Abstract
Competing endogenous RNAs (ceRNAs) are RNA transcripts which can communicate with each other by decreasing targeting concentration of micro-RNA (miRNA) with the derepression of other messenger RNAs (mRNAs) having the common miRNA response elements (MREs). Oncocers are ceRNAs taking crucial roles in oncogenic pathways processed in many types of cancer, and this study analyzes oncocer-mediated cross-talk by sponging microRNAs (miRNAs) in these pathways. While doing this, breast, liver, colon, prostate, gastric, lung, endometrium, thyroid and epithelial cancers and melanoma, rhabdomyosarcoma, glioblastoma, acute promyelocytic leukemia, retinoblastoma, and neuroblastoma were analyzed with respect to ceRNA-based carcinogenesis. This study defines, firstly, oncocers in the literature and contains all oncocer-related findings found up to now. Therefore, it will help to increase our comprehension about oncocer-mediated mechanisms. Via this study, a novel perspective would be produced to make clear cancer mechanisms and suggest novel approaches to regulate ceRNA networks via miRNA competition for cancer therapeutics.

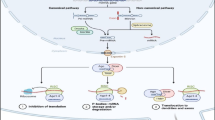
Multiple RNA transcripts have common MREs for the similar miRNA in their 3′-untranslated regions (3′-UTRs). Upregulation of ceRNAs rises the abundance of specific MREs and shifts the miRNA pool distribution, as a result, leading to the increased expression of target mRNA. The depot of genomic mutations and epigenetic alterations changing gene function and expression causes cancers. Herewith, genome-based somatic base-pair mutations, DNA copy number alterations, chromosomal translocation, also transcript fusions, alternative splicing are usually seen in cancer situations. Consequently, such cases causing changed UTR expression in transcripts influence the levels of MRE or present new MREs into the cells. Alterations in MREs of ceRNAs affect the capability of a specific mRNA transcript to attach or titrate miRNAs. As a result, the disturbed ceRNA network can lead to diseases and cancers. As a new term in RNA world, oncocers—the name for ceRNAs taking crucial roles in oncogenic pathways—are processed in many types of cancer, and oncocer-mediated cross-talk are analyzed by sponging miRNAs in these pathways.

Similar content being viewed by others
Abbreviations
- 3′-UTR:
-
3′-Untranslated region
- ARM:
-
Alveolar rhabdomyosarcoma
- ATXN3:
-
Ataxin 3
- ceRNA:
-
Competing endogenous RNA
- ceRNET:
-
ceRNA network
- EMT:
-
Epithelial-to-mesenchymal transition
- ERM:
-
Embryonic rhabdomyosarcoma
- ET:
-
Endometrial tumorspheres
- FGFR4:
-
Fibroblast growth factor receptor 4
- HDACi:
-
Histone deacetylase inhibitors
- HULC:
-
Highly upregulated in liver cancer
- IGFR1:
-
Insulin like growth factor 1 receptor
- lncRNA:
-
Long noncoding RNAs
- miRNA:
-
Micro RNA
- MRE:
-
MiRNA response element
- ncRNA:
-
Noncoding RNA
- PTCSC3:
-
Papillary thyroid carcinoma susceptibility candidate 3
- ZEB:
-
Zinc finger E-box-binding homeobox
References
Kataoka M, Wang D-Z. Non-coding RNAs including miRNAs and lncRNAs in cardiovascular biology and disease. Cells. 2014;3:883–98.
Kameswaran V, Kaestner KH. The missing lnc (RNA) between the pancreatic β-cell and diabetes. Front Genet. 2014;5:200.
Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136:215–33.
Esquela-Kerscher A, Slack FJ. Oncomirs—microRNAs with a role in cancer. Nat Rev Cancer. 2006;6:259–69.
Tan J, Marques A. The miRNA-mediated cross-talk between transcripts provides a novel layer of posttranscriptional regulation. Adv Genet. 2013;85:149–99.
Ergun S, Arman K, Temiz E, Bozgeyik İ, Yumrutaş Ö, et al. Expression patterns of miR-221 and its target caspase-3 in different cancer cell lines. Mol Biol Rep. 2014;41:5877–81.
Arman K, Ergün S, Temiz E, Öztuzcu S. The interrelationship between HER2 and CASP3/8 with apoptosis in different cancer cell lines. Mol Biol Rep. 2014;41(12):1–6.
Ergün S, Ulasli M, Igci Y, Igci M, Kırkbes S, et al. The association of the expression of miR-122-5p and its target ADAM10 with human breast cancer. Mol Biol Rep. 2015;42:497–505.
Costa V, Esposito R, Aprile M, Ciccodicola A. Non-coding RNA and pseudogenes in neurodegenerative diseases:“the (un) usual suspects”. Front Genet. 2012;3:231.
Ergün S, Öztuzcu S (2014) MiR-221: a critical player in apoptosis as a target of caspase-3. Cancer Cell Microenviron 1:10–14800/ccm. 14313.
Sümbül AT, Göğebakan B, Ergün S, Yengil E, Batmacı CY, Yaldız M, Sezer A, Aldemir Ö, Özyılkan Ö. miR-204-5p expression in colorectal cancer: an autophagy-associated gene. Tumor Biol. 2014;35(12):12713–9.
Ergun S, Oztuzcu S. Computational analysis of 3′ UTR Region of CASP3 with respect to miRSNPs and SNPs in targetting miRNAs. Comput Biol Chem. 2014;53PB:235–41.
Eades G, Zhang Y-S, Li Q-L, Xia J-X, Yao Y, et al. Long non-coding RNAs in stem cells and cancer. World J Clin Oncol. 2014;5:134.
Rossi MN, Antonangeli F. LncRNAs: new players in apoptosis control. Int J Cell Biol. 2014;2014, 473857.
Serviss JT, Johnsson P, Grandér D. An emerging role for long non-coding RNAs in cancer metastasis. Front Genet. 2014;5:234.
Khvorova A, Wolfson A. New competition in RNA regulation. Nat Biotechnol. 2012;30:58–9.
Salmena L, Poliseno L, Tay Y, Kats L, Pandolfi PP. A ceRNA hypothesis: the Rosetta Stone of a hidden RNA language? Cell. 2011;146:353–8.
Ala U, Karreth FA, Bosia C, Pagnani A, Taulli R, et al. Integrated transcriptional and competitive endogenous RNA networks are cross-regulated in permissive molecular environments. Proc Natl Acad Sci. 2013;110:7154–9.
Kalyana-Sundaram S, Kumar-Sinha C, Shankar S, Robinson DR, Wu Y-M, et al. Expressed pseudogenes in the transcriptional landscape of human cancers. Cell. 2012;149:1622–34.
Li L, Wang D, Xue M, Mi X, Liang Y, Wang P. 3′ UTR shortening identifies high-risk cancers with targeted dysregulation of the ceRNA network. Sci Rep. 2014;4:5406.
Karreth FA, Pandolfi PP. ceRNA cross-talk in cancer: when ce-bling rivalries go awry. Cancer Discov. 2013;3:1113–21.
Kartha RV, Subramanian S. Competing endogenous RNAs (ceRNAs): new entrants to the intricacies of gene regulation. Front Genet. 2014;5:8.
Sarver AL, Subramanian S. Competing endogenous RNA database. Bioinformation. 2012;8:731.
Karreth FA, Ala U, Provero P, Pandolfi PP (2014) Pseudogenes as competitive endogenous RNAs: target prediction and validation. Pseudogenes. Springer. pp. 199–212.
Cesana M, Daley GQ. Deciphering the rules of ceRNA networks. Proc Natl Acad Sci. 2013;110:7112–3.
Yang J, Li T, Gao C, Lv X, Liu K, et al. FOXO1 3′ UTR functions as a ceRNA in repressing the metastases of breast cancer cells via regulating miRNA activity. FEBS Lett. 2014;588:3218–24.
Sen R, Ghosal S, Das S, Balti S, Chakrabarti J. Competing endogenous RNA: the key to posttranscriptional regulation. Sci World J. 2014;2014, 896206.
Giza DE, Vasilescu C, Calin GA. MicroRNAs and ceRNAs: therapeutic implications of RNA networks. Expert Opin Biol Ther. 2014;14:1285–93.
Su X, Xing J, Wang Z, Chen L, Cui M, et al. microRNAs and ceRNAs: RNA networks in pathogenesis of cancer. Chin J Cancer Res. 2013;25:235.
Karreth FA, Tay Y, Perna D, Ala U, Tan SM, et al. In vivo identification of tumor-suppressive PTEN ceRNAs in an oncogenic BRAF-induced mouse model of melanoma. Cell. 2011;147:382–95.
Berger MF, Levin JZ, Vijayendran K, Sivachenko A, Adiconis X, et al. Integrative analysis of the melanoma transcriptome. Genome Res. 2010;20:413–27.
Tay Y, Kats L, Salmena L, Weiss D, Tan SM, et al. Coding-independent regulation of the tumor suppressor PTEN by competing endogenous mRNAs. Cell. 2011;147:344–57.
Sumazin P, Yang X, Chiu H-S, Chung W-J, Iyer A, et al. An extensive microRNA-mediated network of RNA-RNA interactions regulates established oncogenic pathways in glioblastoma. Cell. 2011;147:370–81.
Cesana M, Cacchiarelli D, Legnini I, Santini T, Sthandier O, et al. A long noncoding RNA controls muscle differentiation by functioning as a competing endogenous RNA. Cell. 2011;147:358–69.
Nie L, Wu H-J, Hsu J-M, Chang S-S, LaBaff AM, et al. Long non-coding RNAs: versatile master regulators of gene expression and crucial players in cancer. Am J Transl Res. 2012;4:127.
de Giorgio A, Krell J, Harding V, Stebbing J, Castellano L. Emerging roles of competing endogenous RNAs in cancer: insights from the regulation of PTEN. Mol Cell Biol. 2013;33:3976–82.
Zhou X, Liu J, Wang W. Construction and investigation of breast-cancer-specific ceRNA network based on the mRNA and miRNA expression data. IET Syst Biol. 2014;8(3):96–103.
Ferracin M, Bassi C, Pedriali M, Pagotto S, D’Abundo L, et al. miR-125b targets erythropoietin and its receptor and their expression correlates with metastatic potential and ERBB2/HER2 expression. Mol Cancer. 2013;12:130.
Paci P, Colombo T, Farina L. Computational analysis identifies a sponge interaction network between long non-coding RNAs and messenger RNAs in human breast cancer. BMC Syst Biol. 2014;8:83.
Hou P, Zhao Y, Li Z, Yao R, Ma M, et al. LincRNA-ROR induces epithelial-to-mesenchymal transition and contributes to breast cancer tumorigenesis and metastasis. Cell Death Dis. 2014;5:e1287.
Cha YH, Kim NH, Park C, Lee I, Kim HS, et al. MiRNA-34 intrinsically links p53 tumor suppressor and Wnt signaling. Cell Cycle. 2012;11:1273–81.
Fang L, Du WW, Yang X, Chen K, Ghanekar A, et al. Versican 3′-untranslated region (3′-UTR) functions as a ceRNA in inducing the development of hepatocellular carcinoma by regulating miRNA activity. FASEB J. 2013;27:907–19.
Zhao L, Li F, Taylor EW. Can tobacco use promote HCV-induced miR-122 hijacking and hepatocarcinogenesis? Med Hypotheses. 2013;80:131–3.
Pilyugin M, Irminger-Finger I. Long non-coding RNA and microRNAs might act in regulating the expression of BARD1 mRNAs. Int J Biochem Cell Biol. 2014;54:356–67.
Poliseno L, Salmena L, Zhang J, Carver B, Haveman WJ, et al. A coding-independent function of gene and pseudogene mRNAs regulates tumour biology. Nature. 2010;465:1033–8.
Shen K, Mao R, Ma L, Li Y, Qiu Y, et al. Post‐transcriptional regulation of the tumor suppressor miR‐139‐5p and a network of miR‐139‐5p‐mediated mRNA interactions in colorectal cancer. FEBS J. 2014;281:3609–24.
Gao X, Fu C, Lao X, Tan Z. Competing endogenous RNA regulation mechanism and its role in the development and progression of colorectal cancer. Chin J Gastrointest Surg. 2012;15:1318–21.
Tay Y, Rinn J, Pandolfi PP. The multilayered complexity of ceRNA crosstalk and competition. Nature. 2014;505:344–52.
Liu X-h, Sun M, Nie F-q, Ge Y-b, Zhang E-b, et al. lnc RNA HOTAIR functions as a competing endogenous RNA to regulate HER2 expression by sponging miR-331-3p in gastric cancer. Mol Cancer. 2014;13:92.
Xia T, Liao Q, Jiang X, Shao Y, Xiao B, et al. Long noncoding RNA associated-competing endogenous RNAs in gastric cancer. Sci Rep. 2014;4:6088. doi:10.1038/srep06088.
Tay Y, Karreth FA, Pandolfi PP. Aberrant ceRNA activity drives lung cancer. Cell Res. 2014;24(3):259–60.
Kumar MS, Armenteros-Monterroso E, East P, Chakravorty P, Matthews N, et al. HMGA2 functions as a competing endogenous RNA to promote lung cancer progression. Nature. 2014;505(7482):212–7.
Sacco J, Yau T, Darling S, Patel V, Liu H, et al. The deubiquitylase ataxin-3 restricts PTEN transcription in lung cancer cells. Oncogene. 2014;33(33):4265–72.
Zhou X, Gao Q, Wang J, Zhang X, Liu K, et al. Linc-RNA-RoR acts as a “sponge” against mediation of the differentiation of endometrial cancer stem cells by microRNA-145. Gynecol Oncol. 2014;133:333–9.
Kovalenko T, Sorokina A, Ozolinya L, Patrushev L. Methylation of the pseudogene PTENP1 5′-terminal region in endometrial cancer and hyperplasia. Russ J Bioorg Chem. 2013;39:397–405.
Fan M, Li X, Jiang W, Huang Y, Li J, et al. A long non-coding RNA, PTCSC3, as a tumor suppressor and a target of miRNAs in thyroid cancer cells. Exp Ther Med. 2013;5:1143–6.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ergun, S., Oztuzcu, S. Oncocers: ceRNA-mediated cross-talk by sponging miRNAs in oncogenic pathways. Tumor Biol. 36, 3129–3136 (2015). https://doi.org/10.1007/s13277-015-3346-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-3346-x