Abstract
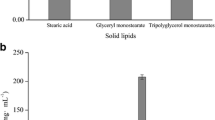
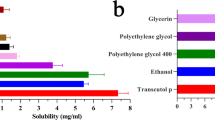
Niosomes are reported to increase the skin permeation and bioavailability of topically applied drug molecules. However, very few studies were reported for nanometer-sized niosome vesicles. The aim of the present study was to prepare minoxidil-loaded niosomal formulation using ethanol injection method. Surfactant screening showed that only Span 60, Span 20, and Tween 20 with cholesterol have capability of nano size vesicle formation. The formed niosomes were characterized for entrapment efficiency, vesicle size, scanning electron microscope, and physical stability. By modulation of surfactant and cholesterol ratio maximum entrapment up to 34.70 ± 1.1 % with size of 470 ± 27 nm was obtained (Span 60/cholesterol ratio of 1:2). The vesicle size obtained was between 150 and 800 nm that was depending on cholesterol ratio and type of nonionic surfactant employed. The in vitro skin permeation study showed that an increase in cholesterol concentration in niosome vesicles increases minoxidil skin retention. Niosome formulation prepared with 1:2 ratio of Span 60 and cholesterol showed 17.21 ± 3.2 % skin retention of minoxidil, which is more than eightfold as compared to control minoxidil gel.





Similar content being viewed by others
References
Baillie AJ, Florence AT, Hume LR, Muirhead GT, Rogerson A. The preparation and properties of niosomes non-ionic surfactant vesicles. J Pharm Pharmacol. 1985;37:863–8.
Azmin MN, Florence AT, Handjani-Vila RM, Stuart JFB, Vanlerberghe G, Whittaker JS. The effect of nonionic surfactant vesicle (niosome) entrapment on the absorption and distribution of methotrexate in mice. J Pharm Pharmacol. 1985;37:237–42.
Handjani-Vila RM, Rlbier A, Rondot B, Vanlerberghe G. Dispersion of lamellar phases of non-ionic lipids in cosmetic products. Int J Cosmetic Sci. 1979;1:303–14.
Toshimitsu Y, Florence AT. Vesicle (niosome)-in-water-in-oil (v/w/o) emulsions: an in vitro study. Int J Pharm. 1994;108(2):117–23.
Reddy DN, Udupa N. Formulation and evaluation of oral and transdermal preparations of flurbiprofen and piroxicam incorporated with different carriers. Drug Dev Ind Pharm. 1993;19:843–52.
Schreier H, Bouwstra J. Liposomes and niosomes as topical drug carriers: dermal and transdermal drug delivery. J Control Rel. 1994;30:1–15.
Bouwstra JA, Hofland HEJ. Niosomes. In: Kreuter J, editor. Colloidal drug delivery systems. New York: Marcell Dekker; 1994. p. 191–217.
Vanhal D, Vanrensen A, Devringer T, Junginger H, Bouwstra J. Diffusion of estradiol from non-ionic surfactant vesicles through human stratum-corneum in-vitro. STP Pharm Sci. 1996;6:72–8.
Manconi M, Sinico C, Valenti D, Lai F, Fadda AM. Niosomes as carriers for tretinoin III A study into the in vitro cutaneous delivery of vesicle-incorporated tretinoin. Int J Pharm. 2006;311:11–9.
Junginger HE, Hofland HEJ, Bouwstra JA. Liposomes and niosomes: interactions with human skin. Cosmet Toilet. 1991;106:45–50.
Touitou E, Junginger HE, Weiner ND, Nagai T, Mezei M. Liposomes as carriers for topical and transdermal delivery. J Pharm Sci. 1994;83:1189–203.
Tata S, Flynn GL, Weiner ND. Penetration of minoxidil from ethanol/propylene glycol solutions: effect of application volume and occlusion. J Pharm Sci. 1995;84:688–91.
Paul SU, Palo A, Yolanda PQ. Non crystalline minoxidil composition, its production and application. US Patent number 4828837; 1989.
Williams AC, Barry BW. Penetration enhancer. Adv Drug Deliv Rev. 2004;56:603–18.
Hagemann T, Schlutter-Bohmer B, Allam JP, Bieber T, Novak N. Contact Dermat. 2005;53:53–5.
Shaikh KS, Chellampillai B, Pawar AP. Studies on nonionic surfactant bilayer vesicles of ciclopiroxolamine. Drug Dev Ind Pharm. 2010;36(8):946–53.
Uchegbu IF, Vyas SP. Non-ionic surfactant based vesicles (niosomes) in drug delivery. Int J Pharm. 1998;172:33–70.
Yoshioka T, Sternberg B, Florence AT. Preparation and properties of vesicles (niosomes) of sorbitan monoesters (Span-20, Span-40, Span-60 and Span-80) and a sorbitan triester (Span-85). Int J Pharm. 1994;105:1–6.
Uchegbu IF, Florence AT. Non-ionic surfactant vesicles (niosomes)—physical and pharmaceutical chemistry. Adv Colloid Interface Sci. 1995;58:1–55.
Nasseri B. Effect of cholesterol and temperature on the elastic properties of niosomal membranes. Int J Pharm. 2005;300:95–101.
Sentjurc M, Vrhovnik K, Kristl J. Liposomes as a topical delivery system: the role of size on transport studied by the EPR imaging method. J Control Rel. 1999;59:87–97.
Acknowledgments
Authors are thankful to the Department of Biotechnology, India and the Council of Scientific and Industrial Research, India for funding the research. The authors are also thankful to AICTE NAFETIC for providing the facility to conduct the research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mali, N., Darandale, S. & Vavia, P. Niosomes as a vesicular carrier for topical administration of minoxidil: formulation and in vitro assessment. Drug Deliv. and Transl. Res. 3, 587–592 (2013). https://doi.org/10.1007/s13346-012-0083-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-012-0083-1