Abstract
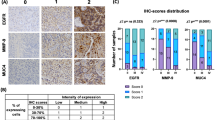
The objective of this study is to investigate the expression level of periostin in cancer stem cells as well as in the glioma tissues and the relationship between periostin expression and clinical and pathological characteristics and prognosis of gliomas. ESA+/CD133+/lin− tumor cells were selected by flow cytometry from glioma tissues, and the periostin expression in ESA+/CD133+/lin− tumor cells and non-ESA+/CD133+/lin− tumor cells was detected by quantitative real-time polymerase chain reaction (RT-PCR) and Western blot analysis. The expression status of periostin in glioma tissues was analyzed by immunohistochemistry staining, and the relationship between periostin and clinicopathological parameters of gliomas was determined. It showed that periostin is expressed higher in ESA+/CD133+/lin− tumor cells compared to non-ESA+/CD133+/lin− tumor cells in both mRNA and protein levels. One hundred eighteen (37.82 %) glioma patients were observed with highly expressed periostin protein in immunohistochemistry. Moreover, we observed that the expression of periostin protein was related to Karnofsky performance scale score (KPS), extent of resection, Ki67, and WHO grade of gliomas in universal analysis (P = 0.008, 0.045, 0.001, and 0.001, respectively). However, only WHO grade was identified to be related to periostin expression in gliomas after multivariate analysis. After survival analysis, the cases with highly expressed periostin protein attained a significantly poorer postoperative disease-specific survival and distant metastasis than those with none/low expressed periostin protein (P = 0.001 and 0.002). In the Cox regression test, KPS, extent of resection, Ki67, WHO grade, and periostin were detected as the independent prognostic factors (P = 0.008, 0.007, 0.032, 0.001, and 0.001, respectively). Periostin can be an important prognostic marker for gliomas, which may present a new therapeutic target for glioma patients.




Similar content being viewed by others
References
Ulukaya E, Frame FM, Cevatemre B, Pellacani D, Walker H, Mann VM, et al. Differential cytotoxic activity of a novel palladium-based compound on prostate cell lines, primary prostate epithelial cells and prostate stem cells. PLoS One. 2013;8(5):e64278.
Ren F, Sheng WQ, Du X. CD133: a cancer stem cells marker, is used in colorectal cancers. World J Gastroenterol. 2013;19(17):2603–11.
Shekhani MT, Jayanthy AS, Maddodi N, Setaluri V. Cancer stem cells and tumor transdifferentiation: implications for novel therapeutic strategies. Am J Stem Cells. 2013;2(1):52–61.
Liao WY, Liaw CC, Huang YC, Han HY, Hsu HW, Hwang SM, et al. Cyclohexylmethyl flavonoids suppress propagation of breast cancer stem cells via downregulation of NANOG. Evid Based Complement Alternat Med. 2013;2013:170261.
Chen SF, Lin YS, Jao SW, Chang YC, Liu CL, Lin YJ, et al. Pulmonary adenocarcinoma in malignant pleural effusion enriches cancer stem cell properties during metastatic cascade. PLoS One. 2013;8(5):e54659.
Nakada M, Nambu E, Furuyama N, Yoshida Y, Takino T, Hayashi Y, et al. Integrin α3 is overexpressed in glioma stem-like cells and promotes invasion. Br J Cancer. 2013;108(12):2516–24.
Tu SM. Cancer: a “stem-cell” disease? Cancer Cell Int. 2013;13(1):40.
Wei Y, Jiang Y, Zou F, Liu Y, Wang S, Xu N, et al. Activation of PI3K/Akt pathway by CD133-p85 interaction promotes tumorigenic capacity of glioma stem cells. Proc Natl Acad Sci U S A. 2013;110(17):6829–34.
Jin X, Jin X, Jung JE, Beck S, Kim H. Cell surface Nestin is a biomarker for glioma stem cells. Biochem Biophys Res Commun. 2013;433(4):496–501.
Dahlrot RH, Hermansen SK, Hansen S, Kristensen BW. What is the clinical value of cancer stem cell markers in gliomas? Int J Clin Exp Pathol. 2013;6(3):334–48.
Singh SK, Clarke ID, Terasaki M, Bonn VE, Hawkins C, Squire J, et al. Identification of a cancer stem cell in human brain tumors. Cancer Res. 2003;63(18):5821–8.
Liu CG, Lu Y, Wang BB, Zhang YJ, Zhang RS, Lu Y, et al. Clinical implications of stem cell gene Oct-4 expression in breast cancer. Ann Surg. 2011;253(6):1165–71.
Malanchi I, Santamaria-Martínez A, Susanto E, Peng H, Lehr HA, Delaloye JF, et al. Interactions between cancer stem cells and their niche govern metastatic colonization. Nature. 2011;481(7379):85.
Kyutoku M, Taniyama Y, Katsuragi N, Shimizu H, Kunugiza Y, Iekushi K, et al. Role of periostin in cancer progression and metastasis: inhibition of breast cancer progression and metastasis by anti-periostin antibody in a murine model. Int J Mol Med. 2011;28(2):181–6.
Xu D, Xu H, Ren Y, Liu C, Wang X, Zhang H, et al. Cancer stem cell-related gene periostin: a novel prognostic marker for breast cancer. PLoS One. 2012;7(10):e46670.
Listernick R, Charrow J, Greenwald M, Mets M. Natural history of optic pathway tumors in children with neurofibromatosis type 1: a longitudinal study. J Pediatr. 1994;125(1):63–6.
Caruso C, Carcaterra M, Donato V. Role of radiotherapy for high grade gliomas management. J Neurosurg Sci. 2013;57(2):163–9.
Ahmed AU, Auffinger B, Lesniak MS. Understanding glioma stem cells: rationale, clinical relevance and therapeutic strategies. Expert Rev Neurother. 2013;13(5):545–55.
Rath P, Lal B, Ajala O, Li Y, Xia S, Kim J, et al. In vivo c-Met pathway inhibition depletes human glioma xenografts of tumor-propagating stem-like cells. Transl Oncol. 2013;6(2):104–11.
Lehnus KS, Donovan LK, Huang X, Zhao N, Warr TJ, Pilkington GJ, et al. CD133 glycosylation is enhanced by hypoxia in cultured glioma stem cells. Int J Oncol. 2013;42(3):1011–7.
Liu X, Chen L, Jiang Z, Wang J, Su Z, Li G, et al. Malignant behavioral characteristics of CD133(+/−) glioblastoma cells from a Northern Chinese population. Exp Ther Med. 2013;5(1):65–72.
Pallini R, Ricci-Vitiani L, Banna GL, Signore M, Lombardi D, Todaro M, et al. Cancer stem cell analysis and clinical outcome in patients with glioblastoma multiforme. Clin Cancer Res. 2008;14(24):8205–12.
Wang H, Wang Y, Jiang C. Stromal protein periostin identified as a progression associated and prognostic biomarker in glioma via inducing an invasive and proliferative phenotype. Int J Oncol. 2013;42(5):1716–24.
Acknowledgments
This work was supported by the Science and Technology Development Project of Liaoning Province in China (Grant No. 2012225019IL-1).
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tian, B., Zhang, Y. & Zhang, J. Periostin is a new potential prognostic biomarker for glioma. Tumor Biol. 35, 5877–5883 (2014). https://doi.org/10.1007/s13277-014-1778-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-014-1778-3