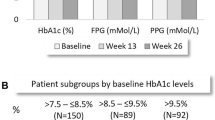
Abstract
Introduction
iGlarLixi (insulin glargine 100 U/mL plus lixisenatide) has demonstrated glycaemic efficacy and safety in adults with inadequately controlled type 2 diabetes mellitus (T2DM). Per the European Medicines Agency’s product label, iGlarLixi should be injected once a day within 1 h prior to a meal, preferably the same meal every day when the most convenient meal has been chosen. It is however unknown whether iGlarLixi administration timing affects glycaemic control and safety, as clinical trial evidence is mainly based on pre-breakfast iGlarLixi administration. Therefore, we assessed the effectiveness and safety of iGlarLixi in clinical practice, according to its administration timing.
Methods
Data were pooled from two prospective observational studies including 1303 European participants with T2DM inadequately controlled on oral antidiabetic drugs with or without basal insulin who initiated iGlarLixi therapy for 24 weeks. Participants were classified into four subgroups based on daily timing of iGlarLixi injection: pre-breakfast (N = 436), pre-lunch (N = 262), pre-dinner (N = 399), and those who switched iGlarLixi injection time during the study (N = 206).
Results
No meaningful differences in baseline characteristics were observed between the study groups. Least-squares mean reductions in haemoglobin A1c (HbA1c) from baseline to week 24 were substantial in all groups, with the numerically largest decrease observed in the pre-breakfast group (1.57%) compared with the pre-lunch (1.27%), pre-dinner (1.42%), or changed injection time (1.33%) groups. Pre-breakfast iGlarLixi injection also resulted in a numerically greater proportion of participants achieving HbA1c < 7.0% at week 24 (33.7% versus 19.0% for pre-lunch, 25.6% pre-dinner, and 23.2% changed injection time). iGlarLixi was well tolerated across all groups, with low rates of gastrointestinal disorders and hypoglycaemia. Mean body weight decreased similarly in all groups (by 1.3–2.3 kg).
Conclusion
iGlarLixi was effective and safe regardless of its daily administration time. However, pre-breakfast iGlarLixi injection resulted in a more effective glycaemic control.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
iGlarLixi (insulin glargine 100 U/mL plus lixisenatide) should be injected once daily within 1 h prior to a meal, preferably before the same meal every day, as per product label. |
It is however unknown whether the administration time of iGlarLixi affects glycaemic control and safety, as clinical trial evidence is mainly based on iGlarLixi administration before breakfast. |
By pooling results from two prospective observational studies in participants with type 2 diabetes inadequately controlled on oral antidiabetic drugs with or without basal insulin, we sought to evaluate in routine clinical practice the effectiveness and safety of iGlarLixi, according to its daily administration timing. |
What was learned from the study? |
iGlarLixi was effective and safe at all administration times, allowing participants flexibility in the timing of iGlarLixi administration to suit their lifestyle. |
However, pre-breakfast iGlarLixi injection was associated with a significantly greater HbA1c reduction compared to pre-lunch injection and changed injection timing, as well as a numerically larger HbA1c reduction compared to pre-dinner injection. |
Introduction
By exploiting complementary mechanisms of action, iGlarLixi, a titratable, once-daily, fixed-ratio combination of insulin glargine 100 U/mL (iGlar) plus lixisenatide, may represent an effective and safe option for therapy intensification in individuals with inadequately controlled type 2 diabetes mellitus (T2DM) [1,2,3]. Lixisenatide is a short-acting glucagon-like peptide 1 (GLP-1) receptor agonist that reduces postprandial plasma glucose (PPG) levels largely by delaying gastric emptying and decreasing postprandial glucagon levels. iGlar is a long-acting basal insulin analogue that primarily reduces fasting plasma glucose (FPG) [2]. iGlarLixi allows individuals with T2DM to achieve glycaemic control in a simple regimen, owing to its low injection burden and ease of use, without the need to increase measurements of self-monitoring blood glucose, which in turn may translate into better treatment adherence [2, 4].
On the basis of their potential benefits, fixed-ratio combinations of GLP-1 receptor agonists plus basal insulin are currently recommended in various clinical guidelines for use in individuals with T2DM inadequately controlled on basal insulin and/or oral antidiabetic drugs (OADs) [5, 6]. Indeed, the efficacy and safety of iGlarLixi has been consistently demonstrated in several large randomised controlled trials (RCTs) conducted in individuals with inadequately controlled T2DM, including the LixiLan clinical programme, consisting of LixiLan-O [7], LixiLan-L [8], and LixiLan-G [9], and more recently the SoliMix trial [10]. The LixiLan RCTs demonstrated robust glycaemic benefit with iGlarLixi versus iGlar, lixisenatide, or continuing prior GLP-1 receptor agonists, without an increased risk of hypoglycaemia [7,8,9]. iGlarLixi was also well tolerated and had a better gastrointestinal safety profile compared with lixisenatide alone and a more favourable body weight profile compared with iGlar alone [7, 8]. Similarly, in SoliMix, which compared iGlarLixi to a premix insulin analogue, biphasic insulin aspart 30 (BIAsp 30), once-daily iGlarLixi provided better glycaemic control with body weight benefit and less hypoglycaemia than twice-daily premix BIAsp 30 [10].
Despite extensive evidence from RCTs, there is currently limited data on the effectiveness and safety of iGlarLixi in routine clinical practice. It thus remains unknown whether the time of administration of iGlarLixi affects glycaemic control and safety, as in most RCTs, iGlarLixi was subcutaneously administered within 1 h before breakfast. However, in the product label of iGlarLixi, it is stated, without specifying the injection time, that iGlarLixi should be injected once a day within 1 h prior to a meal (or first meal as per US label) [11], preferably before the same meal every day, when the most convenient meal has been chosen [12]. By using pooled data from two real-world, prospective, observational studies [13, 14], we sought to evaluate in routine clinical practice the effectiveness and safety of iGlarLixi in individuals with T2DM inadequately controlled on OADs with or without basal insulin, according to its time of administration (i.e. before breakfast, lunch, dinner, or in case the time of the prandial injection was changed during the study period).
Methods
Study Design
This analysis was a part of the larger, comprehensive, European REALI project including pooled data from several multicentre, prospective, open-label studies reflecting clinical practice in different European countries. The aim of REALI was to evaluate the effectiveness and safety of different injectable glucose-lowering medications, particularly insulin glargine 300 U/mL and iGlarLixi, in unselected individuals with inadequately controlled T2DM defined as haemoglobin A1c (HbA1c) ≥ 7.5% (≥ 58.5 mmol/mol) [15,16,17].
The present analysis pooled patient-level data from two 24-week observational studies [13, 14] including adults with T2DM inadequately controlled on OADs with or without basal insulin who initiated iGlarLixi upon the treating physician-investigator’s decision. In both studies, iGlarLixi (Suliqua®, Sanofi, Paris, France) was self-administered subcutaneously once daily within 1 h prior to a meal (preferably the same meal every day) for 24 weeks, using one of the two SoloStar® pen injectors. The Suliqua® 30–60 (olive colour) pen, with a ratio of 3 units iGlar to 1 µg lixisenatide, contains 100 U/mL of iGlar and 33 μg/mL of lixisenatide and delivers dose steps between 30 to 60 units of iGlar in combination with 10–20 µg of lixisenatide. The Suliqua® 10–40 (peach colour) pen, with a ratio of 2 units iGlar to 1 µg lixisenatide, contains 100 U/mL of iGlar and 50 μg/mL of lixisenatide and delivers dose steps between 10 and 40 units of iGlar in combination with 5–20 µg of lixisenatide [12]. The choice of iGlarLixi pen and starting dose were left at the discretion of the treating physician-investigator. iGlarLixi was also titrated at the discretion of the treating physician. All participants recorded the daily time of iGlarLixi injection.
For the purpose of this pooled analysis, participants were classified into four subgroups based on the time of the day of iGlarLixi injection: pre-breakfast, pre-lunch, pre-dinner, and changed injection time during the study period.
Ethics
Both pooled studies [13, 14] were conducted according to the principles of the Declaration of Helsinki and the Good Clinical Practice guidelines, and were approved by the relevant institutional review boards/ethics committees. This pooled analysis was also performed in accordance with the Helsinki Declaration of 1964 and its later amendments. All participants gave written informed consent. Before data pooling, all patient information was de-identified. Moreover, this analysis did not involve primary data collection. Consequently, no ethical approval was required for this pooled analysis.
Data Collection and Assessments
Study-related data were collected at baseline, at 12 weeks and at 24 weeks. Baseline demographics and clinical characteristics in this analysis included age, sex, duration of diabetes, body weight and/or body mass index (BMI), diabetic complications and cardiovascular comorbidities, and details of prior glucose-lowering medications. Data on iGlarLixi treatment, such as iGlarLixi dose, timing of injection, used pen, and concomitant use of other glucose-lowering medications were also collected during the study period.
The primary endpoint of this analysis was the change in HbA1c from baseline to week 24. Secondary effectiveness endpoints included HbA1c change from baseline to week 12, proportions of participants achieving HbA1c targets of < 7.0% (< 53 mmol/mol), < 7.5% (< 58.5 mmol/mol) and < 8.0% (< 63.9 mmol/mol) at week 24, and changes from baseline to weeks 12 and 24 in FPG and 2-h PPG. Two-hour PPG was however collected in only one of the two pooled studies [13]. Safety endpoints included the incidence of hypoglycaemic events (symptomatic and severe) and gastrointestinal adverse events (AEs). During the 24-week treatment period, hypoglycaemic events were reported as percentages of participants with at least one event and as annualised rates (events per patient-year), and were defined on the basis of the American Diabetes Association classification [18]. The pooled analysis also evaluated changes in body weight and in iGlar dose provided by iGlarLixi (expressed in both U/day and in U/kg/day) from baseline to weeks 12 and 24.
Data Analysis
Data are expressed as mean ± standard deviation (SD) or as median (quartile 1–quartile 3) for continuous variables and as counts and percentages for categorical variables. The change in HbA1c from baseline was described using a mixed model for repeated measures (MMRM) with fixed effects of study, visit, subgroup category (pre-breakfast, pre-lunch, pre-dinner, and changed time of iGlarLixi injection), prior insulin use (insulin-naïve or insulin pre-treated), baseline HbA1c, age, baseline BMI, subgroup category-by-visit interaction, prior insulin use-by-visit interaction, baseline HbA1c value-by-visit interaction, age-by-visit interaction, and baseline BMI-by-visit interaction. On the basis of this MMRM, we estimated the least-squares (LS) mean HbA1c changes from baseline to weeks 12 and 24 with the corresponding 95% confidence intervals (CIs) for each subgroup.
All other effectiveness and safety endpoints as well as baseline characteristics were assessed descriptively. No imputation of missing data was performed, and no adjustment for multiple testing was made. All statistical tests were two-sided, with a p value of < 0.05 considered statistically significant. All analyses were performed using SAS version 9.4 (SAS Institute Inc, Cary, NC, USA).
Results
Participants
The pooled study population comprised 1303 adults with T2DM who were treated with iGlarLixi for 24 weeks. Of these participants, 436 (33.5%) self-administered iGlarLixi before breakfast, 262 (20.1%) before lunch, 399 (30.6%) before dinner, and 206 (15.8%) switched the time of iGlarLixi injection during the study period. Overall, there were no meaningful differences in baseline characteristics between the four study groups (Table 1). Participants had a mean age of 61.0 years, a mean BMI of 32.2 kg/m2, and a median diabetes duration of 9.0 years. A total of 590 participants (45.3%) were previously treated with basal insulin for a median duration of 2.5 years, with insulin glargine being the most common (67.8%) prior basal insulin used at baseline. More than half of the study population (56.7%) previously received only one OAD. Except for metformin whose use remained stable during the 24-week observation period (administered in 98% of patients), there was a reduction in the use of all other OADs (Electronic Supplementary Material Table S1).
Glycaemic Control
In the overall study population, mean ± SD HbA1c decreased from 9.11% ± 1.37% (76.04 ± 15.02 mmol/mol) at baseline to 7.70% ± 1.22% (60.65 ± 13.32 mmol/mol) at week 24, corresponding to a LS mean change in HbA1c from baseline to week 24 of − 1.43% (95% CI − 1.50 to − 1.36%). At week 24, pre-breakfast iGlarLixi injection resulted in significantly greater LS mean reductions in HbA1c compared to pre-lunch injection (− 1.57% versus − 1.27%; LS mean difference of 0.30%; p = 0.002) or changed injection time (− 1.33%; LS mean difference of 0.24%; p = 0.02). The pre-dinner group showed a LS mean reduction in HbA1c from baseline to week 24 of − 1.42% (LS mean difference of 0.15% compared to the pre-breakfast group; p = 0.08) (Fig. 1). At week 12, the LS mean change in HbA1c from baseline was − 1.15% (95% CI − 1.21 to − 1.08%) in the overall study population, ranging from − 0.94% in the pre-lunch group to − 1.30% in the pre-breakfast group. Compared to other study groups, pre-breakfast iGlarLixi injection also resulted in greater proportions of participants achieving HbA1c targets of < 7.0%, < 7.5%, and < 8.0% at week 24 (Fig. 2). There were however no noteworthy differences in the changes in FPG (Table 2; Electronic Supplementary Material Table S2) and in 2-h PPG (Electronic Supplementary Material Table S3) from baseline to week 24 between the four study groups.
Mean haemoglobin A1c (HbA1c) (%) over the 24-week study period according to iGlarLixi daily time of administration. N refers to the number of patients with available data at each timepoint. *Correspond to least-squares mean change (95% confidence interval) in HbA1c from baseline to week 24 derived from an adjusted mixed model for repeated measures (MMRM)
Safety
iGlarLixi was well tolerated in all study groups, with overall low reported rates of gastrointestinal AEs and of hypoglycaemic events (Table 3). Mean ± SD body weight also showed a decrease from baseline to weeks 12 and 24 in all four groups. In the total study population, the mean ± SD change in body weight from baseline to week 24 was − 1.8 ± 4.6 kg (Table 4). iGlarLixi dose titration occurred primarily in the first 12 weeks of the study. The mean ± SD dose of iGlar increased from 18.9 ± 9.3 U/day (0.21 ± 0.11 U/kg/day) at baseline to 29.8 ± 11.2 U/day (0.34 ± 0.13 U/kg/day) at week 12 and 33.3 ± 12.7 U/day (0.38 ± 0.14 U/kg/day) at week 24, with comparable changes across study groups (Table 4).
Discussion
In individuals with T2DM, PPG levels typically peak within 2 h after the start of a meal [19]. Hence, given the mode of action of lixisenatide, which specifically decreases post-meal hyperglycaemia, iGlarLixi should be injected within 1 h before a meal, and preferably the main/largest meal [19, 20]. In support of this recommendation, the present pooled analysis, performed in 1303 European adults with T2DM inadequately controlled on OADs with or without basal insulin, demonstrates that iGlarLixi is effective at all administration times during the day. Our findings support flexibility in the timing of iGlarLixi administration, which may be of benefit to both patients and healthcare providers. For instance, flexibility in iGlarLixi administration can improve patient adherence by suiting their lifestyle and can simplify treatment modalities particularly for challenging patient populations with long-standing T2DM or other comorbidities, leading to overall improved health-related quality of life [21, 22]. The favourable safety profile of iGlarLixi across all study groups of this analysis, reflected by its beneficial effect on body weight, the absence of serious AEs, and the occurrence of very few AEs leading to iGlarLixi discontinuation, may further enhance adherence to iGlarLixi therapy.
In line with the reported effectiveness and safety of iGlarLixi at all administration times in the current analysis, two 24-week RCTs, evaluating lixisenatide injected once daily at 20 µg in individuals with T2DM inadequately controlled on metformin, demonstrated that the efficacy and safety of lixisenatide do not vary depending on whether it is administered before breakfast, lunch, or dinner [23, 24]. Similarly, in a more recent in silico simulation study comparing the effect of iGlarLixi administration before either breakfast or an evening meal on blood sugar profiles, both regimens were observed to have acceptable glucose level variability and comparable efficacy, with low hypoglycaemia rates in the simulation [25]. A comparable percentage of time over 24 h was spent with blood glucose levels between 70 and 180 mg/dL when iGlarLixi was administered pre-breakfast or pre-evening (73% versus 71%, respectively) [25]. Despite our analysis’ encouraging effectiveness findings, there was a lower percentage of study participants achieving HbA1c target of < 7.0% (26.4%) when compared with the percentage of iGlarLixi-treated participants who achieved HbA1c < 7.0% (54.9% in LixiLan-L to 73.7% in LixiLan-O) at the end of the LixiLan trials performed in individuals with T2DM inadequately controlled on OADs and/or basal insulin [7,8,9]. Corresponding to real-life clinical practice, no forced titration of iGlarLixi was followed in the two pooled studies [13, 14]. Hence, less stringent titration may explain the lower percentage of participants reaching HbA1c targets in the present pooled analysis compared to the LixiLan RCTs.
Although our overall findings confirm the effectiveness and safety of iGlarLixi regardless of its daily administration time, pre-breakfast iGlarLixi injection was associated with a significantly greater HbA1c reduction compared to pre-lunch injection and changed injection timing but not compared to pre-dinner injection. Hence, pre-breakfast iGlarLixi injection may be preferable if it is convenient for the individuals living with T2DM, with their lifestyle and their typical main/largest meal remaining the most important factors when choosing the timing of the iGlarLixi injection [20]. Morning administration of iGlarLixi is also supported by the facts that PPG levels are typically highest after breakfast in most individuals and that iGlarLixi can cover PPG elevations after two meals if the gap between the two meals is less than 4–5 h. Thus, for pre-breakfast iGlarLixi administration, post-breakfast and post-lunch blood glucose levels are anticipated to be controlled by iGlarLixi assuming a time interval between the two meals of less than 4–5 h [20]. Such benefit may not be seen with a pre-dinner iGlarLixi administration since the time interval between dinner and breakfast is usually much longer than 4–5 h.
To the best of our knowledge, this work represents the first analysis in which the daily administration time of iGlarLixi was prospectively recorded and data regarding glycaemic control were systematically collected and analysed. In such a way, our study addresses the clinical question concerning the impact of iGlarLixi administration timing on its effectiveness and safety. Among other strengths of this analysis are the large data set coming from clinical practice and the analytical methods used to assess the change in HbA1c. Indeed, the change in HbA1c from baseline to week 24 was evaluated using a MMRM that adjusted for several factors including baseline HbA1c, age, baseline BMI, and prior insulin use. Despite this adjustment, caution is nevertheless advised when interpreting the differences in HbA1c reduction between the study groups, given the influence of unmeasured confounding factors. This pooled analysis also has the limitation of the relatively short treatment duration. In addition, there is a potential reporting bias, including missing data, inherent to real-world studies, which may underestimate incidences of AEs including hypoglycaemia. It should be noted that since this is an analysis of European data, our results may not be generalisable to other patient populations, as it is possible that patients’ management and response to iGlarLixi therapy could differ in other healthcare systems and may be affected by culture and ethnicity [26]. Overall, our data are reassuring in that iGlarLixi was effective and safe, irrespective of its administration time. These results strongly support the use of iGlarLixi in a patient-centred approach tailored to patient preferences and meal patterns.
Conclusions
In European people with T2DM inadequately controlled on OADs with or without basal insulin, iGlarLixi was effective and safe regardless of its daily administration time. However, pre-breakfast iGlarLixi injection may be preferable when there is a choice, as it was associated with numerically greater HbA1c reductions compared to other administration times. These data add to the body of evidence on the optimal use of iGlarLixi in clinical practice.
References
Skolnik N, Del Prato S, Blonde L, Galstyan G, Rosenstock J. Translating iGlarLixi evidence for the management of frequent clinical scenarios in type 2 diabetes. Adv Ther. 2021;38(4):1715–31.
Giorgino F, Caruso I, Napoli R. Titratable fixed-ratio combination of insulin glargine plus lixisenatide: a simplified approach to glycemic control in type 2 diabetes mellitus. Diabetes Res Clin Pract. 2020;170: 108478.
Novodvorský P, Haluzík M. An update on the safety of insulin-GLP-1 receptor agonist combinations in type 2 diabetes mellitus. Expert Opin Drug Saf. 2022;21(3):349–61.
McCrimmon RJ, Al Sifri S, Emral R, et al. Advancing therapy with iGlarLixi versus premix BIAsp 30 in basal insulin-treated type 2 diabetes: design and baseline characteristics of the SoliMix randomized controlled trial. Diabetes Obes Metab. 2021;23(6):1221–31.
Davies MJ, D’Alessio DA, Fradkin J, et al. Management of hyperglycemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2018;41(12):2669–701.
American Diabetes Association Professional Practice Committee, American Diabetes Association Professional Practice Committee, Draznin B, Aroda VR, et al. 9. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes-2022. Diabetes Care. 2022;45(Supplement 1):S125–43.
Rosenstock J, Aronson R, Grunberger G, et al. Benefits of LixiLan, a titratable fixed-ratio combination of insulin glargine plus lixisenatide, versus insulin glargine and lixisenatide monocomponents in type 2 diabetes inadequately controlled on oral agents: the LixiLan-O randomized trial. Diabetes Care. 2016;39(11):2026–35.
Aroda VR, Rosenstock J, Wysham C, et al. Efficacy and safety of LixiLan, a titratable fixed-ratio combination of insulin glargine plus lixisenatide in type 2 diabetes inadequately controlled on basal insulin and metformin: the LixiLan-L randomized trial. Diabetes Care. 2016;39(11):1972–80.
Blonde L, Rosenstock J, Del Prato S, et al. Switching to iGlarLixi versus continuing daily or weekly GLP-1 RA in type 2 diabetes inadequately controlled by GLP-1 RA and oral antihyperglycemic therapy: the LixiLan-G randomized clinical trial. Diabetes Care. 2019;42(11):2108–16.
Rosenstock J, Emral R, Sauque-Reyna L, et al. Advancing therapy in suboptimally controlled basal insulin-treated type 2 diabetes: clinical outcomes with iGlarLixi versus premix BIAsp 30 in the SoliMix randomized controlled trial. Diabetes Care. 2021;44(10):2361–70.
Food and Drug Administration. Soliqua® 100/33: Prescribing Information. 2021. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/208673s011lbl.pdf. Accessed 8 Aug 2022.
European Medicines Agency. Suliqua: EPAR-Product information. 2022. https://www.ema.europa.eu/en/documents/product-information/suliqua-epar-product-information_en.pdf. Accessed 4 Mar 2022.
Kis JT, Nagy G, Kovacs G. Effectiveness of iGlarLixi, a fixed-ratio combination of insulin glargine 100 U/mL and lixisenatide, in people with type 2 diabetes. Diabetes Ther. 2021;12(9):2517–29.
Bala C, Cerghizan A, Mihai BM, Moise M, Guja C. Real-world evidence on the use of a fixed-ratio combination of insulin glargine and lixisenatide (iGlarLixi) in people with suboptimally controlled type 2 diabetes in Romania: a prospective cohort study (STAR.Ro). BMJ Open. 2022;12(5):e060852.
Freemantle N, Bonadonna RC, Gourdy P, et al. Rationale and methodology for a European pooled analysis of postmarketing interventional and observational studies of insulin glargine 300 U/mL in diabetes: protocol of REALI project. BMJ Open. 2020;10(4):e033659.
Bonadonna RC, Mauricio D, Müller-Wieland D, et al. Impact of age on the effectiveness and safety of insulin glargine 300 U/mL: results from the REALI European pooled data analysis. Diabetes Ther. 2021;12(4):1073–97.
Gourdy P, Bonadonna RC, Freemantle N, et al. Does gender influence the effectiveness and safety of insulin glargine 300 U/ml in patients with uncontrolled type 2 diabetes? Results from the REALI European pooled analysis. Diabetes Ther. 2022;13(1):57–73.
Seaquist ER, Anderson J, Childs B, et al. Hypoglycemia and diabetes: a report of a workgroup of the American Diabetes Association and the Endocrine Society. Diabetes Care. 2013;36(5):1384–95.
Kapitza C, Forst T, Coester HV, Poitiers F, Ruus P, Hincelin-Méry A. Pharmacodynamic characteristics of lixisenatide once daily versus liraglutide once daily in patients with type 2 diabetes insufficiently controlled on metformin. Diabetes Obes Metab. 2013;15(7):642–9.
Haluzík M, Flekač M, Lengyel C, et al. Expert opinion on the therapeutic use of the fixed-ratio combination of insulin glargine 100 U/mL and lixisenatide: a central/eastern European perspective. Diabetes Ther. 2020;11(4):1029–43.
Peyrot M, Barnett AH, Meneghini LF, Schumm-Draeger PM. Factors associated with injection omission/non-adherence in the Global Attitudes of Patients and Physicians in Insulin Therapy study. Diabetes Obes Metab. 2012;14(12):1081–7.
Davies MJ, Gagliardino JJ, Gray LJ, Khunti K, Mohan V, Hughes R. Real-world factors affecting adherence to insulin therapy in patients with type 1 or type 2 diabetes mellitus: a systematic review. Diabet Med. 2013;30(5):512–24.
Ahrén B, Leguizamo Dimas A, Miossec P, Saubadu S, Aronson R. Efficacy and safety of lixisenatide once-daily morning or evening injections in type 2 diabetes inadequately controlled on metformin (GetGoal-M). Diabetes Care. 2013;36(9):2543–50.
Ahrén B, Vorokhobina N, Souhami E, Demil N, Ye J, Aronson R. Equal improvement in glycaemia with lixisenatide given before breakfast or the main meal of the day. J Diabetes Complicat. 2014;28(5):735–41.
Gautier T, Silwal R, Saremi A, Boss A, Breton MD. Modeling the effect of subcutaneous lixisenatide on glucoregulatory endocrine secretions and gastric emptying in type 2 diabetes to simulate the effect of iGlarLixi administration timing on blood sugar profiles. J Diabetes Sci Technol. 2022;16(2):428–33.
Dailey G, Bajaj HS, Dex T, Groleau M, Stager W, Vinik A. Post hoc efficacy and safety analysis of insulin glargine/lixisenatide fixed-ratio combination in North American patients compared with the rest of world. BMJ Open Diabetes Res Care. 2019;7(1): e000581.
Acknowledgements
Funding
This study (including the Rapid Service fee/Open Access fee) was funded by Sanofi (Paris, France).
Medical Writing Support
Medical writing support in accordance with Good Publication Practice (GPP3) guidelines (http://www.ismpp.org/gpp3) was provided by Thomas Rohban, MD, and Magalie El Hajj, PharmD, of Partner 4 Health (Paris, France) and was funded by Sanofi.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Authors’ Contributions
All authors contributed to the project design and the analysis plan. Mathilde Tournay performed the statistical analysis of the data. All authors were involved in the interpretation of the data, writing and reviewing drafts of the manuscript, and approved the final version for submission.
Prior Presentation
Preliminary results were presented at the American Diabetes Association 82nd Scientific Sessions, New Orleans, Louisiana, June 3–7, 2022.
Disclosures
Martin Haluzík has received honoraria for talks and/or consultancy and/or research funding from Eli Lilly, Novo Nordisk, Sanofi, AstraZeneca, Mundipharma, Bristol-Meyers Squibb, Amgen, Boehringer Ingelheim, Janssen, and Johnson & Johnson. Jochen Seufert has received honoraria for talks and/or consultancy and/or research funding from Apitope, AstraZeneca, Bayer, Berlin Chemie, Boehringer Ingelheim, Bristol-Meyers Squibb, Eli Lilly, GI-Dynamics, GlaxoSmithKline, Intarcia, Ipsen, Janssen, LifeScan, MedScape, MSD, Novartis, Novo Nordisk, OmniaMed, Pfizer, Roche, Sanofi, Servier, Takeda, and Ypsomed. Cristian Guja has participated in scientific advisory boards for and received consulting fees from AstraZeneca, Boehringer Ingelheim, Eli Lilly, KRKA, Merck KGaA, MSD, Novo Nordisk, Sanofi, and Servier. Mireille Bonnemaire is a Sanofi employee. Gregory Bigot is an IVIDATA employee. Mathilde Tournay is an IDDI employee, and has acted as a biostatistics contractor for Sanofi. János Tibor Kis has received research funding from Sanofi. Nick Freemantle has received research support and has acted as a consultant for Allergan, Ipsen, Sanofi, AstraZeneca, Vertex, Aimmune, ALK, Gedeon Richter, Abbott Singapore, Galderma, Thea, and Novartis.
Compliance with Ethics Guidelines
This analysis did not involve primary data collection by the authors; consequently, ethical approval was not required. Both included studies were approved by the appropriate ethics committees and were conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines.
Data Availability
The data sets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Haluzík, M., Seufert, J., Guja, C. et al. Effectiveness and Safety of iGlarLixi (Insulin Glargine 100 U/mL Plus Lixisenatide) in Type 2 Diabetes According to the Timing of Daily Administration: Data from the REALI Pooled Analysis. Diabetes Ther 14, 639–652 (2023). https://doi.org/10.1007/s13300-023-01375-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-023-01375-8