Abstract
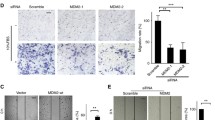
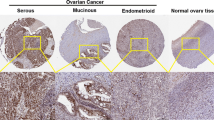
The high mobility group A2 (HMGA2), an oncofetal protein, was shown to play a role in tumor development and progression. However, the molecular and clinical role of HMGA2 in epithelial ovarian carcinomas (EOCs) is still unknown. In the present study, EOC cell line SKOV3 was subjected to in vitro assays. Here, our findings showed that HMGA2 was highly expressed in EOC cell line SKOV3. HMGA2 knockdown promoted cell apoptosis and the cleavage of caspase 3, and decreased the B cell lymphoma 2 (Bcl-2)/Bax ratio in SKOV3. Functionally, HMGA2 knockdown resulted in reduction of SKOV3 cell migration and invasion. Mechanically, HMGA2 knockdown affected the occurrence of EMT by increasing E-cadherin gene and protein expression and decreasing the gene and protein expression of N-cadherin, slug, and vimentin. At the same time, HMGA2 also repressed the expression of matrix metalloproteinase 2 (MMP2) and matrix metalloproteinase 9 (MMP9), which was consistent with the decreased invasion capacity. In conclusion, HMGA2 is associated with migration and invasiveness and regulates the progression of EMT in the development of EOC, and HMGA2 gene and protein may be a novel therapeutic target against EOC in the clinical practice.



Similar content being viewed by others
References
Jayson GC, Kohn EC, Kitchener HC, Ledermann JA. Ovarian cancer. Lancet. 2014;S0140–6736:62146–7.
Harter P, Hilpert F, Mahner S, Heitz F, Pfisterer J, du Bois A. Systemic therapy in recurrent ovarian cancer: current treatment options and new drugs. Expert Rev Anticancer Ther. 2010;10:81–8.
Burger RA, Brady MF, Bookman MA, Fleming GF, Monk BJ, Huang H, et al. Gynecologic Oncology Group Incorporation of bevacizumab in the primary treatment of ovarian cancer. N Engl J Med. 2011;365:2473–83.
Perren TJ, Swart AM, Pfisterer J, Ledermann JA, Pujade-Lauraine E, Kristensen G, et al. A phase 3 trial of bevacizumab in ovarian cancer. N Engl J Med. 2011;365:2484–96.
Chiappetta G, Ammirante M, Basile A, et al. The antiapoptotic protein BAG3 is expressed in thyroid carcinomas and modulates apoptosis mediated by tumor necrosis factor-related apoptosis-inducing ligand. J Clin Endocrinol Metab. 2007;92:1159–63.
Gentilella A, Passiatore G, Deshmane S, et al. Activation of BAG3 by Egr-1 in response to FGF-2 in neuroblastoma cells. Oncogene. 2008;27:5011–8.
Staibano S, Mascolo M, Benedetto M, et al. BAG3 protein delocalization in prostate carcinoma. Tumor Biol. 2010;31:461–9.
Wang HQ, Meng X, Gao Y-Y, et al. Characterization of BAG3 cleavage during apoptosis of pancreatic cancer cells. J Cell Physiol. 2010;224:94–100.
Suzuki M, Iwasaki M, Sugio A, et al. BAG3 (BCL2-associated athanogene 3) interacts with MMP-2 to positively regulate invasion by ovarian carcinoma cells. Cancer Lett. 2011;303:65–71.
Festa M, Del Valle L, Khalili K, et al. BAG3 protein is overexpressed in human glioblastoma and is a potential target for therapy. Am J Pathol. 2011;178:2504–12.
Rosati A, Graziano V, De Laurenzi V, et al. BAG3: a multifaceted protein that regulates major cell pathways. Cell Death Dis. 2011;7:141.
Falco A, Festa M, Basile A, et al. BAG3 controls angiogenesis through regulation of ERK phosphorylation. Oncogene. 2012;31:5153–61.
Li N, Du ZX, Zong ZH, et al. PKC[delta]-mediated phosphorylation of BAG3 at Ser187 site induces epithelial-mesenchymal transition and enhances invasiveness in thyroid cancer FRO cells. Oncogene. 2012;32:4539–48.
He J, Gu D, Wu X, et al. Major causes of death among men and women in China. N Engl J Med. 2005;353:1124–34.
Pang RC, Joh J, Johnson P, et al. Biology of hepatocellular carcinoma. Ann Surg Oncol. 2008;15:962–71.
Semenza GL. HIF-1 and tumor progression: pathophysiology and therapeutics. Trends Mol Med. 2002;8:S62–7.
Déry M-AC, Michaud MD, Richard DE. Hypoxia-inducible factor 1: regulation by hypoxic and non-hypoxic activators. Int J Biochem Cell Biol. 2005;37:535–40.
De A, De A, Papasian C, et al. Extract induces autophagy and inhibits human ovarian cancer cell proliferation, angiogenesis, growth of mouse xenograft tumors. PLoS One. 2013;8:e72748.
Kim KW, Paul P, Qiao J, et al. Enhanced autophagy blocks angiogenesis via degradation of gastrin-releasing peptide in neuroblastoma cells. Autophagy. 2013;9:1579–90.
Stetler-Stevenson WG, Aznavoorian S, Liotta LA. Tumor cell inter-actions with the extracellular matrix during invasion and metastasis. Annu Rev Cell Biol. 1993;9:541–73.
Acknowledgments
We greatly thank other members of Yang Lab for valuable suggestions and writing.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Additional information
The Publisher and Editor retract this article in accordance with the recommendations of the Committee on Publication Ethics (COPE). After a thorough investigation we have strong reason to believe that the peer review process was compromised.
An erratum to this article is available at http://dx.doi.org/10.1007/s13277-017-5487-6.
About this article
Cite this article
Yan, J., Zhang, Y., Shi, W. et al. RETRACTED ARTICLE: The critical role of HMGA2 in regulation of EMT in epithelial ovarian carcinomas. Tumor Biol. 37, 823–828 (2016). https://doi.org/10.1007/s13277-015-3852-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-3852-x