Abstract
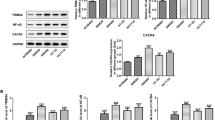
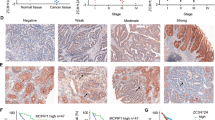
The nuclear factor (NF)-κB pathway has been implicated in colorectal cancer (CRC) tumorigenesis. Here, we investigated the role of a novel NIK- and IKKβ-binding protein (NIBP) in CRC metastasis through activation of the canonical NF-κB pathway. NIBP, p-p65, and matrix metalloproteases (MMPs) were assessed by immunohistochemistry in 114 CRC tissues, and the time to metastasis was recorded after surgery. Furthermore, the activity of the NF-κB pathway, MMP expression, and the metastatic potential of HT-29 cells overexpressing NIBP after treatment with the NF-κB inhibitor pyrrolidinecarbodithioic acid (PDTC) were examined in vitro and in vivo. NIBP-positive CRC exhibited a higher rate of metastasis, and the time to metastasis of NIBP-positive patients was shorter in the early tumor, lymph node, metastasis (TNM) stages (I and II), while NIBP and p-p65 expression was higher in later TNM stages (III and IV). However, there was no difference in terms of the positive rate of NIBP, p-p65, MMP-2, and serum carcinoembryonic antigen (CEA) level was no difference in the pathological type, gender, tumor location, or size. The NF-κB pathway, MMP-2 and MMP-9 activity, and cell motility and invasion were increased in NIBP-overexpressing cells, even after PDTC treatment. Moreover, these cells exhibited high metastasis in mice, and p-p65, MMP-2, and MMP-9 expression levels were elevated in the primary tumor and liver metastases. In conclusion, NIBP overexpression increases the CRC metastatic potential through activation of the NF-κB pathway and increasing MMP-2 and MMP-9 expression. In addition, NIBP overexpression, at least in part, may reduce inhibition of the canonical NF-κB pathway and MMPs caused by PDTC treatment.








Similar content being viewed by others
References
Siegel R, Desantis C, Jemal A. Colorectal cancer statistics, 2014. CA Cancer J Clin. 2014;64:104–17.
Gockel I, Sgourakis G, Lyros O, Polotzek U, Schimanski CC, Lang H, et al. Risk of lymph node metastasis in submucosal esophageal cancer: a review of surgically resected patients. Expert Rev Gastroenterol Hepatol. 2011;5:371–84.
Compton CC. Optimal pathologic staging: defining stage II disease. Clin Cancer Res. 2007;13:6862s–70s.
O’Connell JB, Maggard MA, Ko CY. Colon cancer survival rates with the new American Joint Committee on Cancer sixth edition staging. J Natl Cancer Inst. 2004;96:1420–5.
Markowitz SD, Itzkowitz SH, Berger BM. The effectiveness of colonoscopy in reducing mortality from colorectal cancer. Ann Intern Med. 2009;150:816–7. author reply 9-20.
Chowdhury S, Howell GM, Rajput A, Teggart CA, Brattain LE, Weber HR, et al. Identification of a novel TGFbeta/PKA signaling transduceome in mediating control of cell survival and metastasis in colon cancer. PLoS One. 2011;6:e19335.
Stein U, Walther W, Arlt F, Schwabe H, Smith J, Fichtner I, et al. MACC1, a newly identified key regulator of HGF-MET signaling, predicts colon cancer metastasis. Nat Med. 2009;15:59–67.
Vogelstein B, Fearon ER, Hamilton SR, Kern SE, Preisinger AC, Leppert M, et al. Genetic alterations during colorectal-tumor development. N Engl J Med. 1988;319:525–32.
Markowitz SD, Bertagnolli MM. Molecular origins of cancer: molecular basis of colorectal cancer. N Engl J Med. 2009;361:2449–60.
Vaiopoulos AG, Athanasoula K, Papavassiliou AG. NF-kappaB in colorectal cancer. J Mol Med (Berl). 2013;91:1029–37.
Vaiopoulos AG, Papachroni KK, Papavassiliou AG. Colon carcinogenesis: learning from NF-kappaB and AP-1. Int J Biochem Cell Biol. 2010;42:1061–5.
Chen LF, Greene WC. Shaping the nuclear action of NF-kappaB. Nat Rev Mol Cell Biol. 2004;5:392–401.
Hayden MS, Ghosh S. Shared principles in NF-kappaB signaling. Cell. 2008;132:344–62.
Hinz M, Arslan SC, Scheidereit C. It takes two to tango: IkappaBs, the multifunctional partners of NF-kappaB. Immunol Rev. 2012;246:59–76.
Orlowski RZ, Baldwin Jr AS. NF-kappaB as a therapeutic target in cancer. Trends Mol Med. 2002;8:385–9.
Chaturvedi MM, Sung B, Yadav VR, Kannappan R, Aggarwal BB. NF-kappaB addiction and its role in cancer: ‘One size does not fit all’. Oncogene. 2011;30:1615–30.
Said AH, Raufman JP, Xie G. The role of matrix metalloproteinases in colorectal cancer. Cancers (Basel). 2014;6:366–75.
Zhao Z, Lu P, Zhang H, Xu H, Gao N, Li M, et al. Nestin positively regulates the Wnt/beta-catenin pathway and the proliferation, survival and invasiveness of breast cancer stem cells. Breast Cancer Res. 2014;16:408.
Zong M, Satoh A, Yu MK, Siu KY, Ng WY, Chan HC, et al. TRAPPC9 mediates the interaction between p150 and COPII vesicles at the target membrane. PLoS One. 2012;7:e29995.
Zahoor MA, Yamane D, Mohamed YM, Suda Y, Kobayashi K, Kato K, et al. Bovine viral diarrhea virus non-structural protein 5a interacts with NIK- and IKKbeta-binding protein. J Gen Virol. 2010;91:1939–48.
Hu WH, Pendergast JS, Mo XM, Brambilla R, Bracchi-Ricard V, Li F, et al. NIBP, a novel NIK and IKK(beta)-binding protein that enhances NF-(kappa)B activation. J Biol Chem. 2005;280:29233–41.
Westlake CJ, Baye LM, Nachury MV, Wright KJ, Ervin KE, Phu L, et al. Primary cilia membrane assembly is initiated by Rab11 and transport protein particle II (TRAPPII) complex-dependent trafficking of Rabin8 to the centrosome. Proc Natl Acad Sci U S A. 2011;108:2759–64.
Zhang Y, Liu S, Wang H, Yang W, Li F, Yang F, et al. Elevated NIBP/TRAPPC9 mediates tumorigenesis of cancer cells through NFkappaB signaling. Oncotarget. 2015;6:6160–78.
Kim JC, Kim SY, Roh SA, Cho DH, Kim DD, Kim JH, et al. Gene expression profiling: canonical molecular changes and clinicopathological features in sporadic colorectal cancers. World J Gastroenterol. 2008;14:6662–72.
Blits B, Kitay BM, Farahvar A, Caperton CV, Dietrich WD, Bunge MB. Lentiviral vector-mediated transduction of neural progenitor cells before implantation into injured spinal cord and brain to detect their migration, deliver neurotrophic factors and repair tissue. Restor Neurol Neurosci. 2005;23:313–24.
Rubinson DA, Dillon CP, Kwiatkowski AV, Sievers C, Yang L, Kopinja J, et al. A lentivirus-based system to functionally silence genes in primary mammalian cells, stem cells and transgenic mice by RNA interference. Nat Genet. 2003;33:401–6.
Ongchin M, Sharratt E, Dominguez I, Simms N, Wang J, Cheney R, et al. The effects of epidermal growth factor receptor activation and attenuation of the TGFbeta pathway in an orthotopic model of colon cancer. J Surg Res. 2009;156:250–6.
Rajput A, Dominguez San Martin I, Rose R, Beko A, Levea C, Sharratt E, et al. Characterization of HCT116 human colon cancer cells in an orthotopic model. J Surg Res. 2008;147:276–81.
Kraus S, Nabiochtchikov I, Shapira S, Arber N. Recent advances in personalized colorectal cancer research. Cancer Lett. 2014;347:15–21.
Fletcher RH. Carcinoembryonic antigen. Ann Intern Med. 1986;104:66–73.
Ghosh S, Karin M. Missing pieces in the NF-kappaB puzzle. Cell. 2002;109(Suppl):S81–96.
Tran K, Merika M, Thanos D. Distinct functional properties of IkappaB alpha and IkappaB beta. Mol Cell Biol. 1997;17:5386–99.
Hayden MS, Ghosh S. Signaling to NF-kappaB. Genes Dev. 2004;18:2195–224.
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (No. 81260365), Guangdong Province Universities and Colleges Pearl River Scholar Funded Scheme (2011), Guangzhou Pilot Project of Clinical and Translational Research Center (Early Gastrointestinal Cancers, No. 7415696196402), and Guangdong Province Universities and Colleges Major National Training Project (No. 2014GKXM026).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
None
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Qin, M., Liu, S., Li, A. et al. NIK- and IKKβ-binding protein promotes colon cancer metastasis by activating the classical NF-κB pathway and MMPs. Tumor Biol. 37, 5979–5990 (2016). https://doi.org/10.1007/s13277-015-4433-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-4433-8