Abstract
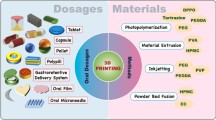
The involvement of recent technologies, such as nanotechnology and three-dimensional printing (3DP), in drug delivery has become the utmost importance for effective and safe delivery of potent therapeutics, and thus, recent advancement for oral drug delivery through 3DP technology has been expanded. The use of computer-aided design (CAD) in 3DP technology allows the manufacturing of drug formulation with the desired release rate and pattern. Currently, the most applicable 3DP technologies in the oral drug delivery system are inkjet printing method, fused deposition method, nozzle-based extrusion system, and stereolithographic 3DP. In 2015, the first 3D-printed tablet was approved by the US Food and Drug Administration (FDA), and since then, it has opened up more opportunities in the discovery of formulation for the development of an oral drug delivery system. 3DP allows the production of an oral drug delivery device that enables tailor-made formulation with customizable size, shape, and release rate. Despite the advantages offered by 3DP technology in the drug delivery system, there are challenges in terms of drug stability, safety as well as applicability in the clinical sector. Nonetheless, 3DP has immense potential in the development of drug delivery devices for future personalized medicine. This article will give the recent advancement along with the challenges of 3DP techniques for the development of oral drug delivery.

Graphical abstract








Similar content being viewed by others
References
Moulton SE, Wallace GG. 3-dimensional (3D) fabricated polymer based drug delivery systems. J Control Release. 2014;193:27–34.
Trenfield SJ, Awad A, Goyanes A, Gaisford S, Basit AW. 3D printing pharmaceuticals: drug development to frontline care. Trends Pharmacol Sci. 2018;39:440–51.
Liang K, Carmone S, Brambilla D, Leroux J-C. 3D printing of a wearable personalized oral delivery device: a first-in-human study. Sci Adv. American Association for the Advancement of Science [cited 2019 Jul 12]. 2018;4:eaat2544. https://doi.org/10.1126/sciadv.aat2544.
Bansal M, Sharma V, Singh G, Harikumar SL. 3D printing for the future of pharmaceuticals dosages forms. Int J Appl Pharm. 2018; 1–7.
Katakam P, Dey B, Assaleh FH, Hwisa NT, Adiki SK, Chandu BR, et al. Top-down and bottom-up approaches in 3D printing technologies for drug delivery challenges. Crit Rev Ther Drug Carrier Syst. 2015;32:61–87 Available from: http://www.ncbi.nlm.nih.gov/pubmed/25746205.[cited 2018 Jul 13].
Jamróz W, Szafraniec J, Kurek M. Jachowicz R. 3D printing in pharmaceutical and medical applications—recent achievements and challenges. Pharm Res . Springer. 2018;35:176.
Afsana JV, Haider N, Jain K. 3D printing in personalized drug delivery. Curr Pharm Des. 2019;24:5062–71 Available from: http://www.ncbi.nlm.nih.gov/pubmed/30767736. [cited 2019 Jul 15].
Norman J, Madurawe RD, Moore CMV, Khan MA, Khairuzzaman A. A new chapter in pharmaceutical manufacturing: 3D-printed drug products. Adv Drug Deliv Rev. 2017;108:39–50.
Osouli-Bostanabad K, Adibkia K. Made-on-demand, complex and personalized 3D-printed drug products. Bioimpacts. 2018;8:77–9 Tabriz University of Medical Sciences; Available from: http://www.ncbi.nlm.nih.gov/pubmed/29977828. [cited 2019 Jul 15].
Goyanes A, Det-Amornrat U, Wang J, Basit AW, Gaisford S. 3D scanning and 3D printing as innovative technologies for fabricating personalized topical drug delivery systems. J Control Release. 2016;234:41–8.
Yi H-G, Choi Y-J, Kang KS, Hong JM, Pati RG, Park MN, et al. A 3D-printed local drug delivery patch for pancreatic cancer growth suppression. J Control Release. 2016;238:231–41 Available from: http://www.ncbi.nlm.nih.gov/pubmed/27288878. [cited 2019 Jul 15].
Pandey M, Choudhury H, Yeun OC, Yin HM, Lynn TW, Tine CLY, et al. Perspectives of nanoemulsion strategies in the improvement of oral, parenteral and transdermal chemotherapy. Curr Pharm Biotechnol. 2018;19:276–92.
Choudhury H, Gorain B, Chatterjee B, Mandal UK, Sengupta P, Tekade RK. Pharmacokinetic and pharmacodynamic features of nanoemulsion following oral, intravenous, topical and nasal route. Curr Pharm Des. 2017;23.
Zeeshan F, Madheswaran T, Pandey M, Gorain B. Three-dimensional (3-D) printing technology exploited for the fabrication of drug delivery systems. Curr Pharm Des. 2018;24:5019–28.
Fina F, Goyanes A, Gaisford S, Basit AW. Selective laser sintering (SLS) 3D printing of medicines. Int J Pharm. 2017;529:285–93. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28668582.Accessed 15 July 2019.
Solanki NG, Tahsin M, Shah AV, Serajuddin AT. Formulation of 3D printed tablet for rapid drug release by fused deposition modeling: screening polymers for drug release, drug-polymer miscibility and printability. J Pharm Sci. 2018;107:390–401. [cited 2019 Jul 15]. https://doi.org/10.1016/j.xphs.2017.10.021.
First 3D-printed pill. Nat Biotechnol. 2015;33:1014 Available from: http://www.nature.com/articles/nbt1015-1014a. [cited 2019 15].
Ishengoma FR, Mtaho AB. 3D printing: developing countries perspectives. Int J Comput Appl. 2014. Available from: https://pdfs.semanticscholar.org/c251/6eba4c2c1d3254ac33c413d52f7ea79f8be4.pdf
Palo M, Holländer J, Suominen J, Yliruusi J, Sandler N. 3D printed drug delivery devices: perspectives and technical challenges. Expert Rev Med Devices. 2017;14:685–96.
FDA approves the first 3D printed drug product. Aprecia Pharm [Internet]. The Economic Times. 2015. http://www.multivu.com/players/English/7577251-aprecia-pharmaceuticals-spritam/. Accessed 15 July 2019.
Goyanes A, Robles Martinez P, Buanz A, Basit AW, Gaisford S. Effect of geometry on drug release from 3D printed tablets. Int J Pharm. 2015;494:657–63.
Prasad LK, Smyth H. 3D printing technologies for drug delivery: a review. Drug Dev Ind Pharm. Taylor & Francis. 2016 [cited 2019 Jan 7];42:1019–31. https://doi.org/10.3109/03639045.2015.1120743.
Ani Jose P, Christoper PG. 3D printing of pharmaceuticals—a potential technology in developing personalized medicine. Asian J Pharm Res Dev. 2018;6:46–54 Available from: http://ajprd.com. [cited 2019 Jul 15].
Okwuosa TC, Pereira BC, Arafat B, Cieszynska M, Isreb A, Alhnan MA. Fabricating a shell-core delayed release tablet using dual FDM 3D printing for patient-centred therapy. Pharm Res. 2017;34:427–37 Available from: http://www.ncbi.nlm.nih.gov/pubmed/27943014. [cited 2019 Jul 15].
Muley S, Nandgude T, Poddar S. Extrusion–spheronization a promising pelletization technique: in-depth review. Asian J Pharm Sci. 2016;11:684–99 Available from: https://www.sciencedirect.com/science/article/pii/S1818087616300721. Elsevier;[cited 2019 Jul 15].
Pietrzak K, Isreb A, Alhnan MA. A flexible-dose dispenser for immediate and extended release 3D printed tablets. Eur J Pharm Biopharm. 2015;96:380–7 Elsevier; [cited 2019 Jul 15]. Available from: https://www.sciencedirect.com/science/article/pii/S0939641115003306.
Konta A, García-Piña M, Serrano D. Personalised 3D printed medicines: which techniques and polymers are more successful? Bioengineering. 2017;4:79 [cited 2019 Jul 15]Available from: http://www.ncbi.nlm.nih.gov/pubmed/28952558.
Scoutaris N, Alexander MR, Gellert PR, Roberts CJ. Inkjet printing as a novel medicine formulation technique. J Control Release. 2011;156:179–85.
Boehm RD, Miller PR, Daniels J, Stafslien S, Narayan RJ. Inkjet printing for pharmaceutical applications. Mater Today. Elsevier Ltd. 2014;17:247–52.
Daly R, Harrington TS, Martin GD, Hutchings IM. Inkjet printing for pharmaceutics—a review of research and manufacturing. Int J Pharm. Elsevier B.V. 2015;494:554–67.
Buanz ABM, Saunders MH, Basit AW, Gaisford S. Preparation of personalized-dose salbutamol sulphate oral films with thermal ink-jet printing. Pharm Res. 2011 [cited 2018 Jul 13];28:2386–92. https://doi.org/10.1007/s11095-011-0450-5.
Meléndez PA, Kane KM, Ashvar CS, Albrecht M, Smith PA. Thermal inkjet application in the preparation of oral dosage forms: dispensing of prednisolone solutions and polymorphic characterization by solid-state spectroscopic techniques. J Pharm Sci. 2008;97:2619–36.
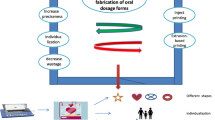
Merits and uses of 3DP in pharmaceutical drug delivery.
Genina N, Janßen EM, Breitenbach A, Breitkreutz J, Sandler N. Evaluation of different substrates for inkjet printing of rasagiline mesylate. Eur J Pharm Biopharm. 2013;85:1075–83.
Pardeike J, Strohmeier DM, Schrödl N, Voura C, Gruber M, Khinast JG, et al. Nanosuspensions as advanced printing ink for accurate dosing of poorly soluble drugs in personalized medicines. Int J Pharm. 2011;420:93–100.
KM S, Kulsum JU. 3D printing: a new avenue in pharmaceuticals. World J Pharm Res. 2016;5:1686–701.
He Y, Wu Y, Fu JZ, Gao Q, Qiu JJ. Developments of 3D printing microfluidics and applications in chemistry and biology: a review. Electroanalysis. 2016;1658–78.
Lewis JA. Direct ink writing of 3D functional materials. Adv Funct Mater. John Wiley & Sons, Ltd. 2006;16:2193–204.
Skowyra J, Pietrzak K, Alhnan MA. Fabrication of extended-release patient-tailored prednisolone tablets via fused deposition modelling (FDM) 3D printing. Eur J Pharm Sci. 2015;68:11–7.
Goyanes A, Chang H, Sedough D, Hatton GB, Wang J, Buanz A, et al. Fabrication of controlled-release budesonide tablets via desktop (FDM) 3D printing. Int J Pharm. 2015;496:414–20.
Kollamaram G, Croker DM, Walker GM, Goyanes A, Basit AW, Gaisford S. Low temperature fused deposition modeling (FDM) 3D printing of thermolabile drugs. Int J Pharm.Elsevier. 2018;545:144–52.
Goole J, Amighi K. 3D printing in pharmaceutics: a new tool for designing customized drug delivery systems. Int J Pharm. 2016;499:376–94.
Wen H, He B, Wang H, Chen F, Li P, Cui M, et al. Structure-based gastro-retentive and controlled-release drug delivery with novel 3D printing. AAPS PharmSciTech. Springer International Publishing. 2019;20:68.
Aprecia Pharmaceuticals. FDA approves the first 3D printed drug product. 2015
Gibson I, Rosen D, Stucker B (Brent). Additive manufacturing technologies: 3D printing, rapid prototyping, and direct digital manufacturing.
BO G, GV S, SB K, DR C, GS A, GP S. 3D Printing in pharmaceutical manufacturing: opportunities and challenges. Int J Bioassays. 2016;5:4723.
Wang J, Goyanes A, Gaisford S, Basit AW. Stereolithographic (SLA) 3D printing of oral modified-release dosage forms. Int J Pharm. 2016;503:207–12.
Alhijjaj M, Belton P, Qi S. An investigation into the use of polymer blends to improve the printability of and regulate drug release from pharmaceutical solid dispersions prepared via fused deposition modeling (FDM) 3D printing. Eur J Pharm Biopharm Elsevier. 2016;108:111–25.
Khaled SA, Burley JC, Alexander MR, Roberts CJ. Desktop 3D printing of controlled release pharmaceutical bilayer tablets. Int J Pharm. 2014;461:105–11 Available from: http://linkinghub.elsevier.com/retrieve/pii/S0378517313010144. [cited 2018 Jul 13].
Maroni A, Melocchi A, Parietti F, Foppoli A, Zema L, Gazzaniga A. 3D printed multi-compartment capsular devices for two-pulse oral drug delivery. J Control Release. 2017;268:10–8.
Genina N, Boetker JP, Colombo S, Harmankaya N, Rantanen J, Bohr A. Anti-tuberculosis drug combination for controlled oral delivery using 3D printed compartmental dosage forms: from drug product design to in vivo testing. J Control Release. 2017;268:40–8.
Kyobula M, Adedeji A, Alexander MR, Saleh E, Wildman R, Ashcroft I, et al. 3D inkjet printing of tablets exploiting bespoke complex geometries for controlled and tuneable drug release. J Control Release. 2017;261:207–15.
Sadia M, Arafat B, Ahmed W, Forbes RT, Alhnan MA. Channelled tablets: an innovative approach to accelerating drug release from 3D printed tablets. J Control Release. 2018;269:355–63.
Muwaffak Z, Goyanes A, Clark V, Basit AW, Hilton ST, Gaisford S. Patient-specific 3D scanned and 3D printed antimicrobial polycaprolactone wound dressings. Int J Pharm. 2017;527:161–70.
Pae A, Yoo R-K, Noh K, Paek J, Kwon K-R. The effects of mouthguards on the athletic ability of professional golfers. Dent Traumatol. 2013;29:47–51.
Nakajima K, Takeda T, Kawamura S, Shibusawa M, Nara K, Kaoru N, et al. A vacuum technique to increase anterior thickness of athletic mouthguards to achieve a full-balanced occlusion. Dent Traumatol. John Wiley & Sons, Ltd (10.1111). 2008;24:50–2.
Yamada A, Niikura F, Ikuta K. A three-dimensional microfabrication system for biodegradable polymers with high resolution and biocompatibility. J Micromechanics Microengineering. IOP Publishing. 2008;18:25035.
Zhang B, Seong B, Nguyen V, Byun D. 3D printing of high-resolution PLA-based structures by hybrid electrohydrodynamic and fused deposition modeling techniques. J Micromechanics Microengineering. IOP Publishing. 2016;26:25015.
Li Q, Wen H, Jia D, Guan X, Pan H, Yang Y, et al. Preparation and investigation of controlled-release glipizide novel oral device with three-dimensional printing. Int J Pharm. 2017;525:5–11.
Goyanes A, Kobayashi M, Martínez-Pacheco R, Gaisford S, Basit AW. Fused-filament 3D printing of drug products: microstructure analysis and drug release characteristics of PVA-based caplets. Int J Pharm. 2016;514:290–5.
Goyanes A, Buanz ABM, Hatton GB, Gaisford S, Basit AW. 3D printing of modified-release aminosalicylate (4-ASA and 5-ASA) tablets. Eur J Pharm Biopharm. 2015;89:157–62 Available from: http://linkinghub.elsevier.com/retrieve/pii/S0939641114003580. [cited 2018 Jul 13].
Tagami T, Fukushige K, Ogawa E, Hayashi N, Ozeki T. 3D printing factors important for the fabrication of polyvinylalcohol filament-based tablets. Biol Pharm Bull. 2017;40:357–64.
Fu J, Yin H, Yu X, Xie C, Jiang H, Jin Y, et al. Combination of 3D printing technologies and compressed tablets for preparation of riboflavin floating tablet-in-device (TiD) systems. Int J Pharm. Elsevier. 2018;549:370–9.
Linares V, Casas M, Caraballo I. Printfills: 3D printed systems combining fused deposition modeling and injection volume filling. Application to colon-specific drug delivery. Eur J Pharm Biopharm. Elsevier. 2019;134:138–43.
Alhnan MA, Okwuosa TC, Sadia M, Wan K-W, Ahmed W, Arafat B. Emergence of 3D printed dosage forms: opportunities and challenges. Pharm Res Springer US. 2016;33:1817–32.
Awad A, Trenfield SJ, Gaisford S, Basit AW. 3D printed medicines: a new branch of digital healthcare. Int J Pharm. 2018;548:586–96.
WHO | Global tuberculosis report 2018. WHO World Health Organization; 2019.
Gallo GG, Radaelli P. Rifampin. Anal Profiles Drug Subst. Academic Press. 1976;5:467–513.
Mariappan TT, Singh S. Regional gastrointestinal permeability of rifampicin and isoniazid (alone and their combination) in the rat. Int J Tuberc Lung Dis. 2003;7:797–803.
Shishoo CJ, Shah SA, Rathod IS, Savale SS, Vora MJ. Impaired bioavailability of rifampicin in presence of isoniazid from fixed dose combination (FDC) formulation. Int J Pharm. 2001;228:53–67.
O’Shaughnessy K. BMA new guide to medicine & drugs. 9th ed. London: Dorling Kindersley; 2015.
Reddymasu SC, Soykan I, McCallum RW. Domperidone: review of pharmacology and clinical applications in gastroenterology. Am J Gastroenterol. 2007;102:2036–45.
Nagarsenker MS, Garad SD, Ramprakash G. Design, optimization and evaluation of domperidone coevaporates. J Control Release. 2000;63:31–9.
Melocchi A, Parietti F, Loreti G, Maroni A, Gazzaniga A, Zema L. 3D printing by fused deposition modeling (FDM) of a swellable/erodible capsular device for oral pulsatile release of drugs. J Drug Deliv Sci Technol. 2015;30:360–7.
Chai X, Chai H, Wang X, Yang J, Li J, Zhao Y, et al. Fused deposition modeling (FDM) 3D printed tablets for intragastric floating delivery of domperidone. Sci Rep. Nature Publishing Group. 2017;7:2829.
Hwang Y-C, Kang M, Ahn CW, Park JS, Baik SH, Chung DJ, et al. Efficacy and safety of glimepiride/metformin sustained release once daily vs. glimepiride/metformin twice daily in patients with type 2 diabetes. Int J Clin Pract. John Wiley & Sons, Ltd (10.1111). 2013;67:236–43.
BNF. 74th ed. BMJ Group and the Royal Pharmaceutical Society of Great Britain; 2014.
McCreight LJ, Bailey CJ, Pearson ER. Metformin and the gastrointestinal tract. Diabetologia. Springer Berlin Heidelberg 2016;59:426–35.
Ning X, Sun J, Han X, Wu Y, Yan Z, Han J, et al. Strategies to improve dissolution and oral absorption of glimepiride tablets: solid dispersion versus micronization techniques. Drug Dev Ind Pharm. 2011;37:727–36.
Paes AH, Bakker A, Soe-Agnie CJ. Impact of dosage frequency on patient compliance. Diabetes Care American Diabetes Association. 1997;20:1512–7.
Gioumouxouzis CI, Baklavaridis A, Katsamenis OL, Markopoulou CK, Bouropoulos N, Tzetzis D, et al. A 3D printed bilayer oral solid dosage form combining metformin for prolonged and glimepiride for immediate drug delivery. Eur J Pharm Sci. 2018;120:40–52.
Song R. Mechanism of metformin: a tale of two sites. Diabetes Care. American Diabetes Association; 2016;39:187–189.
Metformin hydrochloride extended-release tablets type of posting notice of intent to revise posting date. 2018.
Trenfield SJ, Goyanes A, Telford R, Wilsdon D, Rowland M, Gaisford S, et al. 3D printed drug products: non-destructive dose verification using a rapid point-and-shoot approach. Int J Pharm. Elsevier. 2018;549:283–92.
Gohel MC, Jogani PD. A review of co-processed directly compressible excipients. J Pharm Pharm Sci. 2005;8:76–93.
African Journal of Pharmacy and Pharmacology. Academic Journals; 2007.
Khaled SA, Burley JC, Alexander MR, Yang J, Roberts CJ. 3D printing of tablets containing multiple drugs with defined release profiles. Int J Pharm. 2015;494:643–50 Available from: http://linkinghub.elsevier.com/retrieve/pii/S0378517315300855. [cited 2018 Jul 13].
Okwuosa TC, Stefaniak D, Arafat B, Isreb A, Wan K-W, Alhnan MA. A lower temperature FDM 3D printing for the manufacture of patient-specific immediate release tablets. Pharm Res. 2016;33:2704–12.
Khaled SA, Alexander MR, Wildman RD, Wallace MJ, Sharpe S, Yoo J, et al. 3D extrusion printing of high drug loading immediate release paracetamol tablets. Int J Pharm. 2018;538:223–30.
McConnell EL, Liu F, Basit AW. Colonic treatments and targets: issues and opportunities. J Drug Target. 2009;17:335–63.
Gionchetti P, Praticò C, Rizzello F, Calafiore A, Capozzi N, Campieri M, et al. The role of budesonide-MMX in active ulcerative colitis. Expert Rev Gastroenterol Hepatol Taylor & Francis. 2014;8:215–22.
Wang W, Kang Q, Liu N, Zhang Q, Zhang Y, Li H, et al. Enhanced dissolution rate and oral bioavailability of Ginkgo biloba extract by preparing solid dispersion via hot-melt extrusion. Fitoterapia. 2015;102:189–97.
Jamroz W, Kurek M, Lyszczarz E, Brniak W, Jachowicz R. Printing techniques: recent developments in pharmaceutical technology. Acta Pol Pharm. 2017;74:753–63.
Lim SH, Kathuria H, Tan JJY, Kang L. 3D printed drug delivery and testing systems—a passing fad or the future? Adv Drug Deliv Rev. 2018;132:139–68.
Öblom H, Zhang J, Pimparade M, Speer I, Preis M, Repka M, et al. 3D-Printed isoniazid tablets for the treatment and prevention of tuberculosis—personalized dosing and drug release. AAPS PharmSciTech. Springer International Publishing. 2019;20:52.
Solutions F. POLYOXΚ™ water soluble resins combining flexibility with consistency.
Smith D, Kapoor Y, Hermans A, Nofsinger R, Kesisoglou F, Gustafson TP, et al. 3D printed capsules for quantitative regional absorption studies in the GI tract. Int J Pharm. 2018;550:418–28.
Ehtezazi T, Algellay M, Islam Y, Roberts M, Dempster NM, Sarker SD. The application of 3D printing in the formulation of multilayered fast dissolving oral films. J Pharm Sci Elsevier Ltd. 2018;107:1076–85.
Home—British Pharmacopoeia. 2017.
Goyanes A, Wang J, Buanz A, Martínez-Pacheco R, Telford R, Gaisford S, et al. 3D printing of medicines: engineering novel oral devices with unique design and drug release characteristics. Mol Pharm American Chemical Society. 2015;12:4077–84.
Patil H, Tiwari RV, Repka MA. Hot-melt extrusion: from theory to application in pharmaceutical formulation. AAPS PharmSciTech. 2016;17:20–42.
Shah S, Repka MA. Melt extrusion in drug delivery: three decades of progress. New York: Springer; 2013. p. 3–46.
Jamróz W, Kurek M, Łyszczarz E, Szafraniec J, Knapik-Kowalczuk J, Syrek K, et al. 3D printed orodispersible films with aripiprazole. Int J Pharm. Elsevier. 2017;533:413–20.
Genina N, Holländer J, Jukarainen H, Mäkilä E, Salonen J, Sandler N. Ethylene vinyl acetate (EVA) as a new drug carrier for 3D printed medical drug delivery devices. Eur J Pharm Sci. Elsevier. 2016;90:53–63.
Melocchi A, Parietti F, Maroni A, Foppoli A, Gazzaniga A, Zema L. Hot-melt extruded filaments based on pharmaceutical grade polymers for 3D printing by fused deposition modeling. Int J Pharm. Elsevier. 2016;509:255–63.
Qi S, Craig D. Recent developments in micro- and nanofabrication techniques for the preparation of amorphous pharmaceutical dosage forms. Adv Drug Deliv Rev. Elsevier. 2016;100:67–84.
Attaran M. The rise of 3-D printing: the advantages of additive manufacturing over traditional manufacturing. Bus Horiz. Elsevier. 2017;60:677–88.
Gebhardt A, Hötter J-S, Gebhardt A, Hötter J-S. Rapid prototyping. Addit Manuf Hanser. 2016:291–352.
Pîrjan A, Petroşanu D-M. The impact of 3d printing technology on the society and economy. J Inf Syst Oper Manag. Romanian-American University. 2013;7:360–70.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pandey, M., Choudhury, H., Fern, J.L.C. et al. 3D printing for oral drug delivery: a new tool to customize drug delivery. Drug Deliv. and Transl. Res. 10, 986–1001 (2020). https://doi.org/10.1007/s13346-020-00737-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-020-00737-0