayhoff MO (1966) Atlas of protein sequence and structure. National Biomedical Research Foundation, Silver Springs, MD
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32(5):1792–7
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Gams W, Bissett J (1998) Morphology and identification of Trichoderma. In: Harman GE, Kubicek CP (eds) Trichoderma and Gliocladium, vol. 1: basic biology, taxonomy and genetics. Taylor and Francis, London, p 334
Garo E, Starks CM, Jensen PR, Fenical W, Lobkovsky E, Clardy J (2003) Trichodermamides A and B, cytotoxic modified dipeptides from the marine-derived fungus Trichoderma virens. J Nat Prod 66:423–426
Gautheret A, Dromer F, Bourhis JH, Andremont A (1995) Trichoderma pseudokoningii as a cause of fatal infection in a bone marrow transplant recipient. Clin Infect Dis 20:1063–1064
Gherbawy Y, Druzhinina I, Shaban GM, Wuczowsky M, Yaser M, El-Naghy MA, Prillinger HJ, Kubicek CP (2004) Trichoderma populations from alkaline agricultural soil in the Nile valley, Egypt, consist of only two species. Mycol Prog 3:211–218
Harman GE, Howell CR, Viterbo A, Chet I, Lorito M (2004) Trichoderma species—opportunistic, avirulent plant symbionts. Nat Rev Microbiol 2:43–56
Hatvani L, Antal Z, Manczinger L, Szekeres A, Druzhinina IS, Kubicek CP, Nagy A, Nagy E, Vágvölgyi C, Kredics L (2007) Green Mold Diseases of Agaricus and Pleurotus spp. are caused by related but phylogenetically different Trichoderma species. Phytopathology 97:532–537
Hermosa MR, Keck E, Chamorro I, Rubio B, Sanz L, Vizcaíno JA, Grondona I, Monte E (2004) Genetic diversity shown in Trichoderma biocontrol isolates. Mycol Res 108:897–906
Hjeljord L, Tronsmo A (1998) Trichoderma and Gliocladium in biological control: an overview. In: Harman GE, Kubicek CP (eds) Trichoderma and Gliocladium, vol. 2: enzymes, biological control and commercial application. Taylor and Francis, London, pp 131–151
Hoyos-Carvajal L, Orduz S, Bissett J (2009) Genetic and metabolic biodiversity of Trichoderma from Colombia and adjacent neotropic regions. Fungal Genet Biol 46:615–631
Humphris SN, Bruce A, Buultjens E, Wheatley RE (2002) The effects of volatile microbial secondary metabolites on protein synthesis in Serpula lacrymans. FEMS Microbiol Lett 210:215–219
Jaklitsch WM (2009) European species of Hypocrea Part I. The green-spored species. Stud Mycol 63:1–91
Kredics L, Antal Z, Dóczi I, Manczinger L, Kevei F, Nagy E (2003) Clinical importance of the genus Trichoderma. A review. Acta Microbiol Immunol Hung 50:105–117
Krupke OA, Castle AJ, Rinker DL (2003) The North American mushroom competitor, Trichoderma aggressivum f. aggressivum, produces antifungal compounds in mushroom compost that inhibit mycelial growth of the commercial mushroom Agaricus bisporus. Mycol Res 107(12):1467–1475
Kubicek CP, Penttilä ME (1998) Regulation of production of plant polysaccharide degrading enzymes by Trichoderma. In: Harman GE, Kubicek CP (eds) Trichoderma and Gliocladium, vol. 1: basic biology, taxonomy and genetics. Taylor and Francis, London, pp 49–71
Kubicek CP, Bissett J, Druzhinina I, Kullnig-Gradinger C, Szakacs G (2003) Genetic and metabolic diversity of Trichoderma: a case study on South-East Asian isolates. Fungal Genet Biol 38:310–319
Kubicek CP, Komoń-Zelazowska M, Druzhinina IS (2008) Fungal genus Hypocrea/Trichoderma: from barcodes to biodiversity. J Zhejiang Univ Sci B 9(10):753–763
Kuhls K, Lieckfeldt E, Börner T, Guého E (1999) Molecular reidentification of human pathogenic Trichoderma isolates as Trichoderma longibrachiatum and Trichoderma citrinoviride. Med Mycol 37:25–33
Kullnig CM, Krupica T, Woo SL, Mach RL, Rey M, Benítez T, Lorito M, Kubicek CP (2001) Confusion abounds over identities of Trichoderma biocontrol isolates. Mycol Res 105:769–772
Kullnig-Gradinger C, Szakacs G, Kubicek CP (2002) Phylogeny and evolution of the genus Trichoderma: a multigene approach. Mycol Res 106:757–767
Liu R, Gu QQ, Zhu WM, Cui CB, Fan GT (2005) Trichodermamide A and aspergillazine A, two cytotoxic modified dipeptides from a marine-derived fungus Spicaria elegans. Arch Pharm Res 28:1042–1046
Mańka K (1974) Zbiorowiska grzybów jako kryterium oceny wpływu środowiska na choroby roślin. Zesz Probl Post Nauk Roln 160:9–23
Migheli Q, Balmas V, Komoñ-Zelazowska M, Scherm B, Fiori S, Kopchinskiy AG, Kubicek CP, Druzhinina IS (2009) Soils of a Mediterranean hot spot of biodiversity and endemism (Sardinia, Tyrrhenian Islands) are inhabited by pan-European, invasive species of Hypocrea/Trichoderma. Environ Microbiol 11(1):35–46
Mułenko W, Majewski T, Ruszkiewicz-Michalska M (eds) (2008) Biodiversity of Poland, vol. 9A. A preliminary checklist of micromycetes in Poland. Krytyczna lista grzybów mikroskopijnych Polski. pp. 500–504
Nei M, Kumar S (2000) Molecular evolution and phylogenetics. Oxford University Press, New York
Nielsen KF, Gräfenhan T, Zafari D, Thrane U (2005) Trichothecene production by Trichoderma brevicompactum. J Agric Food Chem 53:8190–8196
Nirenberg HI (1976) Untersuchungen über die morphologische und biologische Differenzierung in der Fusarium-Sektion Liseola. Mitteilungen aus der Biologischen Bundesanstalt für Land- und Forstwirtschaft Berlin-Dahlem 169:1–117
Piens M-A, Celard M, de Monbrison F, Grando J, Vandenesch F, Mottolese C, Picot S (2004) Trichoderma infection of cerebrospinal fluid shunt device in a non immunocompromised patient. J Mycol Med 14:49–51
Samuels GJ (2006) Trichoderma: systematics, the sexual state, and ecology. Phytopathology 96:195–206
Samuels GJ, Dodd SL, Gams W, Castlebury LA, Petrini O (2002) Trichoderma species associated with the green mold epidemic of commercially grown Agaricus bisporus. Mycologia 94:146–170
Samuels GJ, Chaverri P, Farr DF, McCray EB (2009) Trichoderma online. Systematic Mycology and Microbiology Laboratory. Home page at: http://nt.ars-grin.gov/taxadescriptions/keys/TrichodermaIndex.cfm
Samuels GJ, Ismaiel A, Bon M-C, De Respinis S, Petrini O (2010) Trichoderma asperellum sensu lato consists of two cryptic species. Mycologia 102:944–966
Sivasithamparam K, Ghisalberti EL (1998) Secondary metabolism in Trichoderma and Gliocladium. In: Kubicek CP, Harman GE (eds) Trichoderma and Gliocladium, vol. 1: basic biology, taxonomy and genetics. Taylor and Francis, London, pp 139–191
Szczech M, Staniaszek M, Habdas H, Uliński Z, Szymański J (2008) Trichoderma spp.—the cause of green mold on Polish mushroom farms. Veg Crops Res Bull 69:105–114
Tamura K, Nei M, Kumar S (2004) Prospects for inferring very large phylogenies by using the neighbor-joining method. Proc Natl Acad Sci USA 101:11030–11035
Tang P, Mohan S, Sigler L, Witterick I, Summerbell R, Campbell I, Mazzulli T (2003) Allergic fungal sinusitis associated with Trichoderma longibrachiatum. J Clin Microbiol 41:5333–5336
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
White TJ, Bruns T, Lee S, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Shinsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press, San Diego, pp 315–322
Wuczkowski M, Druzhinina I, Gherbawy Y, Klug B, Prillinger H-J, Kubicek CP (2003) Species pattern and genetic diversity of Trichoderma in a mid-European, primeval floodplain-forest. Microbiol Res 158:125–133
Zhang CL, Druzhinina IS, Kubicek CP, Xu T (2005) Trichoderma biodiversity in China: evidence for a North to South distribution of species in East Asia. FEMS Microbiol Lett 251:251–257
Acknowledgments
This work was supported by the Ministry of Science and Higher Education in Poland, Project No. NN310 203037. We thank Profs. H. Kwaśna and M. Mańka, Department of Forest Pathology, Poznań University of Life Sciences, and Dr. M. Szczech, Department of Plant Protection, Research Institute of Vegetable Crops, Skierniewice, for providing some of the isolates included in this study.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Błaszczyk, L., Popiel, D., Chełkowski, J. et al. Species diversity of Trichoderma in Poland. J Appl Genetics 52, 233–243 (2011). https://doi.org/10.1007/s13353-011-0039-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13353-011-0039-z