Abstract
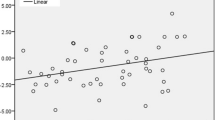
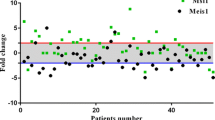
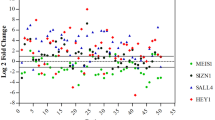
The three amino acid loop extension (TALE) class myeloid ecotropic viral integration site 1 (MEIS1) homeobox gene is known to play a crucial role in normal and tumor development. In contrast with its well-described cancer stemness properties in hematopoietic cancers, little is known about its role in solid tumors like esophageal squamous cell carcinoma (ESCC). Here, we analyzed MEIS1 expression and its clinical relevance in ESCC patients and also investigated its correlation with the SOX2 self-renewal master transcription factor in the ESCC samples and in the KYSE-30 ESCC cell line. MEIS1 mRNA and protein expression were significantly decreased in ESCC disease (P < 0.05). The inverse correlation between MEIS1 mRNA expression and tumor cell metastasis to the lymph nodes (P = 0.004) was significant. Also, MEIS1 protein levels inversely correlated to lymph node involvement (P = 0.048) and high tumor stage (stages III/IV, P = 0.030). The low levels of DNA methylation in the MEIS1 promoter showed that this suppression does not depend on methylation. We showed that downregulation of EZH2 restored MEIS1 expression significantly. Also, we investigated that MEIS1 downregulation is concomitant with increased SOX2 expression. To the best of our knowledge, this is the first report on the MEIS1 gene in ESCC. The inverse correlation of MEIS1 with metastasis, tumor staging, and the role of EZH2 in methylation, together with its correlation with stemness factor SOX2 expression, led us to predict cancer stemness properties for MEIS1 in ESCC.






Similar content being viewed by others
Abbreviations
- EC:
-
Esophageal carcinoma
- ESCC:
-
Esophageal squamous cell carcinoma
- ESC:
-
Embryonic stem cells
- CSC:
-
Cancer stem cell
- MEIS1:
-
Myeloid ecotropic viral integration site 1
- MLL:
-
Myeloid/lymphoid or mixed lineage leukemia
- MSP-PCR:
-
Methylation-specific PCR
- LSC:
-
Leukemia stem cell
- PBX:
-
Pre-B cell leukemia homeobox
- TALE:
-
Three amino acid loop extension
References
Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics. CA Cancer J Clin. 2005;55:74–108.
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90.
Kamangar F, Malekzadeh R, Dawsey SM, Saidi F. Esophageal cancer in Northeastern Iran: a review. Arch Iran Med. 2007;10:70–82.
Sadjadi A, Marjani H, Semnani S, Nasseri-Moghaddam S. Esophageal cancer in Iran: a review. Middle East J Cancer. 2010;1:11–20.
Chang AC, Ji H, Birkmeyer NJ, Orringer MB, Birkmeyer JD. Outcomes after transhiatal and transthoracic esophagectomy for cancer. Ann Thorac Surg. 2008;85:424–9.
Herman JG, Baylin SB. Gene silencing in cancer in association with promoter hypermethylation. New Engl J Med. 2003;349:2042–54.
Islam F, Gopalan V, Wahab R, Smith RA, Lam AK. Cancer stem cells in oesophageal squamous cell carcinoma: identification, prognostic and treatment perspectives. Crit Rev Oncol Hematol. 2015. doi:10.1016/j.critrevonc.2015.04.007.
Moskow JJ, Bullrich F, Huebner K, Daar IO, Buchberg AM. Meis1, a PBX1-related homeobox gene involved in myeloid leukemia in BXH-2 mice. Mol Cell Biol. 1995;15:5434–43.
Moens CB, Selleri L. Hox cofactors in vertebrate development. Dev Biol. 2006;291:193–206.
Penkov D, San Martín DM, Fernandez-Díaz LC, Rosselló CA, Torroja C, Sánchez-Cabo F, et al. Analysis of the DNA-binding profile and function of TALE homeoproteins reveals their specialization and specific interactions with Hox genes/proteins. Cell Rep. 2013;3:1321–33.
Dekel B, Metsuyanim S, Schmidt-Ott KM, Fridman E, Jacob-Hirsch J, Simon A, et al. Multiple imprinted and stemness genes provide a link between normal and tumor progenitor cells of the developing human kidney. Cancer Res. 2006;66:6040–9.
Cai M, Langer EM, Gill JG, Satpathy AT, Albring J, Wumesh KC, et al. Dual actions of Meis1 inhibit erythroid progenitor development and sustain general hematopoietic cell proliferation. Blood. 2012;120:335–46.
Hisa T, Spence SE, Rachel RA, Fujita M, Nakamura T, Ward JM, et al. Hematopoietic, angiogenic and eye defects in Meis1 mutant animals. EMBO J. 2004;23:450–9.
Tucker ES, Lehtinen MK, Maynard T, Zirlinger M, Dulac C, Rawson N, et al. Proliferative and transcriptional identity of distinct classes of neural precursors in the mammalian olfactory epithelium. Development. 2010;137:2471–81.
Yamada T, Urano-Tashiro Y, Tanaka S, Akiyama H, Tashiro F. Involvement of crosstalk between Oct4 and Meis1a in neural cell fate decision. PLoS One. 2013;8, e56997.
Graham V, Khudyakov J, Ellis P, Pevny L. SOX2 functions to maintain neural progenitor identity. Neuron. 2003;39:749–65.
Adachi K, Suemori H. Yasuda Sy, Nakatsuji N, Kawase E. Role of SOX2 in maintaining pluripotency of human embryonic stem cells. Genes Cells. 2010;15:455–70.
Bass AJ, Watanabe H, Mermel CH, Yu S, Perner S, Verhaak RG, et al. SOX2 is an amplified lineage-survival oncogene in lung and esophageal squamous cell carcinomas. Nat Genet. 2009;41:1238–42.
Gen Y, Yasui K, Zen Y, Zen K, Dohi O, Endo M, et al. SOX2 identified as a target gene for the amplification at 3q26 that is frequently detected in esophageal squamous cell carcinoma. Cancer Genet Cytogenet. 2010;202:82–93.
Forghanifard MM, Khales SA, Javdani-Mallak A, Rad A, Farshchian M, Abbaszadegan MR. Stemness state regulators SALL4 and SOX2 are involved in progression and invasiveness of esophageal squamous cell carcinoma. Med Oncol. 2014;31:1–8.
Alonso MM, Diez-Valle R, Manterola L, Rubio A, Liu D, Cortes-Santiago N, et al. Genetic and epigenetic modifications of Sox2 contribute to the invasive phenotype of malignant gliomas. PLoS One. 2011;6, e26740.
Han X, Fang X, Lou X, Hua D, Ding W, Foltz G, et al. Silencing SOX2 induced mesenchymal-epithelial transition and its expression predicts liver and lymph node metastasis of CRC patients. PLoS One. 2012;7, e41335.
Li X, Xu Y, Chen Y, Chen S, Jia X, Sun T, et al. SOX2 promotes tumor metastasis by stimulating epithelial-to-mesenchymal transition via regulation of WNT/β-catenin signal network. Cancer Lett. 2013;336:379–89.
Bareiss PM, Paczulla A, Wang H, Schairer R, Wiehr S, Kohlhofer U, et al. SOX2 expression associates with stem cell state in human ovarian carcinoma. Cancer Res. 2013;73:5544–55.
Sobin LH, Gospodarowicz MK, Wittekind C. TNM classification of malignant tumours. 7th ed. Oxford: Wiley-Blackwell; 2009.
Shimada Y, Imamura M. Prognostic significance of cell culture in carcinoma of the oesophagus. Br J Surg. 1993;80:605–7.
Forghanifard MM, Gholamin M, Farshchian M, Moaven O, Memar B, Forghani MN, et al. Cancer-testis gene expression profiling in esophageal squamous cell carcinoma: Identification of specific tumor marker and potential targets for immunotherapy. Cancer Biol Ther. 2011;12:191–7.
Sinicrope FA, Ruan S, Cleary KR, Stephens LC, Lee JJ, Levin B. bcl-2 and p53 oncoprotein expression during colorectal tumorigenesis. Cancer Res. 1995;55:237–41.
Hu N, Clifford RJ, Yang HH, Wang C, Goldstein AM, Ding T, et al. Genome wide analysis of DNA copy number neutral loss of heterozygosity (CNNLOH) and its relation to gene expression in esophageal squamous cell carcinoma. BMC Genomics. 2010;11:576.
Kimchi ET, Posner MC, Park JO, Darga TE, Kocherginsky M, Karrison T, et al. Progression of Barrett's metaplasia to adenocarcinoma is associated with the suppression of the transcriptional programs of epidermal differentiation. Cancer Res. 2005;65:3146–54.
Su H, Hu N, Yang HH, Wang C, Takikita M, Wang Q-H, et al. Global gene expression profiling and validation in esophageal squamous cell carcinoma and its association with clinical phenotypes. Clin Cancer Res. 2011;17:2955–66.
Root DE, Hacohen N, Hahn WC, Lander ES, Sabatini DM. Genome-scale loss-of-function screening with a lentiviral RNAi library. Nat Methods. 2006;3:715–9.
Barde I, Salmon P, Trono D. Production and titration of lentiviral vectors. Curr Protoc Neurosci. 2010;4:4.21.
Mahmood T, Yang P-C. Western blot: Technique, theory, and trouble shooting. N Am J Med Sci. 2012;4:429–34.
Xiang P, Lo C, Argiropoulos B, Lai CB, Rouhi A, Imren S, et al. Identification of E74-like factor 1 (ELF1) as a transcriptional regulator of the Hox cofactor MEIS1. Exp Hematol. 2010;38:798–808.
Wong P, Iwasaki M, Somervaille TC, So CW, Cleary ML. Meis1 is an essential and rate-limiting regulator of MLL leukemia stem cell potential. Genes Dev. 2007;21:2762–74.
Okumura K, Saito M, Isogai E, Aoto Y, Hachiya T, Sakakibara Y, et al. Meis1 Regulates Epidermal Stem Cells and Is Required for Skin Tumorigenesis. PLoS One. 2014;9, e102111.
Geerts D, Revet I, Jorritsma G, Schilderink N, Versteeg R. MEIS homeobox genes in neuroblastoma. Cancer Lett. 2005;228:43–50.
Crijns APG, de Graeff P, Geerts D, Ten Hoor KA, Hollema H, Van Der Sluis T, et al. MEIS and PBX homeobox proteins in ovarian cancer. Eur J Cancer. 2007;43:2495–505.
Crist RC, Roth JJ, Waldman SA, Buchberg AM. A conserved tissue-specific homeodomain-less isoform of MEIS1 is downregulated in colorectal cancer. PLoS One. 2011;6, e23665.
Chen JL, Li J, Kiriluk KJ, Rosen AM, Paner GP, Antic T, et al. Deregulation of a hox protein regulatory network spanning prostate cancer initiation and progression. Clin Cancer Res. 2012;18:4291–302.
Lasa A, Carnicer M, Aventin A, Estivill C, Brunet S, Sierra J, et al. MEIS 1 expression is downregulated through promoter hypermethylation in AML1-ETO acute myeloid leukemias. Leukemia. 2004;18:1231–7.
Kondo Y, Shen L, Cheng AS, Ahmed S, Boumber Y, Charo C, et al. Gene silencing in cancer by histone H3 lysine 27 trimethylation independent of promoter DNA methylation. Nat Genet. 2008;40:741–50.
Beukers W, Hercegovac A, Vermeij M, Kandimalla R, Blok AC, van der Aa MM, et al. Hypermethylation of the polycomb group target gene PCDH7 in bladder tumors from patients of all ages. J Urol. 2013;190:311–6.
Acknowledgments
The authors gratefully acknowledge the scientific and technical support of colleagues at the Division of Human Genetic, Avicenna Research Institute (Mashhad University) and the Department of Biology, Ferdowsi University of Mashhad. In addition, we thank Dr. Heydar Parsaee (Department of Pharmacology, Mashhad University) and Dr. Thomas Mikeska (Department of Pathology, Peter MacCallum Cancer Centre, East Melbourne, Australia) for their technical assistances. This study was a MSc thesis and supported by a grant from Mashhad University of Medical Sciences (# 89751).
Conflict of interest
None.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study was approved beforehand by the local Ethics Committee. Informed, declared consent was obtained from all individual participants included in the study, and is on record. This article does not contain any studies with animals performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Abolfazl Rad and Moein Farshchian contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Figure 1
SOX2 mRNA detection in 50 ESCC and matched adjacent non-tumoral tissue samples. SOX2 mRNA expression is higher in tumor than in adjacent non-tumoral tissue, but this difference does not reach significance. SOX2 mRNA expression was determined with qRT-PCR, with GAPDH as the reference gene. Shown are the 2log median centered mean values, the error bars represent the SD. A two-sided t-test was used to assess the statistical difference between ESCC and adjacent non-tumoral tissue. (TIFF 329 kb)
Rights and permissions
About this article
Cite this article
Rad, A., Farshchian, M., Forghanifard, M.M. et al. Predicting the molecular role of MEIS1 in esophageal squamous cell carcinoma. Tumor Biol. 37, 1715–1725 (2016). https://doi.org/10.1007/s13277-015-3780-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-3780-9