Abstract
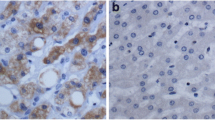
Armadillo repeat-containing protein 8 (Armc8) is a key factor to regulate cell membrane adhesion complex through promoting α-catenin degradation. However, its expression and function in human malignant tumors are largely unknown. Here, we present our study investigating Armc8 expression in tumor and non-tumor breast tissues including 45 normal epithelia, 53 lesions of hyperplasia with or without dysplasia, 22 benign tumors, and 92 carcinomas including 28 carcinomas in situ and 64 infiltrating carcinomas using immunohistochemistry (IHC) and Western blotting study. Armc8 expression was detected mainly in the cytoplasm with occasional membrane immunostaining. The positive rate of Armc8 expression in normal breast epithelia (8.9 %, four out of 45) was very low. No significant difference was found between Armc8 expression in usual ductal hyperplasia (UDH) (11.1 %, two out of 18), benign breast tumors including intraductal papilloma (10.0 %, one out of 10) and fibroadenoma (8.3 %, one out of 12), and normal breast epithelia (p > 0.05). Elevated expression of Armc8 was found in breast epithelia with dysplasia (24.0 %, six out of 25) compared to that in normal breast epithelia, UDH, and benign breast tumors (p < 0.05). Armc8 expression in breast carcinoma including breast carcinoma in situ (10/28, 35.7 %), infiltrating ductal carcinoma (60.7 %, 34/56), and infiltrating lobular carcinoma (50.0 %, 4/8) was higher than that in normal breast epithelia, UDH, benign breast tumors, and breast epithelia with dysplasia (p < 0.05). The highest expression of Armc8 was found in infiltrating breast carcinoma (59.4 %, 38/64) compared to all the other breast tissues. Higher Armc8 expression was found to be linked to lymph node metastasis and advanced tumor–node–metastasis (TNM) stages (III + IV) in infiltrating breast carcinoma (p < 0.05). We further confirmed Armc8 expression in breast epithelial cell line MCF10A and breast carcinoma cell lines including MCF-7, MDA-MB-231, and ZR751 using Western blotting and immunofluorescent study. These results indicate that the elevated expression of Armc8 may be involved in carcinogenesis including atypia-to-carcinoma progression and cancer development of breast carcinoma.






Similar content being viewed by others
References
Takeyuki S, Atsuhisa U, Nobuaki K, et al. Proteasome-dependent degradation of α-catenin is regulated by interaction with Armc8α. Biochem. 2008;411:581–91.
Nawrocki B, Polette M, Van Hengel J, et al. Cytoplasmic redistribution of E-cadherin-catenin adhesion complex is associated with down-regulated tyrosine phosphorylation of E-cadherin in human bronchopulmonary carcinomas. Am J Pathol. 1998;153(5):1521–30.
Sadot E, Conacci-Sorrell M, Zhurinsky J, et al. Regulation of S33/S37 phosphorylated β-Catenin in normal and transformed cells. J Cell Sci. 2002;115(13):2771–80.
Alan W, Clayton Y, Christopher R, et al. E-cadherin as an indicator of mesenchymal to epithelial reverting transitions during the metastatic seeding of disseminated carcinomas. Clin Exp Metastasis. 2008;25(6):621–8.
Somesh B, Suresh K. Molecular mechanisms controlling E-cadherin expression in breast cancer. Biochem Biophys Res Commun. 2009;384(1):6–11.
Liu Y, Wang Y, Zhang Y, et al. Abnormal expression of p120-catenin, E-cadherin, and small GTPases is significantly associated with malignant phenotype of human lung cancer. Lung Cancer. 2009;63(3):375–82.
Travis W, Muller-Hermelink H, Harris C. World Health Organization classification of tumors: pathology and genetics of tumors of the female reproduction system. Lyon: IARC Press; 2004.
Carlson R, Allred C, Anderson B, et al. NCCN guidelines for patients: breast cancer. 2010.
Fan C, Yu J, Liu Y, et al. Increased NDRG1 expression is associated with advanced T stages and poor vascularization in non-small cell lung cancer. Pathol Oncol Res. 2012;18(3):549–56.
Nobuaki K, Jun Y, Atsuhisa U, et al. RanBPM, muskelin, p48EMLP, p44CTLH, and the armadillo-repeat proteins ARMC8α and ARMC8β are components of the CTLH complex. Gene. 2007;396:236–47.
Koji T, Atsuhisa U, Takeyuki S, et al. Armadillo repeat containing 8α binds to HRS and promotes HRS interaction with ubiquitinated proteins. Open Biochem J. 2010;4:1–8.
Bai M, Agnantis N, Kamina S, et al. In vivo cell kinetics in breast carcinogenesis. Breast Cancer Res. 2011;3:276–83.
Arpino G, Laucirica R, Elledge R. Premalignant and in situ breast disease: biology and clinical implications. Ann Intern Med. 2005;143:446–57.
Steinman S, Wang J, Bourne P, et al. Expression of cytokeratin markers, ER-alpha, PR, HER-2/neu, and EGFR in pure ductal carcinoma in situ (DCIS) and DCIS with co-existing invasive ductal carcinoma (IDC) of the breast. Ann Clin Lab Sci. 2007;37:127–34.
Liu T, Niu Y, Yu Y, et al. Increased gamma-tubulin expression and P16INK4A promoter methylation occur together in preinvasive lesions and carcinomas of the breast. Ann Oncol. 2009;20:441–8.
Raynaud C, Hernandez J, Llorca F, et al. DNA damage repair and telomere length in normal breast, preneoplastic lesions, and invasive cancer. Am J Clin Oncol. 2010;33:341–5.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fan, C., Zhao, Y., Mao, X. et al. Armc8 expression was elevated during atypia-to-carcinoma progression and associated with cancer development of breast carcinoma. Tumor Biol. 35, 11337–11343 (2014). https://doi.org/10.1007/s13277-014-2473-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-014-2473-0