Abstract
Objective
To evaluate the effect of clofibrate for unconjugated hyperbilirubinemia in neonates.
Methods
A systematic review with meta-analysis of randomized controlled trials or quasi-randomized controlled trials was conducted to evaluate the clofibrate treatment in neonates with unconjugated hyperbilirubinemia. We followed the guidelines from the Cochrane review group and the PRISMA statement.
Results
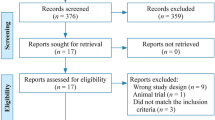
Of 148 studies identified, a total of 13 studies on 867 infants were included. A single oral administration of clofibrate was associated with decreased need of phototherapy (RR:.38, 95% CI: 0.21 to 0.68), shortened duration of phototherapy (mean duration: 23.88 h, 95% CI: 33.03 to −14.72 h) and reduced peak total serum bilirubin (mean duration: −1.62 mg/dL, 95% CI: 2.13 to −1.11 mg/dL). These effects were especially obvious in term infants and infants without hemolytic diseases. Data regarding mortality or kernicterus were not available from included studies.
Conclusions
Clofibrate may have short-term benefits for the infants with hyperbilirubinaemia, especially for population of term infants and infants without hemolytic diseases. Large RCTs with long-term followup are required to verify the safety of clofibrate and assess its long-term effects.
Similar content being viewed by others
References
Rennie J, Burman-Roy S, Murphy MS. Neonatal jaundice: summary of NICE guidance. BMJ. 2010;340:c2409.
Suresh GK, Martin CL, Soll RF. Metalloporphyrins for treatment of unconjugated hyperbilirubinemia in neonates. Cochrane Database Syst Rev. 2003:CD004207.
Steiner LA, Bizzarro MJ, Ehrenkranz RA, Gallagher PG. A decline in the frequency of neonatal exchange transfusions and its effect on exchange-related morbidity and mortality. Pediatrics. 2007;120:27–32.
Alcock GS, Liley H. Immunoglobulin infusion for isoimmune haemolytic jaundice in neonates. Cochrane Database Syst Rev. 2002:CD003313.
Cuperus FJ, Hafkamp AM, Hulzebos CV, Verkade HJ. Pharmacological therapies for unconjugated hyperbilirubinemia. Curr Pharm Dis. 2009;15:2927–2938.
Wang G, Shen H, Rajaraman G, Roberts MS, Gong Y, Jiang P, et al. Expression and antioxidant function of liver fatty acid binding protein in normal and bile-duct ligated rats. Eur J Pharmacol. 2007;560:61–68.
Lindenbaum A, Hernandorena X, Vial M, Benattar C, Janaud JC, Dehan M, et al. Clofibrate for the treatment of hyperbilirubinemia in neonates born at term: a double blind controlled study (author’s transl). Arch Fr Pediatr. 1981;38:867–873. (Article in French).
Cochrane Neonatal Review Group. Guidelines for Reviewers and Editors Available from: http://neonatalcochraneorg. Accessed October 1, 2010.
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–560.
Mantel N, Haenszel W. Statistical aspects of the analysis of data from retrospective studies of disease. J Natl Cancer Inst. 1959;22:719–748.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–188.
Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–634.
Lindenbaum A, Delaporte B, Benattar C, Dehan M, Magny JF, Gerbet D, et al. Preventive treatment of jaundice in premature newborn infants with clofibrate. Double-blind controlled therapeutic trial. Arch Fr Pediatr. 1985;42:759–763. (Article in French).
Flores Nava G, Vargas Perez C, Lopez Padilla M, Escobedo Chavez E. Clofibrate in the prevention of neonatal hyperbilirubinemia. Practica Pediatrica. 1996;5:40–46.
Mohammadzadeh A, Farhat A, Iranpour R. Effect of clofibrate in jaundiced term newborns. Indian J Pediatr. 2005;72:123–126.
Moslehi MA, Pishva N. Determination of effect of low dose vs moderate dose clofibrate on decreasing serum bilirubin in healthy term neonates. Iranian Journal of Pediatrics. 2007;17:108–112.
Eghbalian F, Pourhossein A, Zandevakili H. Effect of clofibrate in non-hemolytic indirect hyperbiliru-binemia in full term neonates. Indian J Pediatr. 2007;74:1003–1006.
Zahedpasha Y, Ahmadpour-Kacho M, Hajiahmadi M, Naderi S. Effect of clofibrate in jaundiced full-term infants:a randomized clinical trial. Arch Iran Med. 2007;10:349–353.
Badeli H, Sharafi R, Sajedi S. The effect of clofibrate on neonatal hyperbilirubinemia in uncomplicated jaundice. Iranian Journal of Pediatrics. 2008;18:20–24.
Mohammadzadeh A, Farhat A, Jafarzadeh M, Mirzarahimi M, Esmaili H, Amiri R. Prophylactic effect of clofibrate in low birth weight neonates, hyperbilirubinemia. J Chinese Clinical Med. 2008;3:140–144.
Zahedpasha Y, Ahmadpour-Kacho M, Hajiahmadi M, Naderi S, Kamali AA. Efficacy of clofibrate on severe neonatal jaundice associated with glucose-6-phosphate dehydrogenase deficiency (a randomized clinical trial). Southeast Asian J Trop Med Public Health. 2008;39:557–561.
Mohammadzadeh A, Farhat AS, Amiri R, Esmaely H, Bagheri S. Treatment effect of clofibrate in jaundiced low birth weight neonates. International J Hematol Oncol. 2009;19:100–105.
Sakha SH, Gharehbaghi MM, Rahbani ME. The effect of clofibrate with phototherapy in late pre-term newborns with non-hemolytic jaundice. Indian J Med Sci. 2009;63:174–179.
Sharafi R, Mortazavi Z, Sharafi S, Parashkouh R. The effect of clofibrate on decreasing serum bilirubin in healthy term neonates under home phototherapy. Iran J Pediatr. 2010;20:48–52.
Penna F, Bonelli G, Baccino FM, Costelli P. Cytotoxic properties of clofibrate and other peroxisome proliferators: relevance to cancer progression. Curr Med Chem. 2010;17:309–320.
Loomba RS, Arora R. Prevention of cardiovascular disease utilizing fibrates—a pooled meta-analysis. Am J Ther. 2010;17:e182–e188.
Fidaleo M. Human health risk assessment for peroxisome proliferators: more than 30 years of research. Exp Toxicol Pathol. 2009;61:215–221.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Xiong, T., Chen, D., Duan, Z. et al. Clofibrate for unconjugated hyperbilirubinemia in neonates: A systematic review . Indian Pediatr 49, 35–41 (2012). https://doi.org/10.1007/s13312-012-0012-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13312-012-0012-x