Abstract
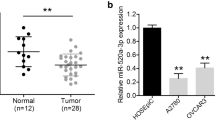
A growing body of evidence suggests that microRNA-494 (miR-494) could act as tumor-suppressive or oncogenic microRNAs (miRNAs) in different types of tumors. However, the biological roles and underlying mechanisms of miR-494 remain unknown in human epithelial ovarian carcinoma (EOC). Therefore, the aims of this study were to investigate the miR-494 expression and the significance of its clinical diagnosis in patients suffering EOC and to analyze its role and underlying molecular mechanism on the carcinogenesis of EOC. Here, we found that miR-494 was significantly decreased in EOC cell lines and tissues and its expression was negatively correlated with advanced International Federation of Gynecology and Obstetrics (FIGO) stage, high pathological grade, and lymph node metastasis (all P < 0.01). Functional studies showed that overexpression of miR-494 in EOC cells could remarkably inhibit proliferation, colony formation, migration, and invasion and induce cell apoptosis, G0/G1 phase arrest. An in vivo analysis revealed that the overexpression of miR-494 suppressed tumor growth in a nude mouse xenograft model system. Bioinformatic assay and dual-luciferase assay confirmed that insulin-like growth factor 1 receptor (IGF1R) was as a direct target of miR-494 in EOC cells. Western blot assay showed that overexpression of miR-494 inhibited IGF1R expression and its downstream signal protein expression. In addition, downregulation of IGF1R has similar effects with miR-494 overexpression on EOC cells and overexpression of IGF1R effectively rescued the inhibition of overexpressed miR-494 in EOC cells. These data suggested that miR-494 functions as a tumor suppressor in EOC by targeting IGF1R.







Similar content being viewed by others
Change history
21 December 2021
This article has been retracted. Please see the retraction notice for more detail: https://doi.org/10.3233/TUB-219010
References
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30.
Permuth-Wey J, Sellers TA. Epidemiology of ovarian cancer. Methods Mol Biol. 2009;472:413–37.
Legge F, Ferrandina G, Salutari V, Scambia G. Biological characterization of ovarian cancer: prognostic and therapeutic implications. Ann Oncol. 2005;16:95–101.
Coleman RL, Monk BJ, Sood AK, Herzog TJ. Latest research and treatment of advanced-stage epithelial ovarian cancer. Nat Rev Clin Oncol. 2013;10:211–24.
Miska EA. How microRNAs control cell division, differentiation and death. Curr Opin Genet Dev. 2005;15:563.
Ambros V. The functions of animal microRNAs. Nature. 2004;431:350–5.
Brennecke J, Hipfner DR, Stark A, Russell RB, Cohen SM. Bantam encodes a developmentally regulated microRNA that controls cell proliferation and regulates the proapoptotic gene hid in Drosophila. Cell. 2003;113:25–36.
Sun K, Lai EC. Adult-specific functions of animal microRNAs. Nat Rev Genet. 2013;14:535–48.
Seton-Rogers S. Tumour suppressors: Hippo promotes microRNA processing. Nat Rev Cancer. 2014;14:216–7.
Shenouda SK, Alahari SK. MicroRNA function in cancer: oncogene or a tumor suppressor? Cancer Metastasis Rev. 2009;28:369–78.
Zhang X, Han C, He J: Research progress of oncogene and tumor suppressor gene in bladder cancer. Panminerva Med. 2015 (in press).
Diakos C, Zhong S, Xiao Y, Zhou M, Vasconcelos GM, Krapf G, et al. TEL-AML1 regulation of survivin and apoptosis via miRNA-494 and miRNA-320a. Blood. 2010;116:4885–93.
Duan H, Jiang Y, Zhang H, Wu Y. MiR-320 and miR-494 affect cell cycles of primary murine bronchial epithelial cells exposed to benzo[a]pyrene. Toxicol In Vitro. 2010;24:928–35.
Zhao JJ, Yang J, Lin J, Yao N, Zhu Y, Zheng J, et al. Identification of miRNAs associated with tumorigenesis of retinoblastoma by miRNA microarray analysis. Childs Nerv Syst. 2009;25:13–20.
Liu K, Liu S, Zhang W, Jia B, Tan L, Jin Z, et al. miR-494 promotes cell proliferation, migration and invasion, and increased sorafenib resistance in hepatocellular carcinoma by targeting PTEN. Oncol Rep. 2015;34:1003–10.
Lim L, Balakrishnan A, Huskey N, Jones KD, Jodari M, Ng R, et al. MicroRNA-494 within an oncogenic microRNA megacluster regulates G1/S transition in liver tumorigenesis through suppression of mutated in colorectal cancer. Hepatology. 2014;59:202–15.
Liu Y, Li X, Zhu S, Zhang JG, Yang M, Qin Q, et al. Ectopic expression of miR-494 inhibited the proliferation, invasion and chemoresistance of pancreatic cancer by regulating SIRT1 and c-Myc. Gene Ther. 2015;22:729–38.
Shen PF, Chen XQ, Liao YC, Chen N, Zhou Q, Wei Q, et al. MicroRNA-494-3p targets CXCR4 to suppress the proliferation, invasion, and migration of prostate cancer. Prostate. 2014;74:756–67.
Romano G, Acunzo M, Garofalo M, Di Leva G, Cascione L, Zanca C, et al. MiR-494 is regulated by ERK1/2 and modulates trail-induced apoptosis in non-small-cell lung cancer through BIM down-regulation. Proc Natl Acad Sci U S A. 2012;109:16570–5.
Kim WK, Park M, Kim YK, Tae YK, Yang HK, Lee JM, et al. MicroRNA-494 downregulates kit and inhibits gastrointestinal stromal tumor cell proliferation. Clin Cancer Res. 2011;17:7584–94.
Yamanaka S, Campbell NR, An F, Kuo SC, Potter JJ, Mezey E, et al. Coordinated effects of microRNA-494 induce G2/M arrest in human cholangiocarcinoma. Cell Cycle. 2012;11:2729–38.
Kim YW, Kim EY, Jeon D, Liu JL, Kim HS, Choi JW, et al. Differential microRNA expression signatures and cell type-specific association with taxol resistance in ovarian cancer cells. Drug Des Devel Ther. 2014;8:293–314.
Sanabria-Figueroa E, Donnelly SM, Foy KC, Buss MC, Castellino RC, Paplomata E, et al. Insulin-like growth factor-1 receptor signaling increases the invasive potential of human epidermal growth factor receptor 2-overexpressing breast cancer cells via Src-focal adhesion kinase and forkhead box protein M1. Mol Pharmacol. 2015;87:150–61.
King SM, Modi DA, Eddie SL, Burdette JE. Insulin and insulin-like growth factor signaling increases proliferation and hyperplasia of the ovarian surface epithelium and decreases follicular integrity through upregulation of the PI3-kinase pathway. J Ovarian Res. 2013;6:12.
Osaki LH, Gama P. MAPKs and signal transduction in the control of gastrointestinal epithelial cell proliferation and differentiation. Int J Mol Sci. 2013;14:10143–61.
Cao Z, Liu LZ, Dixon DA, Zheng JZ, Chandran B, Jiang BH. Insulin-like growth factor-I induces cyclooxygenase-2 expression via PI3K, MAPK and PKC signaling pathways in human ovarian cancer cells. Cell Signal. 2007;19:1542–53.
Gest C, Mirshahi P, Li H, Pritchard LL, Joimel U, Blot E, et al. Ovarian cancer: Stat3, RhoA and IGF-IR as therapeutic targets. Cancer Lett. 2012;317:207–17.
Gotlieb WH, Bruchim I, Gu J, Shi Y, Camirand A, Blouin MJ, et al. Insulin-like growth factor receptor I targeting in epithelial ovarian cancer. Gynecol Oncol. 2006;100:389–96.
Coppola D, Saunders B, Fu L, Mao W, Nicosia SV. The insulin-like growth factor 1 receptor induces transformation and tumorigenicity of ovarian mesothelial cells and down-regulates their Fas-receptor expression. Cancer Res. 1999;59:3264–70.
Xue M, Cao X, Zhong Y, Kuang D, Liu X, Zhao Z, et al. Insulin-like growth factor-1 receptor (IGF1R) kinase inhibitors in cancer therapy: advances and perspectives. Curr Pharm Des. 2012;18:2901–13.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
None
Additional information
This article has been retracted. Please see the retraction notice for more detail: https://doi.org/10.3233/TUB-219010
Manhua Cui and Jin He contributed equally to this work.
About this article
Cite this article
Li, N., Zhao, X., Wang, L. et al. RETRACTED ARTICLE: miR-494 suppresses tumor growth of epithelial ovarian carcinoma by targeting IGF1R. Tumor Biol. 37, 7767–7776 (2016). https://doi.org/10.1007/s13277-015-4603-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-4603-8