Abstract
Objective
To evaluate the efficacy of enalapril treatment on decline in glomerular filtration rate and reduction in proteinuria in children with chronic kidney disease (CKD).
Design
Open-label, randomized controlled trial.
Setting
Pediatric nephrology clinic at a tertiary-care referral hospital.
Intervention
Children with GFR between 15–60 mL/min/1.73 m2 were randomized to receive either enalapril at 0.4 mg/kg /day or no enalapril for 1 year.
Outcome measures
Change in GFR using 99mTc-DTPA and urine protein to creatinine ratio. Secondary outcomes included occurrence of composite outcome (30% decline in GFR or end stage renal disease) and systolic and diastolic blood pressure SDS during the study period.
Results
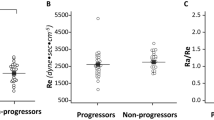
41 children were randomized into two groups; 20 received enalapril while 21 did not receive enalapril. During 1 year, GFR decline was not different in the two groups (regression coefficient (r) 0.40, 95% CI -4.29 to 5.09, P=0.86). The mean proteinuria reduction was 65% in the enalapril group, significantly higher than control group. The difference was significant even after adjustment for blood pressure was 198.5 (CI 97.5, 299.3; P<0.001). 3 (17.6%) patients in enalapril and 7 (36.8%) in nonenalapril group attained the composite outcome.
Conclusions
Enalapril is effective in reducing proteinuria in children with CKD and might be renoprotective in proteinuric CKD.
Similar content being viewed by others
References
Warady BA, Chadha V. Chronic kidney disease in children: the global perspective. Pediatr Nephrol. 2007;22:1999–2009.
Hari P, Singla IK, Mantan M, Kanitkar M, Batra B, Bagga A. Chronic renal failure in children. Indian Pediatr. 2003;40:1035–1042.
Bakris GL. Slowing nephropathy progression: focus on proteinuria reduction. Clin J Am Soc Nephrol. 2008;3:S3–10.
Wong CS, Pierce CB, Cole SR, Warady BA, Mak RH, Benador NM, et al. CKiD Investigators. Association of proteinuria with race, cause of chronic kidney disease, and glomerular filtration rate in the chronic kidney disease in children study. Clin J Am Soc Nephrol. 2009;4:812–819.
Ardissino G, Viganò S, Testa S, Daccò V, Paglialonga F, Leoni A, et al. Italkid Project. No clear evidence of ACEi efficacy on the progression of chronic kidney disease in children with hypodysplastic nephropathy-report from the ItalKid Project database. Nephrol Dial Transplant. 2007;22:2525–2530.
ESCAPE Trial Group, Wühl E, Trivelli A, Picca S, Litwin M, Peco-Antic A, Zurowska A, et al. Strict blood-pressure control and progression of renal failure in children. N Engl J Med. 2009;22:1639–1650.
Schwartz GJ, Haycock GB, Edelmann CM Jr, Spitzer A. A simple estimateof glomerular filtration rate in children derived from body length and plasma creatinine. Pediatrics. 1976;58:259–263.
National High Blood Pressure Education Program Working Group on High Blood Pressure in Children and Adolescents. The fourth report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents. Pediatrics. 2004;114:555–756.
Piepsz A, Colarinha P, Gordon I, Hahn K, Olivier P, Sixt R, et al. Guidelines for glomerular filtration rate determination in children. Eur J Nucl Med. 2001;28:BP31–BP36.
Romeo J. Evaluation of a kinetic method for creatinine. Lab Med. 1975;6:15–18.
Ruggenenti P, Perna A, Gherardi G, Garini G, Zoccali C, Salvadori M, et al. Renoprotective properties of ACE-inhibition in non-diabetic nephropathies with nonnephrotic proteinuria. Lancet. 1999;31:359–364.
Seeman T, Dusek J, Vondrák K, Flögelová H, Geier P, Janda J. Ramipril in the treatment of hypertension and proteinuria in children with chronic kidney diseases. Am J Hypertens. 2004;17:415–420.
Zeger SL, Liang KY. Longitudinal data analysis for discrete and continuous outcomes. Biometrics. 1986;42:121–130.
Ellis D, Vats A, Moritz ML, Reitz S, Grosso MJ, Janosky JE. Long-term antiproteinuric and renoprotective efficacy and safety of losartan in children with proteinuria. J Pediatr. 2003;143:89–97.
Van Dyck M, Proesmans W. Renoprotection by ACE inhibitors after severe hemolytic uremic syndrome. Pediatr Nephrol. 2004;19:688–690.
Litwin M, Grenda R, Sladowska J, Antoniewicz J. Add-on therapy with angiotensin II receptor 1 blocker in children with chronic kidney disease already treated with angiotensin-converting enzyme inhibitors. Pediatr Nephrol. 2006;21:1716–1722.
Ruggenenti P, Schieppati A, Remuzzi G. Progression, remission, regression of chronic renal diseases. Lancet. 2001;19:1601–1608.
Jafar TH, Schmid CH, Landa M, Giatras I, Toto R, Remuzzi G, et al. Angiotensin-converting enzyme inhibitors and progression of nondiabetic renal disease. A meta-analysis of patient-level data. Ann Intern Med. 2001;17:73–87.
Wingen AM, Fabian-Bach C, Schaefer F, Mehls O. Randomised multicentre study of a low-protein diet on the progression of chronic renal failure in children. European Study Group of Nutritional Treatment of Chronic Renal Failure in Childhood. Lancet. 1997;19:1117–1123.
Webb NJ, Shahinfar S, Wells TG, Massaad R, Gleim GW, Santoro EP, et al. Losartan and enalapril are comparable in reducing proteinuria in children. Kidney Int. 2012;Jun 27. doi: 10.1038/ki.2012.210.
Webb NJ, Lam C, Loeys T, Shahinfar S, Strehlau J, Wells TG, et al. Randomized, double-blind, controlled study of losartan in children with proteinuria. Clin J Am Soc Nephrol. 2010;5:417–424.
Webb NJ, Lam C, Shahinfar S, Strehlau J, Wells TG, Gleim GW, et al. Efficacy and safety of losartan in children with Alport syndrome-results from a subgroup analysis of a prospective, randomized, placebo- or amlodipine-controlled trial. Nephrol Dial Transplant. 2011;26:2521–2526.
Izuhara Y, Nangaku M, Inagi R, Tominaga N, Aizawa T, Kurokawa K, et al. Renoprotective properties of angiotensin receptor blockers beyond blood pressure lowering. J Am Soc Nephrol. 2005;16:3631–3641.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hari, P., Sahu, J., Sinha, A. et al. Effect of enalapril on glomerular filtration rate and proteinuria in children with chronic kidney disease: A randomized controlled trial . Indian Pediatr 50, 923–928 (2013). https://doi.org/10.1007/s13312-013-0262-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13312-013-0262-2